Advanced Synthesis of Piperidine Carboxamide Derivatives for Serotonin Antagonists
Introduction to Novel Synthetic Pathways
The pharmaceutical industry constantly seeks robust methodologies for constructing complex heterocyclic scaffolds essential for modern therapeutics. Patent CN1126740C introduces a transformative approach for the preparation of piperidine carboxamide derivatives, which serve as critical intermediates for potent serotonin antagonists and antiplatelet agents. This technology addresses long-standing safety and efficiency challenges associated with traditional alkylation routes by leveraging the unique reactivity of 2-oxazoline compounds. By shifting the synthetic paradigm from hazardous alkyl halides to stable cyclic imino ethers, this method ensures a safer operational environment while maintaining high chemical fidelity. The process is particularly relevant for the commercial scale-up of complex pharmaceutical intermediates where impurity profiles and operator safety are paramount concerns for regulatory compliance.
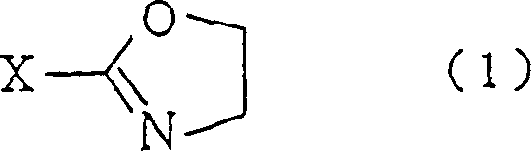
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ethyl-linked piperidine amides relied heavily on the nucleophilic substitution of piperidine derivatives with 2-aminoethyl bromide or its protected variants. As explicitly noted in the background art of the patent, 2-aminoethyl bromide is a highly toxic substance that presents severe handling hazards in an industrial setting. Furthermore, under basic reaction conditions often required for these alkylations, there is a significant risk of intramolecular cyclization leading to the formation of aziridine, a known carcinogen. This side reaction not only compromises the purity of the final active pharmaceutical ingredient but also necessitates expensive and complex purification steps to remove genotoxic impurities. Consequently, conventional methods are increasingly viewed as unsustainable for large-scale manufacturing due to their poor atom economy and stringent environmental, health, and safety (EHS) requirements.
The Novel Approach
The innovative methodology disclosed in CN1126740C circumvents these dangers by utilizing 2-oxazoline compounds as stable acylating equivalents. In this acid-catalyzed process, the 2-oxazoline ring undergoes a controlled ring-opening reaction upon exposure to a piperidine derivative, directly forming the desired amide bond without generating toxic halide by-products. This route allows for the use of mild proton acids like p-toluenesulfonic acid, which effectively suppresses side reactions and drives the equilibrium toward the product. The versatility of this approach is further enhanced by the ability to use commercially available 2-oxazolines or easily synthesized variants, providing a flexible platform for generating diverse libraries of analogs. This strategic shift from alkylation to ring-opening represents a significant advancement in process chemistry, aligning with green chemistry principles by eliminating hazardous reagents.
Mechanistic Insights into Acid-Catalyzed Oxazoline Ring Opening
The core of this technology lies in the electrophilic activation of the 2-oxazoline ring system. Under acidic conditions, the nitrogen atom of the oxazoline ring is protonated, significantly increasing the electrophilicity of the adjacent carbon atoms, particularly the C2 position. The nucleophilic nitrogen of the piperidine derivative then attacks this activated center, initiating the ring-opening cascade. The patent specifies that the reaction temperature generally ranges from 50°C to 140°C, with an optimal window between 80°C and 125°C to balance reaction kinetics and solvent stability. Solvent selection is critical; aromatic hydrocarbons like toluene or xylene are preferred because they facilitate the necessary thermal energy without promoting the hydrolytic decomposition of the oxazoline, which can occur in protic solvents like water or alcohols. This precise control over reaction parameters ensures a clean conversion profile.
Impurity control is inherently built into the mechanism through the choice of catalyst and stoichiometry. The patent highlights that using p-toluenesulfonic acid monohydrate at a loading of 10 to 17 mol% relative to the oxazoline provides the best balance between reaction rate and by-product suppression. Excessive acid loading (>100 mol%) is noted to be uneconomical and detrimental to yield, likely due to over-protonation or degradation of sensitive functional groups on the piperidine scaffold. Conversely, insufficient acid (<5 mol%) leads to incomplete conversion. This mechanistic understanding allows process chemists to fine-tune the reaction for maximum purity, ensuring that the resulting high-purity pharmaceutical intermediates meet the rigorous specifications required for downstream drug substance manufacturing.
