Advanced One-Pot Synthesis of N-Boc-N-methyl-4-aminostyrene for High-Volume Pharmaceutical Manufacturing
Introduction to Patent CN111499544B: A Breakthrough in Diagnostic Intermediate Synthesis
The pharmaceutical industry is constantly seeking more efficient pathways to produce critical intermediates for diagnostic imaging, particularly for positron emission tomography (PET) agents used in neurology. Patent CN111499544B introduces a transformative synthetic method for preparing N-Boc-N-methyl-4-aminostyrene, a pivotal precursor for the Alzheimer's disease diagnostic reagent 18F-AV-45 and its analogues. This innovation addresses the longstanding challenges associated with the traditional synthesis of this valuable building block, which has historically relied on expensive and unstable starting materials. By shifting the synthetic strategy to a one-pot methylation and elimination protocol, this technology offers a robust solution for reliable pharmaceutical intermediate supplier networks aiming to secure stable supply chains for high-demand diagnostic tools. The method leverages inexpensive, commercially available substrates bearing various leaving groups, thereby circumventing the bottlenecks of previous routes.
Furthermore, the technical significance of this patent extends beyond mere cost savings; it represents a fundamental improvement in process safety and operational simplicity. The ability to perform both N-methylation and vinyl formation in a single reactor vessel drastically reduces the processing time and solvent consumption typically associated with multi-step syntheses. For R&D teams focused on impurity control, this streamlined approach minimizes the generation of side products that often arise during intermediate isolation steps. As the demand for amyloid imaging agents continues to grow globally, adopting such advanced manufacturing techniques becomes essential for maintaining competitiveness. This report analyzes the technical merits and commercial implications of this novel route, providing actionable insights for decision-makers in the fine chemical and pharmaceutical sectors.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-Boc-N-methyl-4-aminostyrene has been fraught with significant economic and technical hurdles that hinder large-scale production efficiency. Traditional routes, as cited in prior art such as CN102271716A and CN102432529A, typically commence with 4-aminostyrene as the primary raw material. This starting compound is notoriously difficult to handle due to its inherent instability and tendency to polymerize, which necessitates stringent storage conditions and often results in substantial material loss during processing. Moreover, the commercial availability of high-purity 4-aminostyrene is limited, driving up procurement costs and creating supply chain vulnerabilities for manufacturers. The conventional process requires an initial protection step of the amino group followed by a separate methylation reaction, effectively doubling the number of unit operations and increasing the overall environmental footprint through higher solvent and reagent usage.
In addition to the raw material issues, the multi-step nature of the legacy synthesis introduces compounding yield losses at each stage. Every isolation and purification step carries the risk of product degradation and the accumulation of difficult-to-remove impurities, which is particularly critical for intermediates destined for radiopharmaceutical applications where purity standards are exceptionally high. The reliance on specialized, high-cost precursors means that cost reduction in pharmaceutical intermediate manufacturing is nearly impossible to achieve without a fundamental redesign of the synthetic pathway. Consequently, producers have been forced to accept low overall yields and high production costs, which ultimately translates to higher prices for the final diagnostic agents. These inefficiencies highlight the urgent need for a more direct and economically viable synthetic strategy that can bypass these inherent limitations.
The Novel Approach
The methodology disclosed in patent CN111499544B presents a paradigm shift by utilizing a one-pot tandem reaction that simultaneously achieves N-methylation and elimination to form the styrene double bond. Instead of starting with the unstable aminostyrene, this innovative route begins with stable, Boc-protected phenethyl derivatives equipped with a leaving group (LG) such as tosylate, mesylate, triflate, or iodide. This strategic change allows manufacturers to utilize cheap and abundant raw materials that are easy to store and handle, effectively eliminating the supply risks associated with 4-aminostyrene. The reaction proceeds by treating the substrate with a strong base and a methylating agent in a dry organic solvent, facilitating the methylation of the nitrogen atom followed immediately by an E2 elimination to generate the vinyl group. This consolidation of two distinct chemical transformations into a single operational step significantly simplifies the workflow and reduces the requirement for intermediate purification.
