Advanced Synthesis of Phosphonate-Containing Sulfonamides for Next-Generation Antibacterial Applications
Advanced Synthesis of Phosphonate-Containing Sulfonamides for Next-Generation Antibacterial Applications
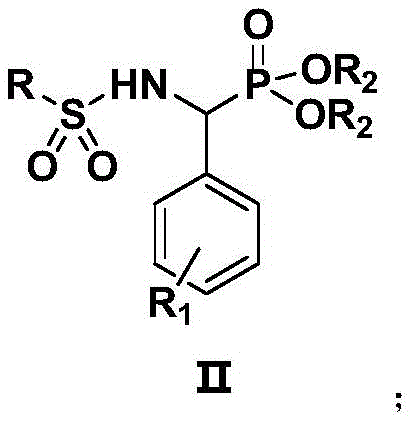
The escalating global crisis of antimicrobial resistance (AMR) has necessitated the urgent development of novel chemical entities capable of overcoming multidrug-resistant pathogens. Patent CN109678900B addresses this critical challenge by disclosing a series of innovative sulfonamide derivatives characterized by a unique phosphonate ester structural motif. These compounds, represented generally by Formula II, exhibit remarkable antibacterial potency against formidable superbugs including Methicillin-resistant Staphylococcus aureus (MRSA) and fluoroquinolone-resistant Escherichia coli (MREC). As a leading reliable antibacterial intermediate supplier, we recognize that the structural integration of phosphonate groups into the classic sulfonamide scaffold represents a strategic evolution in medicinal chemistry, potentially offering multi-targeting mechanisms that bypass traditional resistance pathways. The following technical insight report dissects the proprietary synthesis methodology and commercial viability of these high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted sulfonamides has relied heavily on classical Schotten-Baumann conditions or the use of stoichiometric organic bases such as triethylamine or inorganic bases like potassium carbonate in organic solvents. While these methods are well-established, they often suffer from significant inefficiencies when applied to sterically hindered or electronically complex substrates. The background data within the patent highlights that conventional base-mediated coupling of aminophosphonates with sulfonyl chlorides can result in prolonged reaction times exceeding 13 hours and disappointing isolated yields hovering around 30% to 40%. Such inefficiencies translate directly into increased operational expenditures due to higher solvent consumption, extended reactor occupancy, and substantial loss of valuable starting materials, thereby creating a bottleneck for the cost reduction in pharmaceutical manufacturing of complex antibacterial candidates.
The Novel Approach
In a transformative departure from traditional protocols, the disclosed invention employs an alkaline ionic liquid, specifically 1-butyl-3-methylimidazolium hydroxide ([Bmim]OH), as a highly efficient reaction promoter. This novel approach facilitates the nucleophilic attack of the aminophosphonate on the sulfonyl chloride under mild reflux conditions in anhydrous dichloromethane. The implementation of this ionic liquid catalytic system results in a dramatic enhancement of reaction kinetics, reducing the requisite heating time to merely 3 to 4 hours. More critically, this methodology consistently delivers isolated yields in the range of 80% to 85%, effectively doubling the output compared to conventional base methods. This leap in efficiency not only optimizes resource utilization but also simplifies the downstream purification process, establishing a robust foundation for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ionic Liquid Promoted Sulfonylation
The exceptional performance of the [Bmim]OH ionic liquid in this transformation can be attributed to its dual functionality as both a basic catalyst and a phase-transfer mediator. Unlike traditional bulky organic bases that may suffer from steric hindrance or poor solubility profiles, the ionic liquid creates a highly polar microenvironment that stabilizes the transition state of the nucleophilic substitution. The hydroxide anion of the ionic liquid likely deprotonates the amino group of the phosphonate intermediate, generating a more nucleophilic nitrogen species that rapidly attacks the electrophilic sulfur center of the sulfonyl chloride. Furthermore, the imidazolium cation may assist in stabilizing the chloride leaving group through electrostatic interactions, thereby lowering the activation energy barrier for the formation of the sulfonamide bond. This synergistic effect ensures high conversion rates even with diverse substituents on the aromatic rings, maintaining consistent quality across the compound library.
From an impurity control perspective, the mild yet effective nature of this ionic liquid system minimizes the formation of common by-products such as sulfonic acids or hydrolysis products that often plague aqueous or harsh basic conditions. The use of anhydrous dichloromethane as the bulk solvent further suppresses hydrolytic degradation of the sensitive phosphonate ester groups. By avoiding excessive thermal stress and harsh reagents, the process preserves the stereochemical integrity of the chiral center adjacent to the phosphorus atom, which is often crucial for biological activity. This high level of chemoselectivity reduces the burden on downstream purification steps, allowing for the isolation of high-purity intermediates that meet stringent regulatory specifications required for preclinical and clinical evaluation of new antibacterial agents.
