Advanced Synthesis of Adrenoceptor Agonist Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value active pharmaceutical ingredients, particularly those targeting respiratory conditions such as asthma and chronic obstructive pulmonary disease (COPD). Patent CN1753874A introduces a groundbreaking process for preparing 5-[(R)-2-(5,6-diethyl-indan-2-ylamino)-1-hydroxy-ethyl]-8-hydroxy-(1H)-quinolin-2-one salts, which serve as potent beta-selective adrenoceptor agonists. This technical insight report analyzes the novel methodology that bypasses the isolation of unstable free bases, a critical bottleneck in traditional manufacturing. By leveraging specific salt formation and crystallization techniques, this process addresses long-standing challenges in regioselectivity and purification efficiency. For global procurement teams and R&D directors, understanding this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with enhanced supply chain continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
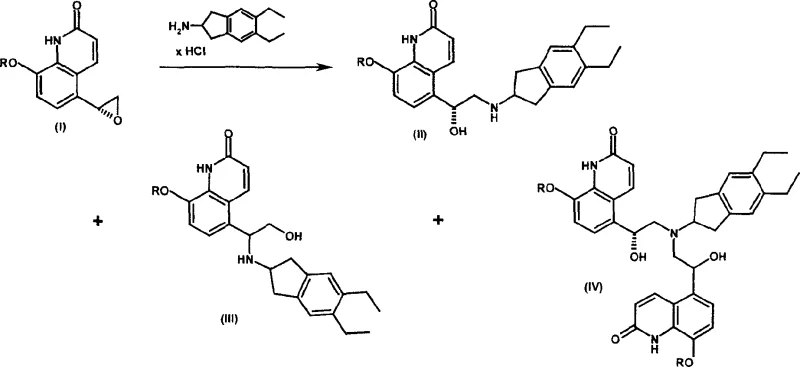
Traditional synthetic routes for complex quinolinone derivatives often suffer from significant regioselectivity issues during the amine opening of epoxide intermediates. In conventional processes, the reaction between the epoxide and the amine typically generates a mixture containing the desired intermediate alongside substantial amounts of regioisomers and dimers. Historically, purifying these mixtures has relied heavily on silica gel chromatography, a technique that is notoriously difficult to scale for industrial production. The reliance on chromatography introduces severe operational inefficiencies, including high solvent consumption, labor-intensive handling, and significant product loss during the purification stages. Furthermore, the instability of the free base form of the target molecule in organic solvents complicates isolation, leading to inconsistent yields and quality issues that can disrupt the supply of high-purity pharmaceutical intermediates.
The Novel Approach
The innovative process described in the patent data overcomes these limitations by introducing a strategic salt formation step that eliminates the need to isolate the unstable free base. Instead of attempting to purify the free base through chromatography, the method involves treating the crude reaction mixture directly with an acid to form a stable salt. This salt can then be purified through crystallization, a unit operation that is far more scalable and cost-effective than chromatography. This approach not only simplifies the workflow but also significantly enhances the overall yield by minimizing material loss during purification. By converting the intermediate into a crystalline salt form early in the process, manufacturers can achieve superior impurity profiles and ensure consistent quality, which is vital for cost reduction in API manufacturing and meeting stringent regulatory standards.
Mechanistic Insights into Chiral Reduction and Salt Formation
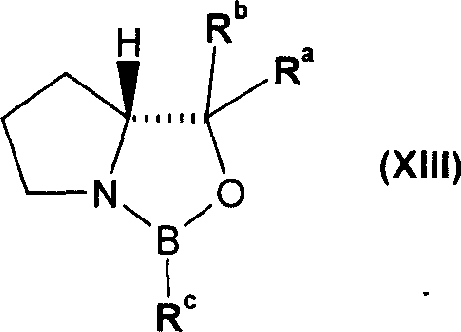
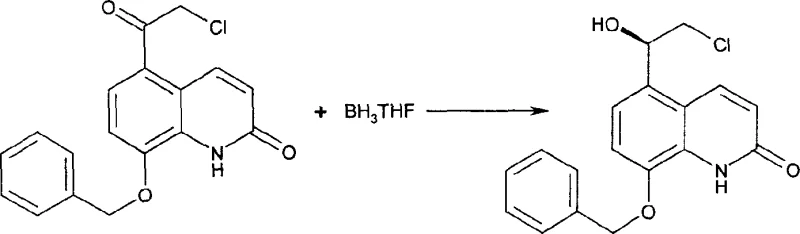
The core of this synthetic strategy lies in the precise control of stereochemistry and the management of reaction byproducts. The synthesis begins with the preparation of the chiral epoxide intermediate, often achieved through asymmetric reduction of a haloacetyl precursor using a chiral catalyst. The patent highlights the use of oxazaborolidine compounds, such as (R)-2-methyl-CBS-oxazaborole, which facilitate highly enantioselective reduction. This ensures that the resulting epoxide possesses the correct (R)-configuration required for the biological activity of the final adrenoceptor agonist. The subsequent ring-opening reaction with 2-amino-(5,6-diethyl)-indan is carefully controlled to minimize the formation of regioisomers, although some byproduct formation is inevitable. The key innovation is not preventing all byproducts but managing them effectively through downstream processing.