How to Synthesize Piperidine Carboxamide Derivatives Efficiently
Implementing this synthesis requires careful attention to the order of addition and thermal management to maximize yield and safety. The process typically involves heating the 2-oxazoline and piperidine derivative in a non-polar solvent with the acid catalyst under reflux conditions. Following the reaction, the mixture is worked up via aqueous extraction to remove the acid catalyst, followed by crystallization to isolate the pure product. For detailed operational parameters and specific stoichiometric ratios tailored to your specific substrate, please refer to the standardized protocol below.
- React a 2-oxazoline compound with a piperidine derivative in the presence of an acid catalyst such as p-toluenesulfonic acid.
- Maintain reaction temperature between 50°C and 140°C, preferably using aromatic hydrocarbons like toluene as solvent.
- Isolate the product through extraction, concentration, and crystallization to obtain high-purity piperidine carboxamide derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this oxazoline-based synthetic route offers profound benefits for supply chain stability and cost management in pharmaceutical manufacturing. By eliminating the need for toxic alkyl halides, facilities can significantly reduce the costs associated with hazardous waste disposal and specialized containment equipment. The use of stable, commercially available starting materials mitigates the risk of supply disruptions often caused by the strict regulatory controls on toxic precursors. Furthermore, the high yields reported in the patent examples suggest a more efficient use of raw materials, directly contributing to cost reduction in API manufacturing. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures and ensuring consistent delivery timelines for critical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous alkylating agents like 2-aminoethyl bromide removes the need for complex quenching and purification steps required to remove genotoxic impurities. This simplification of the downstream processing workflow leads to substantial cost savings by reducing solvent consumption and processing time. Additionally, the ability to use cheaper, bulk-grade solvents like toluene instead of specialized polar aprotic solvents further lowers the overall production cost structure.
- Enhanced Supply Chain Reliability: The starting materials, specifically 2-oxazoline derivatives, are either commercially available or can be synthesized from abundant commodity chemicals like amino alcohols and nitriles. This reduces dependency on single-source suppliers of controlled toxic substances. The improved safety profile of the reagents also simplifies logistics and storage requirements, allowing for larger inventory buffers without triggering excessive regulatory scrutiny, thereby enhancing supply continuity.
- Scalability and Environmental Compliance: The reaction operates effectively under atmospheric pressure in standard glass-lined or stainless steel reactors, avoiding the need for high-pressure vessels often required for gas-phase alkylations. The absence of halogenated by-products simplifies effluent treatment, making it easier to meet increasingly stringent environmental discharge standards. This ease of scale-up from kilogram to multi-ton quantities ensures that the process remains viable and compliant as production volumes increase to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. These insights are derived directly from the experimental data and claims within the patent documentation to assist technical teams in evaluating feasibility.
Q: Why is the 2-oxazoline method preferred over traditional alkylation?
A: Traditional methods using 2-aminoethyl bromide pose significant toxicity risks and potential carcinogen (aziridine) formation. The 2-oxazoline route operates under milder, safer conditions with superior yield control.
Q: What catalysts are optimal for this ring-opening reaction?
A: While Lewis acids like BF3·Et2O work, proton acids such as p-toluenesulfonic acid monohydrate are preferred for suppressing by-products and maximizing yield, typically used at 10-17 mol%.
Q: Can this intermediate be further functionalized?
A: Yes, the resulting piperidine carboxamide can be hydrolyzed to a free amine intermediate, allowing for subsequent coupling with various carboxylic acid derivatives to create diverse serotonin antagonists.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperidine Carboxamide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for complex pharmaceutical intermediates. Our technical team has extensively analyzed the methodology described in CN1126740C and possesses the expertise to implement this acid-catalyzed oxazoline ring-opening strategy at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the absence of genotoxic impurities often associated with older synthetic methods.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for serotonin antagonist precursors. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis specific to your project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your drug development timeline.