From a process engineering perspective, this novel approach offers remarkable flexibility and robustness. The patent demonstrates that a variety of leaving groups can be employed successfully, giving process chemists the freedom to optimize based on local raw material availability and cost structures. For instance, substrates with tosylate or mesylate leaving groups are often derived from inexpensive alcohols, while iodide substrates offer high reactivity. The use of common bases like sodium hydride or potassium tert-butoxide in polar aprotic solvents such as DMF ensures that the reaction conditions are easily replicable on a large scale. By achieving yields exceeding 90% in a single pot, this method not only maximizes atom economy but also drastically cuts down on waste generation. This represents a significant advancement for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials with a reduced environmental impact and lower total cost of ownership.
Mechanistic Insights into One-Pot Methylation and Elimination
The core chemical innovation of this patent lies in the precise orchestration of nucleophilic substitution and elimination reactions within a single reaction vessel. The mechanism initiates with the deprotonation of the Boc-protected amine by a strong base, such as sodium hydride (NaH), generating a nucleophilic nitrogen anion. This anion subsequently attacks the methylating agent, typically methyl iodide (CH3I) or dimethyl sulfate, in an SN2 fashion to install the N-methyl group. Crucially, the reaction conditions are tuned such that the base remains active or is present in sufficient excess to facilitate the subsequent elimination step. Once the N-methyl group is installed, the base abstracts a proton from the beta-carbon relative to the leaving group. This abstraction triggers a concerted E2 elimination mechanism, where the electrons from the C-H bond collapse to form the carbon-carbon double bond, expelling the leaving group (LG) simultaneously. This tandem sequence is highly efficient because the intermediate N-methyl species does not need to be isolated; it reacts in situ to form the final styrene product.
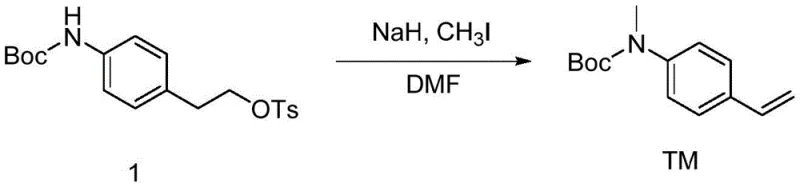
Controlling the impurity profile in this one-pot system is achieved through careful management of reaction temperature and stoichiometry. The patent specifies an initial cooling phase, typically in an ice bath at 0°C, during the addition of reagents. This thermal control is vital to suppress potential side reactions, such as over-alkylation or premature elimination before methylation is complete. By maintaining the temperature at 0°C initially and then allowing the reaction to warm to room temperature overnight, the process ensures complete conversion while minimizing the formation of polymeric byproducts or decomposition of the sensitive styrene moiety. Furthermore, the choice of solvent plays a critical role; polar aprotic solvents like DMF stabilize the ionic intermediates and enhance the nucleophilicity of the amine anion, promoting the desired pathway over competing hydrolysis or substitution reactions. This mechanistic understanding allows for the production of high-purity pharmaceutical intermediates that meet the rigorous specifications required for downstream radiolabeling processes.
How to Synthesize N-Boc-N-methyl-4-aminostyrene Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure safety and reproducibility. The process begins with the preparation of a dry reaction environment under an inert atmosphere, typically nitrogen, to prevent moisture from quenching the strong base. A suspension of sodium hydride is prepared in dry DMF and cooled to 0°C before the slow addition of the substrate and methylating agent. This controlled addition is critical to manage the exotherm and maintain the selectivity of the reaction. Following the addition, the mixture is stirred at low temperature to allow methylation to proceed, followed by a period at room temperature to drive the elimination to completion. The detailed standardized synthesis steps below outline the precise quantities and conditions validated in the patent examples, serving as a foundational guide for process optimization.
- Under nitrogen protection, suspend sodium hydride (60%) in dry DMF and cool the mixture to 0°C using an ice bath.
- Dropwise add a solution containing the substrate (e.g., Boc-protected phenethyl tosylate) and methyl iodide in DMF while maintaining the temperature at 0°C.
- Stir the reaction mixture at 0°C for 1 hour, then allow it to warm to room temperature and stir overnight to complete methylation and elimination.
- Quench the reaction with distilled water, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis method offers compelling strategic advantages that directly impact the bottom line and operational resilience. The most immediate benefit is the drastic reduction in raw material costs. By replacing expensive and scarce 4-aminostyrene with inexpensive, stable Boc-protected phenethyl derivatives, manufacturers can secure a much more predictable and cost-effective supply of starting materials. These precursors are commodity chemicals that can be sourced from multiple vendors, reducing dependency on single-source suppliers and mitigating the risk of price volatility. Additionally, the elimination of intermediate isolation steps means that less solvent is consumed, and less waste is generated, leading to significant savings in waste disposal and solvent recovery costs. This aligns perfectly with modern green chemistry initiatives and helps companies meet their sustainability goals without compromising on output.