Impurity control is further enhanced by the specific choice of acid used for salt formation. The patent data indicates that different acids, such as benzoic acid, maleic acid, or fumaric acid, can be employed to form salts with varying solubility and crystallization properties. For instance, the benzoate salt of the protected intermediate can be crystallized to high purity, effectively excluding regioisomers and dimers from the crystal lattice. This selective crystallization is a powerful tool for purification that does not require complex separation equipment. Following the deprotection of the phenolic group, typically via hydrogenolysis if a benzyl protecting group is used, the final salt is formed using a pharmaceutically acceptable acid like maleic acid. This multi-step salt manipulation ensures that the final product meets the rigorous purity specifications demanded by the pharmaceutical industry.
How to Synthesize 5-[(R)-2-(5,6-diethyl-indan-2-ylamino)-1-hydroxy-ethyl]-8-hydroxy-(1H)-quinolin-2-one Salt Efficiently
Implementing this synthesis requires a thorough understanding of the reaction conditions and the physical properties of the intermediates. The process is designed to be robust, utilizing common solvents and reagents that are readily available in a commercial setting. The initial coupling reaction is typically performed at elevated temperatures to ensure complete conversion of the epoxide, followed by cooling to facilitate salt precipitation. The crystallization steps are critical and must be optimized to maximize yield and purity. Detailed standard operating procedures are essential for reproducibility, particularly regarding the seeding of crystals and the control of cooling rates. For technical teams looking to adopt this route, the following guide outlines the standardized synthesis steps derived from the patent examples.
- React 8-substituted oxy-5-(R)-oxiranyl-(1H)-quinolin-2-one with 2-amino-(5,6-diethyl)-indan to form a reaction mixture containing regioisomers.
- Treat the reaction mixture with an acid in the presence of a solvent to form the corresponding salt without isolating the free base.
- Isolate and crystallize the salt, remove the protecting group, and treat with acid to obtain the final quinolin-2-one salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for procurement managers and supply chain heads focused on cost efficiency and reliability. The elimination of silica gel chromatography is a major driver for cost reduction in API manufacturing, as it removes the need for large volumes of chromatographic solvents and the associated waste disposal costs. This simplification of the process flow also reduces the manufacturing cycle time, allowing for faster turnaround and improved responsiveness to market demand. Additionally, the use of crystallization for purification enhances the scalability of the process, making it suitable for commercial scale-up of complex pharmaceutical intermediates without the technical risks associated with chromatographic scaling.
- Cost Reduction in Manufacturing: The process significantly lowers production costs by replacing expensive and labor-intensive chromatographic purification with efficient crystallization steps. By avoiding the isolation of the unstable free base, the method reduces material loss and minimizes the need for reprocessing, leading to substantial cost savings. The use of readily available reagents and solvents further contributes to a more economical manufacturing profile, ensuring that the final intermediate is cost-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain reliability by reducing the risk of production delays caused by purification bottlenecks. The ability to produce high-purity intermediates consistently ensures a steady supply of materials for downstream API synthesis. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing pharmaceutical companies to maintain their production schedules and meet patient demand without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, as crystallization is a standard unit operation in the chemical industry that can be easily transferred from pilot to production scale. Furthermore, the reduction in solvent usage and waste generation aligns with environmental compliance goals, making the process more sustainable. This environmental advantage is increasingly important for companies seeking to minimize their carbon footprint and adhere to strict regulatory standards regarding waste management and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis process. These answers are derived from the detailed patent specifications and are intended to provide clarity on the feasibility and advantages of the technology. Understanding these aspects is critical for decision-makers evaluating this route for potential integration into their supply chain.
Q: How does this process improve purity compared to conventional methods?
A: The process utilizes selective salt formation and crystallization to purify the intermediate, avoiding the need for silica gel chromatography which often leads to yield loss and impurity retention.
Q: What are the scalability advantages of this synthetic route?
A: By eliminating chromatographic purification and using robust crystallization steps, the process is highly amenable to large-scale manufacturing, reducing solvent consumption and operational complexity.
Q: Is the chiral catalyst used in this process commercially viable?
A: Yes, the process employs oxazaborolidine catalysts which are well-established in industrial asymmetric synthesis, ensuring consistent enantiomeric purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-[(R)-2-(5,6-diethyl-indan-2-ylamino)-1-hydroxy-ethyl]-8-hydroxy-(1H)-quinolin-2-one Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for high-value pharmaceutical intermediates. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the one described in CN1753874A can be successfully translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to handle chiral synthesis and precise salt formation makes us an ideal partner for your respiratory drug development projects.
We invite you to collaborate with us to optimize your supply chain and achieve your cost targets. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can support your commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
