Scalable Production of Vinyl Imidazole Derivatives for Antifungal Applications
Introduction to Advanced Imidazole Synthesis
The development of efficient synthetic routes for heterocyclic compounds remains a cornerstone of modern pharmaceutical and agrochemical manufacturing. Patent CN85104009A introduces a transformative methodology for the preparation of vinyl imidazole derivatives, which serve as critical intermediates for potent antimycotic agents and agricultural fungicides. This technology addresses longstanding challenges in the industry, specifically regarding reagent safety, solvent stability, and byproduct management. By shifting from traditional, hazardous reagents to more benign alternatives, this process offers a robust pathway for producing high-purity intermediates essential for downstream drug synthesis. The innovation lies not only in the final coupling step but also in a clever recycling mechanism that converts potential waste into valuable product, thereby enhancing the overall economic viability of the synthesis.
For R&D directors and process chemists, the significance of this patent extends beyond simple yield improvements; it represents a fundamental optimization of the reaction ecosystem. The traditional reliance on sodium hydride and dimethylformamide (DMF) has long been a bottleneck due to safety concerns and impurity profiles. This new approach leverages alkali metal hydroxides and dimethyl sulfoxide (DMSO) to create a cleaner, more controllable reaction environment. Furthermore, the ability to salvage bis-imidazole byproducts through acid-catalyzed rearrangement demonstrates a sophisticated understanding of reaction kinetics and thermodynamics, ensuring that raw material costs are minimized while output is maximized. This holistic view of the synthesis makes it an attractive candidate for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing vinyl imidazole derivatives typically relied on the use of sodium hydride as a base in dried dimethylformamide (DMF). While effective in small-scale laboratory settings, this approach presents severe limitations for industrial application. Sodium hydride is a pyrophoric solid that poses significant safety risks during handling and storage, requiring specialized equipment and rigorous safety protocols that drive up operational costs. Moreover, DMF is prone to decomposition under strongly basic conditions, breaking down into dimethylamine. This decomposition product is not merely a solvent impurity; it actively participates in side reactions with benzyl halides to form 4-benzylchloro-dimethylamine byproducts. These impurities are structurally similar to the target molecule, making purification extremely difficult and resulting in low isolated yields of the desired compound.
The Novel Approach
The methodology described in the patent fundamentally re-engineers the synthesis to overcome these deficits. By substituting sodium hydride with inexpensive and safe alkali metal hydroxides such as potassium hydroxide, the process eliminates the hazards associated with pyrophoric reagents. Crucially, the switch from DMF to DMSO as the solvent prevents the formation of dimethylamine, thereby eradicating the source of the troublesome amine byproducts. This change alone drastically simplifies the workup procedure and improves the purity of the crude product. Additionally, the process introduces a unique strategy for managing the bis-imidazole byproduct (Compound IV) that inevitably forms during the initial condensation step. Instead of viewing this as waste, the novel approach converts it back into the desired mono-imidazole intermediate (Compound II) through heating or acid treatment.
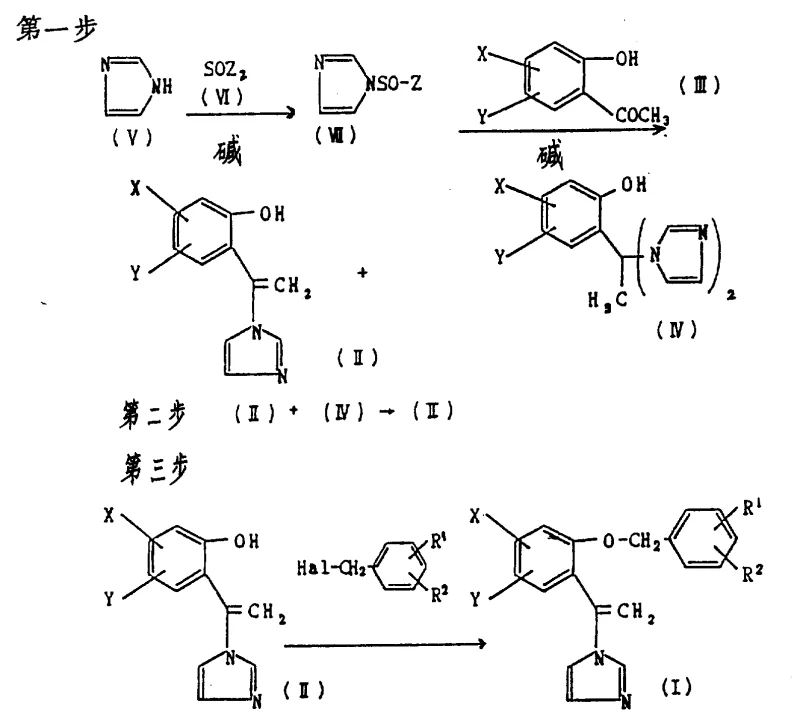
This comprehensive reaction sequence illustrates the efficiency of the new process. It highlights how the starting material (III) is converted into a mixture of the target intermediate (II) and the byproduct (IV), which is then recycled. The final step involves the alkylation to form the active derivative (I). This integrated approach ensures that the productive rate of the purpose compound is significantly higher than in conventional methods, providing a reliable supply of high-quality intermediates for the production of antifungal agents.
