Advanced Synthesis of N-(2-Amino-4,6-Dichloropyrimidin-5-Yl) Formamide for Antiviral Intermediates
Advanced Synthesis of N-(2-Amino-4,6-Dichloropyrimidin-5-Yl) Formamide for Antiviral Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for constructing complex heterocyclic scaffolds, particularly those serving as critical backbones for antiviral therapeutics. Patent CN1113237A introduces a groundbreaking methodology for producing N-(2-amino-4,6-dichloropyrimidin-5-yl) formamide, a novel pyrimidine derivative that addresses significant bottlenecks in nucleoside analog synthesis. This specific compound represents a pivotal advancement because it serves as a highly reactive precursor that can be efficiently transformed into nucleotide derivatives, a capability often lacking in traditional protected intermediates. By leveraging a unique sequence of cyclization, chlorination, and selective hydrolysis, this technology enables the production of high-purity intermediates essential for next-generation antiviral drugs. As a reliable pharmaceutical intermediate supplier, understanding the structural nuances of this molecule is crucial for optimizing downstream coupling reactions.
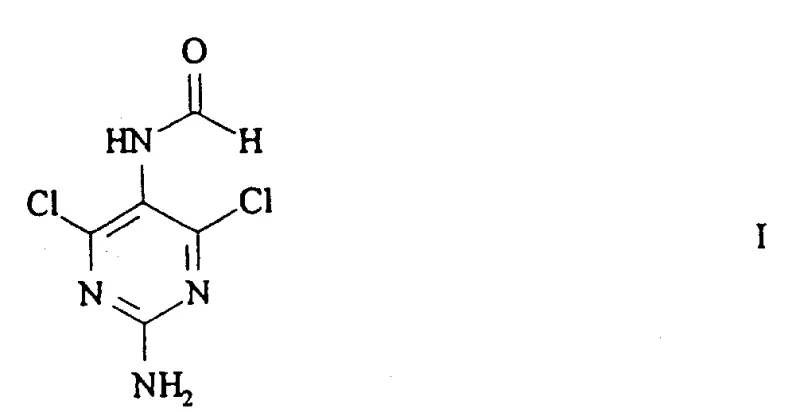
The strategic value of this innovation lies in its ability to bypass the limitations of prior art, specifically the difficulties associated with converting N-5 protected diamino-dichloropyrimidines into active pharmaceutical ingredients. The disclosed method not only improves overall yield but also simplifies the purification profile, reducing the burden on quality control laboratories. For research and development teams focused on antiviral pipelines, adopting this synthetic route could significantly accelerate lead optimization phases by providing a more versatile building block. Furthermore, the economic implications of this process are substantial, as it utilizes readily available starting materials and avoids the need for expensive transition metal catalysts or cryogenic conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of antiviral nucleoside precursors has relied heavily on N-5 protected 2,5-diamino-4,6-dichloropyrimidine intermediates, as documented in earlier patents such as EP-A0552758. While these compounds served their purpose, they present a formidable challenge in downstream processing: they are notoriously difficult to convert into the corresponding nucleotide derivatives required for final drug assembly. This recalcitrance often forces process chemists to employ harsh deprotection conditions or multi-step activation sequences, which inevitably degrade overall yield and introduce complex impurity profiles. Additionally, the removal of protecting groups from the N-5 position can generate hazardous waste streams and require extensive chromatographic purification, driving up both the cost of goods sold and the environmental footprint of the manufacturing process. These inefficiencies create a supply chain vulnerability, as any delay in intermediate conversion can stall the entire production timeline for critical antiviral medications.
The Novel Approach
In stark contrast, the methodology outlined in patent CN1113237A offers a streamlined solution by introducing a formamide moiety that acts as a superior leaving group and activating handle. This novel approach facilitates a direct and high-productivity conversion into nucleotide derivatives, effectively eliminating the stubborn conversion barriers associated with legacy intermediates. The process achieves this through a clever manipulation of reaction conditions, utilizing common reagents like phosphorus oxychloride and formamides to install the necessary functionality in fewer steps. By integrating the formyl group early in the synthesis, the route ensures that the final intermediate possesses the exact electronic and steric properties needed for efficient nucleophilic substitution. This paradigm shift not only enhances the chemical efficiency of the synthesis but also aligns perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing by minimizing unit operations and solvent consumption.
Mechanistic Insights into Pyrimidine Cyclization and Chlorination
The core of this synthetic strategy involves a sophisticated three-step cascade that begins with the construction of the pyrimidine ring itself. The initial phase utilizes amidomalonic acid ester, or its salts, which undergoes cyclization with guanidine or its salts in the presence of a strong base such as sodium methylate. This reaction, typically conducted under reflux in alcoholic solvents, generates a 2,5-diamino-4,6-dihydroxypyrimidine intermediate. The choice of base and solvent is critical here, as it dictates the rate of enolate formation and subsequent nucleophilic attack on the guanidine carbon. Following cyclization, the process moves to a critical chlorination step where the hydroxyl groups are replaced by chlorine atoms. This transformation is mediated by phosphorus oxychloride in the presence of an acid amide, such as N,N-dimethylformamide (DMF), which acts as a Vilsmeier-Haack type reagent to activate the chlorinating agent.

