Scalable Green Catalysis for High-Purity 2-Amino-4H-Chromene Pharmaceutical Intermediates
Scalable Green Catalysis for High-Purity 2-Amino-4H-Chromene Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are increasingly demanding sustainable manufacturing processes that do not compromise on yield or purity. Patent CN103788050A introduces a groundbreaking methodology for the synthesis of 2-amino-4H-chromene derivatives, a critical scaffold found in numerous bioactive compounds ranging from antimicrobial agents to central nervous system therapeutics. This technology leverages a Brönstedt basic ionic liquid catalyst to facilitate a three-component one-pot reaction in an aqueous medium, effectively replacing hazardous volatile organic solvents. The process operates under mild reflux conditions, achieving high conversion rates within a remarkably short timeframe of 15 to 45 minutes. By integrating green chemistry principles with robust synthetic efficiency, this patent offers a compelling solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring that manufacturers can meet stringent regulatory standards while optimizing their production footprint.
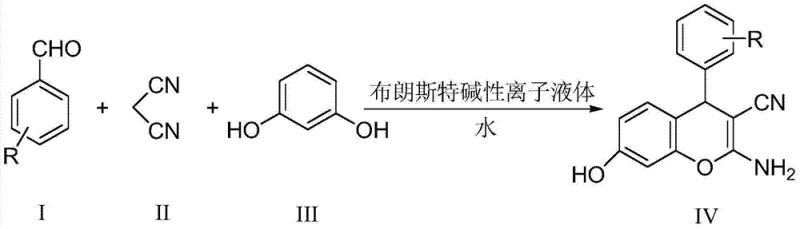
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-4H-chromene derivatives has relied on traditional catalytic systems that present significant operational and environmental challenges. Conventional protocols often necessitate the use of volatile organic solvents such as ethanol, acetonitrile, or dichloromethane, which pose serious safety risks regarding flammability and toxicity, while also generating substantial hazardous waste that requires costly disposal. Furthermore, many existing methods suffer from prolonged reaction times, often extending several hours, which limits throughput and increases energy consumption in large-scale reactors. Traditional catalysts, including piperidine or ammonium salts, frequently exhibit lower catalytic activity, requiring higher loading amounts that complicate downstream purification and increase the impurity profile of the final API intermediate. These inefficiencies create bottlenecks in cost reduction in fine chemical manufacturing, as the cumulative expenses of solvent recovery, waste treatment, and extended reactor occupancy erode profit margins.
The Novel Approach
In stark contrast, the methodology disclosed in CN103788050A utilizes a specialized Brönstedt basic ionic liquid that functions as a highly efficient, reusable catalyst in a purely aqueous environment. This novel approach eliminates the need for toxic organic solvents entirely, utilizing water which is non-flammable, inexpensive, and environmentally benign. The reaction proceeds rapidly under reflux, typically completing within 15 to 45 minutes, which dramatically enhances reactor turnover rates and overall plant productivity. The unique properties of the ionic liquid allow for a simplified workup procedure where the product precipitates upon cooling, enabling isolation via simple filtration rather than complex extraction. This streamlining of the process not only reduces the operational complexity for reliable pharmaceutical intermediate supplier networks but also ensures a cleaner crude product, minimizing the burden on purification teams and facilitating faster time-to-market for new drug candidates.
Mechanistic Insights into Brönstedt Basic Ionic Liquid Catalysis
The core of this technological advancement lies in the specific structure and function of the Brönstedt basic ionic liquid catalyst, identified as a choline hydroxide derivative. As illustrated in the catalyst structure below, the molecule features a quaternary ammonium cation paired with a hydroxide anion, providing strong basic sites that are essential for activating the reactants. The hydroxide anion acts as a potent base to deprotonate the active methylene compound, malononitrile, generating a nucleophilic carbanion species that initiates the cascade reaction. This high density of active basic sites ensures that the reaction kinetics are accelerated significantly compared to weaker organic bases, allowing the process to proceed efficiently even at low catalyst loadings of 3 to 5 mol%. The ionic nature of the catalyst also enhances its solubility in the aqueous phase while maintaining stability under reflux conditions, preventing degradation that often plagues molecular catalysts in harsh environments.
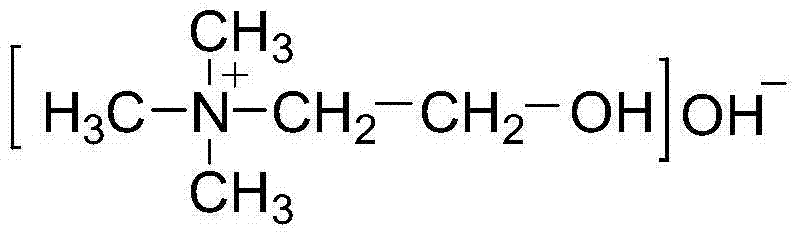
Beyond mere acceleration, the mechanistic pathway facilitated by this ionic liquid contributes to superior impurity control and product quality. The reaction follows a classic Knoevenagel condensation between the aromatic aldehyde and malononitrile, followed by a Michael addition of the resorcinol and subsequent intramolecular cyclization. The mild yet effective basicity of the ionic liquid promotes these steps sequentially without inducing side reactions such as polymerization or over-alkylation, which are common pitfalls in strongly basic traditional media. Furthermore, the aqueous environment helps to suppress the formation of hydrophobic byproducts, driving the equilibrium towards the desired chromene derivative through precipitation. This inherent selectivity results in a high-purity pharmaceutical intermediate with a simplified impurity profile, reducing the need for extensive chromatographic purification and ensuring that the final material meets the rigorous specifications required for downstream drug synthesis.