Mechanistic Insights into Acid-Catalyzed Byproduct Conversion
A standout feature of this technology is the mechanistic understanding applied to the conversion of the bis-imidazole byproduct (IV) into the mono-imidazole intermediate (II). In traditional syntheses, the formation of double imidazole compounds is often an irreversible loss of material, reducing atom economy. However, this patent reveals that Compound IV can be quantitatively reverted to Compound II under specific conditions. The mechanism involves the protonation of the imidazole nitrogen or the cleavage of the C-N bond under acidic or thermal stress. Experimental data within the patent demonstrates that various acids, including p-toluenesulfonic acid (P-TsOH), trifluoroacetic acid, and Lewis acids like boron trifluoride etherate, can catalyze this transformation with high efficiency.
The kinetic data presented in the patent underscores the versatility of this conversion step. As shown in the graphical analysis, the addition of catalytic amounts of acid significantly accelerates the conversion rate compared to thermal treatment alone. For instance, using 0.1 molar equivalent of p-toluenesulfonic acid allows for nearly complete conversion within a few hours at reflux temperatures. This mechanistic insight allows process chemists to fine-tune the reaction conditions to minimize residence time and energy consumption. By controlling the acidity and temperature, manufacturers can ensure that the equilibrium favors the mono-imidazole species, effectively turning a potential waste stream into a valuable resource and enhancing the overall sustainability of the manufacturing process.
How to Synthesize Vinyl Imidazole Derivatives Efficiently
The synthesis of these valuable intermediates is achieved through a streamlined three-step protocol that balances reactivity with selectivity. The process begins with the condensation of an o-hydroxyacetophenone derivative with a 1-halosulfinyl-1H-imidazole reagent in the presence of a tertiary amine base. This step generates the crude mixture of mono- and bis-imidazoles. Following this, the mixture undergoes a crucial recycling phase where acid or heat is applied to convert the bis-imidazole fraction into the desired mono-imidazole. Finally, the purified intermediate is alkylated with a benzyl halide in a DMSO solvent system containing potassium hydroxide to yield the final vinyl imidazole derivative. Detailed standardized synthetic steps for implementing this route are provided in the guide below.
- React o-hydroxyacetophenone derivatives with 1-halosulfinyl-1H-imidazoles in the presence of a base to form a mixture of mono- and bis-imidazole compounds.
- Treat the reaction mixture with acid or heat to convert the bis-imidazole byproduct back into the desired mono-imidazole intermediate, significantly improving yield.
- Perform nucleophilic substitution on the mono-imidazole intermediate using benzyl halides in a DMSO solvent system with alkali metal hydroxide to obtain the final vinyl imidazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond the laboratory bench. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for sodium hydride, companies remove a hazardous material from their inventory, reducing insurance costs and regulatory compliance burdens. Furthermore, the replacement of DMF with DMSO mitigates the risk of solvent-related impurities that often lead to batch failures and costly re-processing. The ability to recycle the bis-imidazole byproduct internally means that less starting material is required per kilogram of final product, directly translating to substantial cost savings in raw material procurement.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents like sodium hydride in favor of commodity chemicals like potassium hydroxide significantly lowers the direct material cost of the synthesis. Additionally, the internal recycling of the bis-imidazole byproduct improves the overall yield, meaning less feedstock is wasted. This efficiency gain reduces the cost per kilogram of the active intermediate, allowing for more competitive pricing in the final pharmaceutical or agrochemical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on stable, non-hazardous reagents ensures a more resilient supply chain. Potassium hydroxide and DMSO are widely available commodity chemicals with robust global supply networks, unlike specialized reagents that may face shortages. The simplified purification process, free from difficult-to-separate amine byproducts, also reduces the lead time for batch release. This reliability ensures consistent delivery schedules for downstream customers, minimizing the risk of production stoppages due to intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up. The use of aqueous workups and common organic solvents facilitates easy transition from pilot plant to commercial production volumes. From an environmental perspective, the reduction in hazardous waste (no sodium hydride quenching) and the minimization of solvent decomposition byproducts align with green chemistry principles. This compliance with stricter environmental regulations future-proofs the manufacturing process against evolving regulatory landscapes, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on reaction conditions and material compatibility. Understanding these nuances is critical for process engineers aiming to replicate the high yields and purity levels reported in the intellectual property.
Q: Why is DMSO preferred over DMF in this synthesis?
A: DMF tends to decompose under basic conditions to form dimethylamine, which reacts with benzyl halides to create difficult-to-remove impurities. DMSO avoids this side reaction, leading to higher purity and easier isolation of the final product.
Q: How does this process handle the bis-imidazole byproduct?
A: Instead of discarding the bis-imidazole byproduct formed in the first step, the process utilizes an acid-catalyzed or thermal treatment to convert it back into the valuable mono-imidazole intermediate, thereby maximizing atom economy and overall yield.
Q: What safety advantages does this method offer over prior art?
A: The method replaces hazardous sodium hydride (NaH) with safer and cheaper alkali metal hydroxides like potassium hydroxide (KOH). This eliminates the risks associated with handling pyrophoric reagents and simplifies the operational safety profile for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinyl Imidazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antifungal therapies and crop protection agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative processes described in patents like CN85104009A can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of vinyl imidazole derivative meets the exacting standards required by global pharmaceutical regulators. Our commitment to quality assurance ensures that your downstream synthesis proceeds without interruption or impurity-related delays.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your specific product pipelines. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can reduce your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in delivering efficient, scalable, and cost-effective chemical solutions for the global market.
