Advanced Synthesis of Fluorinated 1-Alkoxycarbonyl-2H-Isoindole Derivatives for High-Value Applications
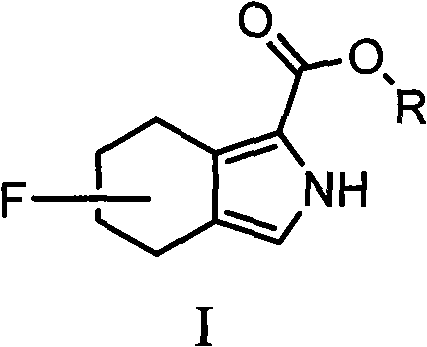
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to incorporate fluorine atoms into complex molecular scaffolds, driven by the unique metabolic stability and electronic properties that fluorine imparts to bioactive molecules. Patent CN101906062B presents a significant technological breakthrough in this domain by disclosing a novel class of fluorine-containing 1-alkoxycarbonyl-2H-isoindole derivatives. These compounds serve as critical intermediates for the synthesis of fluorine-containing porphyrins, which are increasingly valued in photodynamic therapy and advanced material science, as well as serving as core structures for indigo dyes and phthalocyanine compounds. The innovation lies not just in the molecule itself, but in the strategic retrosynthetic approach that bypasses the notorious difficulties associated with direct aliphatic fluorination.
Traditionally, introducing a fluorine atom directly onto a saturated aliphatic ring requires harsh conditions, specialized reagents, and often results in low regioselectivity and poor yields. This patent elegantly circumvents these obstacles by starting with readily available industrial fluorinated aromatic hydrocarbon derivatives. By leveraging a classical yet optimized catalytic hydrogenation reduction, the technology converts the aromatic precursor into the desired tetrahydro-isoindole structure while meticulously preserving the carbon-fluorine bond. This approach transforms a complex synthetic challenge into a manageable, high-yielding industrial process, offering a reliable fluorinated isoindole supplier pathway for downstream applications in agrochemicals and advanced organic electronics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the realm of organic synthesis, the construction of fluorinated alicyclic systems has historically been a bottleneck for process chemists. Conventional methods often rely on electrophilic fluorination of enolates or nucleophilic substitution on activated aliphatic substrates, both of which suffer from significant drawbacks. Electrophilic fluorination reagents are frequently expensive, hazardous, and difficult to handle on a multi-kilogram scale, posing severe safety risks and environmental burdens. Furthermore, achieving high regioselectivity on a saturated ring is notoriously difficult, often leading to complex mixtures of isomers that require costly and yield-eroding purification steps. The instability of certain fluorinated intermediates under standard reaction conditions further complicates the synthesis, limiting the practical utility of these routes for commercial scale-up of complex polymer additives or pharmaceutical intermediates.
The Novel Approach
The methodology described in CN101906062B represents a paradigm shift by utilizing a "fluorine-first" strategy on an aromatic system followed by reduction. This approach capitalizes on the maturity of industrial fluorobenzene chemistry, where fluorinated aromatics are commodity chemicals available at competitive prices. By subjecting these stable aromatic precursors to catalytic hydrogenation, the process achieves the saturation of the ring system under relatively mild conditions. This novel approach ensures that the fluorine atom, once installed on the robust aromatic ring, remains intact during the reduction phase. The result is a streamlined synthesis that eliminates the need for exotic fluorinating agents, drastically simplifies the workflow, and delivers the target fluorinated tetrahydro-isoindole derivatives with impressive consistency and purity profiles suitable for high-value applications.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation
The core of this synthetic transformation relies on the heterogeneous catalytic hydrogenation of the isoindole aromatic system using palladium on carbon (Pd/C). In this mechanism, molecular hydrogen is adsorbed onto the surface of the palladium particles, where it dissociates into reactive atomic hydrogen species. Simultaneously, the fluorinated aromatic substrate adsorbs onto the catalyst surface. The reaction proceeds through a stepwise addition of hydrogen atoms to the double bonds of the aromatic ring. A critical aspect of this mechanism is the chemoselectivity required to reduce the carbon-carbon double bonds of the benzene ring without triggering hydrodefluorination, a side reaction where the carbon-fluorine bond is cleaved by hydrogen. The patent specifies optimal conditions that favor ring saturation over defluorination, ensuring the fluorine substituent is retained in the final aliphatic structure.