How to Synthesize 2-Amino-4H-Chromene Derivatives Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and robustness, making it ideal for translation from laboratory benchtop to pilot plant scales. The process involves charging a reactor with water, the Brönstedt basic ionic liquid catalyst, and the three solid or liquid reactants in a precise 1:1:1 molar ratio. The mixture is then heated to reflux with vigorous stirring to ensure homogeneous mass transfer, a critical factor in heterogeneous aqueous systems. Monitoring via TLC confirms the rapid disappearance of the starting aldehyde, signaling reaction completion. Upon cooling, the product crystallizes out of the aqueous solution, allowing for immediate isolation. For detailed operational parameters, stoichiometry, and specific workup instructions, please refer to the standardized synthesis guide below.
- Combine aromatic aldehyde, malononitrile, and resorcinol in a 1: 1:1 molar ratio with 3-5% Brönstedt basic ionic liquid catalyst in water.
- Reflux the mixture under vigorous stirring for 15 to 45 minutes until TLC indicates complete consumption of starting materials.
- Cool to room temperature to precipitate solids, filter, wash with water, and recrystallize from methanol to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this green catalytic technology translates into tangible strategic benefits that extend beyond simple chemical yield. The shift from organic solvents to water fundamentally alters the cost structure of production by eliminating the procurement, storage, and recycling costs associated with volatile organic compounds. Additionally, the biodegradable nature of the choline-based ionic liquid aligns with increasingly strict environmental regulations, reducing the risk of compliance penalties and enhancing the sustainability profile of the supply chain. The ability to recycle the catalyst filtrate directly for subsequent batches further amplifies these economic advantages, as it minimizes raw material consumption and waste generation. This creates a more resilient and cost-effective supply model for reducing lead time for high-purity pharmaceutical intermediates, ensuring consistent availability of critical building blocks for drug development.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents and the reduction in catalyst loading directly lower the variable costs of production. Since the catalyst can be recovered and reused multiple times without significant loss of activity, the effective cost per kilogram of catalyst consumed is drastically reduced. Furthermore, the simplified workup involving filtration rather than extraction reduces labor hours and energy consumption associated with solvent evaporation and distillation. These factors combine to deliver substantial cost savings that improve the overall margin profile of the manufactured intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing water as the primary reaction medium mitigates supply chain risks associated with the volatility and regulatory restrictions of organic solvents. Water is universally available and不受 supply fluctuations, ensuring uninterrupted production capabilities even during global logistics disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, leading to more consistent batch-to-batch performance. This reliability is crucial for maintaining steady inventory levels and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The aqueous nature of the reaction simplifies heat transfer and safety management during scale-up, as there is no risk of solvent flammability or explosive vapor formation. This allows for safer operation in larger reactors, facilitating the transition from pilot scale to commercial tonnage production. Moreover, the biodegradability of the catalyst and the absence of halogenated solvents significantly reduce the environmental footprint of the manufacturing process. This alignment with green chemistry principles facilitates easier permitting and regulatory approval, accelerating the timeline for bringing new processes online.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the primary advantages of using Brönstedt basic ionic liquids over traditional catalysts for chromene synthesis?
A: Unlike traditional catalysts that often require volatile organic solvents and lengthy reaction times, Brönstedt basic ionic liquids enable rapid reactions in water with high catalytic activity. They are also biodegradable and can be recycled multiple times without significant loss of efficiency, addressing both environmental and cost concerns.
Q: How does the water-based solvent system impact the purification process?
A: The use of water as a solvent simplifies purification significantly. Upon cooling, the product precipitates as a solid, allowing for simple filtration. This eliminates the need for complex extraction procedures and reduces the generation of hazardous organic waste streams.
Q: Is the ionic liquid catalyst recoverable for industrial scale-up?
A: Yes, the patent data indicates that the filtrate containing the ionic liquid catalyst can be directly reused for subsequent batches. The catalyst maintains its activity over at least five cycles, making it highly suitable for continuous or batch-wise industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4H-Chromene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies like the one described in CN103788050A for the future of fine chemical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-amino-4H-chromene derivatives meets the exacting standards required by the global pharmaceutical industry. We are committed to leveraging such advanced synthetic routes to deliver superior value to our partners.
We invite you to collaborate with us to explore how this efficient, water-based synthesis can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in green chemistry can support your long-term strategic goals.