- Cost Reduction in Manufacturing: The consolidation of methylation and elimination into a single pot eliminates the need for separate reactors, filtration units, and drying equipment for intermediate steps. This reduction in unit operations translates directly to lower capital expenditure (CAPEX) requirements for new production lines and reduced operational expenditure (OPEX) for existing facilities. The high yields reported in the patent, consistently above 90%, mean that less raw material is wasted per kilogram of final product, further enhancing the overall cost efficiency. Furthermore, the use of common reagents like sodium hydride and methyl iodide avoids the need for specialized, high-cost catalysts or ligands, simplifying the procurement process and reducing inventory holding costs.
- Enhanced Supply Chain Reliability: The stability of the starting materials used in this new method significantly enhances supply chain reliability. Unlike 4-aminostyrene, which requires cold storage and has a short shelf life, the Boc-protected substrates with leaving groups are stable solids or liquids that can be stored for extended periods without degradation. This stability allows manufacturers to maintain larger safety stocks of raw materials, buffering against supply disruptions and ensuring continuous production even during market fluctuations. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures and ensuring a consistent flow of product to customers. This reliability is crucial for maintaining the trust of downstream partners in the diagnostic imaging supply chain.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the use of standard chemical engineering principles and equipment. The reaction does not require extreme pressures or temperatures, making it safe to operate in large-scale stainless steel reactors. The simplified workup procedure, involving aqueous quenching and extraction, is easily adaptable to industrial separation units. From an environmental compliance perspective, the reduction in solvent usage and waste generation simplifies the permitting process and reduces the burden on effluent treatment plants. The ability to produce commercial scale-up of complex pharmaceutical intermediates with a smaller environmental footprint positions companies favorably in markets with strict regulatory oversight regarding chemical manufacturing emissions and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN111499544B, providing a factual basis for evaluating the feasibility of this route for your specific application. Understanding these details is essential for R&D teams planning pilot runs and procurement teams assessing vendor capabilities. The versatility of the leaving groups and the robustness of the reaction conditions make this a highly adaptable platform for various production scales.
Q: What are the key advantages of this one-pot synthesis method over conventional routes?
A: This method eliminates the need for expensive and unstable 4-aminostyrene as a starting material. By using readily available Boc-protected precursors with leaving groups, it combines methylation and elimination into a single pot, significantly reducing operational complexity and raw material costs while achieving yields over 90%.
Q: Which leaving groups are compatible with this synthesis protocol?
A: The patent specifies that a variety of leaving groups are effective, including tosylate (-OTs), mesylate (-OMs), triflate (-OTf), and iodide (-I). This flexibility allows manufacturers to select the most cost-effective or readily available substrate for their specific supply chain needs.
Q: Is this process scalable for commercial production of diagnostic reagents?
A: Yes, the process uses common industrial solvents like DMF and standard reagents like sodium hydride and methyl iodide. The one-pot nature simplifies scale-up by reducing unit operations, making it highly suitable for the commercial scale-up of complex pharmaceutical intermediates required for PET imaging agents like 18F-AV-45.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Boc-N-methyl-4-aminostyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and manufacture of next-generation diagnostic agents. Our technical team has extensively analyzed the methodology described in CN111499544B and possesses the expertise to implement this one-pot synthesis at an industrial scale. We understand that transitioning to a new synthetic route requires confidence in the partner's ability to manage process risks and ensure product consistency. With our state-of-the-art facilities and rigorous QC labs, we are well-equipped to handle the specific requirements of this chemistry, from the handling of moisture-sensitive reagents like sodium hydride to the precise temperature control needed for optimal yield. Our commitment to excellence ensures that every batch of N-Boc-N-methyl-4-aminostyrene meets the stringent purity specifications required for radiopharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Whether you are looking to reduce lead time for high-purity pharmaceutical intermediates or seeking a long-term partner for commercial production, NINGBO INNO PHARMCHEM offers the capacity and technical depth to support your needs. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements as your demand grows. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis specific to your project. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can deliver value to your organization.