During the chlorination stage, the reaction proceeds through the formation of a highly reactive iminium species, which facilitates the substitution of the hydroxyl groups at the 4 and 6 positions of the pyrimidine ring. The patent specifies that the molar ratio of chlorinating reagent to acid amide is carefully controlled, typically ranging from 1:0.55 to 1:10, to ensure complete conversion while minimizing side reactions. The resulting intermediate, represented generally by Formula V in the patent data, contains the essential dichloro motif required for subsequent nucleophilic displacements. Crucially, the process allows for the isolation of specific precursors or the direct progression to the final formamide depending on the desired substitution pattern. The final step involves a controlled hydrolysis or reaction with aqueous carboxylic acids, which selectively modifies the substituents to yield the target N-(2-amino-4,6-dichloropyrimidin-5-yl) formamide. This mechanistic precision ensures that the amino group at the 2-position remains intact while the 4 and 6 positions are activated for future coupling.
How to Synthesize N-(2-Amino-4,6-Dichloropyrimidin-5-Yl) Formamide Efficiently
Executing this synthesis requires strict adherence to the reaction parameters defined in the patent to maximize yield and purity. The process is designed to be robust, tolerating slight variations in temperature while maintaining high selectivity for the desired isomer. Operators must pay close attention to the stoichiometry of the chlorinating agents and the timing of the quenching steps to prevent over-chlorination or degradation of the sensitive formamide group. The detailed standardized synthesis steps below outline the precise conditions for scaling this reaction from laboratory benchtop to pilot plant volumes.
- Cyclize amidomalonic acid ester with guanidine salt in the presence of alkali to form the diaminodihydroxypyrimidine intermediate.
- Perform chlorination using phosphorus oxychloride and a formamide derivative (like DMF) to generate the dichloro-pyrimidine intermediate.
- React the chlorinated intermediate with aqueous carboxylic acid to yield the final N-(2-amino-4,6-dichloropyrimidin-5-yl) formamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend far beyond simple chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials; the key starting materials, such as amidomalonic acid esters and guanidine salts, are commodity chemicals available from multiple global sources, reducing the risk of single-supplier dependency. Furthermore, the elimination of difficult-to-remove protecting groups means that the purification process is significantly less resource-intensive, translating directly into lower operational expenditures. By avoiding the need for specialized catalysts or extreme low-temperature reactors, facilities can utilize existing general-purpose equipment, thereby accelerating the timeline for technology transfer and commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the reduction in unit operations and the avoidance of expensive deprotection reagents. Traditional routes often require distinct steps to install and subsequently remove protecting groups, each adding labor, solvent, and energy costs. By utilizing a formamide group that serves a dual purpose as both a protecting and activating group, this novel method collapses multiple steps into a more efficient sequence. This consolidation leads to substantial cost savings in terms of raw material consumption and waste disposal, as fewer byproducts are generated that require neutralization or incineration. Additionally, the higher overall yield means that less starting material is required per kilogram of final product, further enhancing the cost competitiveness of the manufactured intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical sector, and this synthesis route bolsters reliability through the use of stable, non-hazardous reagents. Unlike processes relying on unstable diazo compounds or pyrophoric reagents, the chemicals used here—such as phosphorus oxychloride and common organic solvents—are well-understood and easily sourced. This stability reduces the likelihood of production stoppages due to reagent shortages or safety incidents. Moreover, the robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuations in demand for antiviral APIs. The ability to produce high-purity intermediates consistently ensures that downstream customers face fewer delays due to quality failures or specification deviations.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is inherently greener than many legacy alternatives. The reaction solvents, primarily alcohols and toluene, are readily recyclable, and the absence of heavy metal catalysts simplifies the wastewater treatment profile. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental compliance burden on manufacturing sites. Scalability is further supported by the fact that the exothermic profiles of the reactions are manageable with standard cooling systems, allowing for safe operation in large-scale reactors. The potential to isolate stable intermediates also provides flexibility, allowing manufacturers to stockpile key precursors and decouple the production stages, thereby smoothing out production flows and ensuring a steady supply of the final active intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within patent CN1113237A, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing portfolios.
Q: Why is this new pyrimidine derivative superior to existing N-5 protected intermediates?
A: Unlike traditional N-5 protected 2,5-diamino-4,6-dichloropyrimidines which are difficult to convert into corresponding nucleotide derivatives, this novel formamide derivative allows for high-productivity conversion, streamlining the synthesis of antiviral nucleosides.
Q: What are the key reagents required for the chlorination step?
A: The process utilizes phosphorus oxychloride as the primary chlorinating agent in combination with acid amides such as N,N-dimethylformamide (DMF) or N-formylpiperidine, ensuring efficient substitution at the 4 and 6 positions.
Q: Can this process be scaled for commercial production?
A: Yes, the reaction conditions involve standard reflux temperatures and common solvents like toluene or alcohols, making the pathway highly adaptable for large-scale manufacturing without requiring exotic catalysts or extreme pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(2-Amino-4,6-Dichloropyrimidin-5-Yl) Formamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against viral diseases. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis modifications or bulk supply of the standard intermediate, our infrastructure is designed to support your project from early-stage development through to full commercialization.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your specific manufacturing requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this superior intermediate. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your antiviral drug development pipeline, ensuring a partnership built on technical excellence and mutual success.