Impurity control in this process is inherently managed by the choice of catalyst and reaction parameters. The use of Pd/C, a standard and well-understood catalyst, minimizes the formation of heavy metal residues compared to homogeneous catalysts, simplifying downstream purification. The reaction conditions, specifically the temperature range of 20-80°C and hydrogen pressure of 40-60 atm, are tuned to drive the reaction to completion while suppressing thermal degradation or over-reduction of the ester functionality. Post-reaction, the heterogeneous nature of the catalyst allows for simple removal via filtration, preventing catalyst leaching into the product stream. This mechanistic robustness translates directly to a cleaner crude product profile, reducing the burden on recrystallization steps and ensuring the final high-purity OLED material or pharmaceutical intermediate meets stringent quality specifications.
How to Synthesize Fluorinated Isoindole Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the dissolution of the fluorinated aromatic precursor in a protic solvent, typically ethanol or methanol, which serves both as a reaction medium and a proton source. Following the addition of the Pd/C catalyst, the system is pressurized with hydrogen gas. The reaction is monitored until completion, typically requiring 8 to 16 hours depending on the specific substitution pattern and reaction temperature. The detailed standardized synthesis steps below outline the precise operational parameters required to achieve the reported yields of 72% to 80%, ensuring reproducibility and safety during execution.
- Dissolve the fluorinated aromatic isoindole precursor (Compound II) in a protic solvent such as ethanol or methanol.
- Add a palladium on carbon (Pd/C) catalyst (5-10% by weight) to the reaction mixture under an inert atmosphere.
- Conduct hydrogenation at 40-60 atm pressure and 20-80°C for 8-16 hours, followed by filtration and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from complex, multi-step fluorination routes to this streamlined hydrogenation process offers tangible economic and logistical benefits. The primary advantage lies in the raw material strategy; by utilizing industrial-grade fluorinated aromatics, the process taps into an established, high-volume supply chain that is less susceptible to the volatility seen with specialized fluorinating reagents. This shift significantly reduces the cost of goods sold (COGS) by eliminating expensive reagents and minimizing waste disposal costs associated with hazardous fluorination byproducts. Furthermore, the simplicity of the workup—filtration followed by concentration and recrystallization—reduces processing time and energy consumption, contributing to substantial cost savings in manufacturing overheads.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous electrophilic fluorinating agents dramatically lowers the direct material costs associated with production. Additionally, the use of a recoverable heterogeneous catalyst (Pd/C) avoids the financial loss associated with precious metal ligands used in homogeneous catalysis. The simplified purification process, which relies on standard crystallization rather than complex chromatography, further reduces solvent usage and labor hours, leading to a more economically efficient production model that enhances margin potential for high-volume contracts.
- Enhanced Supply Chain Reliability: Sourcing fluorinated aromatic precursors is significantly more reliable than sourcing specialized aliphatic fluorinating reagents, which often have limited suppliers and long lead times. The robustness of the hydrogenation process also means that production schedules are less likely to be disrupted by sensitive reaction failures. This stability allows for better inventory planning and ensures consistent delivery timelines for downstream customers who rely on these intermediates for their own API or dye manufacturing processes, thereby reducing lead time for high-purity dye intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard high-pressure hydrogenation reactors that are common in fine chemical facilities. The absence of toxic fluorinating byproducts simplifies waste treatment and aligns with increasingly stringent environmental regulations. The ability to run the reaction in common solvents like ethanol or methanol further enhances the green chemistry profile of the process, making it easier to obtain regulatory approvals for commercial scale-up of complex heterocyclic intermediates in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated isoindole derivatives. The answers are derived directly from the experimental data and technical specifications provided in patent CN101906062B, ensuring accuracy and relevance for technical decision-makers evaluating this technology for integration into their supply chains.
Q: What is the primary advantage of this hydrogenation method over direct fluorination?
A: Direct fluorination of aliphatic rings is chemically challenging and often yields poor results. This patent utilizes readily available fluorinated aromatic precursors and converts them via hydrogenation, overcoming the difficulty of introducing fluorine atoms onto saturated rings while maintaining high yields.
Q: What are the critical reaction conditions for maintaining the C-F bond integrity?
A: The process employs mild temperatures (20-80°C) and specific Pd/C catalysts under controlled hydrogen pressure (40-60 atm). These conditions ensure chemoselective reduction of the aromatic ring without causing hydrodefluorination, preserving the crucial fluorine substituent.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights industrial suitability. The use of standard heterogeneous catalysts, common solvents like ethanol, and simple post-treatment steps like filtration and recrystallization makes the process highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Isoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation pharmaceuticals and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of fluorinated 1-alkoxycarbonyl-2H-isoindole derivative meets the exacting standards required for sensitive downstream applications such as porphyrin synthesis.
We invite global partners to collaborate with us to leverage this innovative synthetic route for their specific project needs. Whether you require cost reduction in pharmaceutical intermediate manufacturing or need a secure supply of specialized dye precursors, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential clients to reach out for specific COA data and route feasibility assessments to determine how our capabilities can best support your R&D and commercial production goals.