How to Synthesize Sulfonamide Derivative II Efficiently
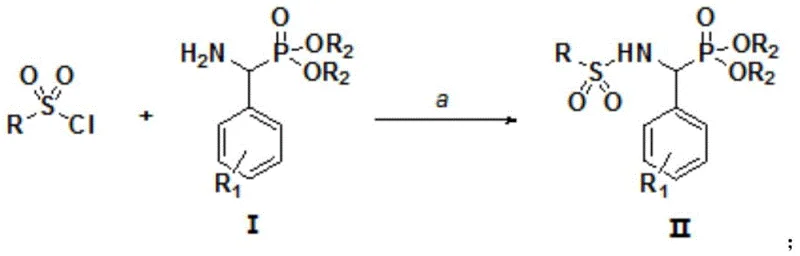
The preparation of these bioactive molecules follows a streamlined one-pot procedure that is amenable to both laboratory optimization and industrial production. The process begins with the precise metering of sulfonyl chloride and the aminophosphonate intermediate in a 1:1 molar ratio, followed by the addition of the [Bmim]OH ionic liquid promoter at 0.5 equivalents. The reaction mixture is then diluted with anhydrous dichloromethane and subjected to controlled heating. Detailed standardized synthetic steps for optimizing temperature profiles and workup procedures are provided in the guide below to ensure reproducibility and maximum yield recovery.
- Mix sulfonyl chloride, aminophosphonate intermediate I, and [Bmim]OH ionic liquid in a 1: 1:0.5 molar ratio.
- Add anhydrous dichloromethane (10-15 mL per 1 mmol sulfonyl chloride) and stir the mixture thoroughly.
- Heat under reflux for 3-4 hours, concentrate under reduced pressure, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-mediated synthesis route offers compelling economic and logistical advantages over legacy manufacturing processes. The primary value driver lies in the substantial increase in process mass intensity (PMI) efficiency; by nearly doubling the reaction yield from roughly 40% to over 80%, the consumption of expensive starting materials such as functionalized sulfonyl chlorides and chiral aminophosphonates is drastically reduced per kilogram of final product. This reduction in raw material waste directly correlates to a lower cost of goods sold (COGS), enabling more competitive pricing strategies for the final active pharmaceutical ingredients. Additionally, the shortened reaction cycle time of 3 to 4 hours allows for higher throughput in existing reactor fleets, effectively increasing production capacity without the need for capital-intensive infrastructure expansion.
- Cost Reduction in Manufacturing: The elimination of stoichiometric amounts of traditional bases and the associated salt waste generation simplifies the effluent treatment process. Traditional methods often generate large volumes of amine hydrochloride salts or inorganic sludge that require costly disposal and neutralization steps. In contrast, the ionic liquid system generates minimal inorganic waste, and the potential for recycling the ionic liquid catalyst in future iterations could further drive down operational costs. This alignment with green chemistry principles not only reduces direct waste disposal fees but also mitigates regulatory risks associated with environmental compliance, ensuring long-term sustainability of the supply chain.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, including various substituted benzenesulfonyl chlorides and diethyl aminobenzylphosphonates, are commercially available from multiple global vendors, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of electronic substituents (such as halogens and trifluoromethyl groups), means that supply disruptions for specific precursors can often be managed by switching to alternative analogues without re-validating the entire process. This flexibility ensures continuous availability of high-purity antibacterial intermediates, safeguarding downstream drug development timelines against raw material shortages.
- Scalability and Environmental Compliance: The use of dichloromethane, a standard industrial solvent, ensures that the process can be easily scaled from gram-scale laboratory synthesis to multi-ton commercial production using existing equipment. The straightforward workup procedure involving simple concentration and column chromatography avoids complex distillation or crystallization steps that often pose scaling challenges. Furthermore, the high atom economy of the reaction minimizes the generation of hazardous by-products, facilitating easier adherence to increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions and chemical waste discharge, thus securing the social license to operate for large-scale manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel sulfonamide derivatives. The answers are derived directly from the experimental data and mechanistic understanding provided in the patent literature, ensuring accuracy and relevance for technical decision-makers evaluating this technology for their pipeline.
Q: What is the primary advantage of using [Bmim]OH in this sulfonamide synthesis?
A: The use of [Bmim]OH ionic liquid significantly accelerates the reaction rate and improves yield compared to traditional bases like triethylamine or potassium carbonate, reducing reaction time from over 13 hours to just 3-4 hours while boosting yields from ~40% to over 80%.
Q: Which bacterial strains show the highest sensitivity to these new derivatives?
A: The synthesized derivatives demonstrate potent activity against both Gram-positive and Gram-negative bacteria, with particularly significant efficacy against drug-resistant strains such as Methicillin-resistant Staphylococcus aureus (MRSA) and fluoroquinolone-resistant Escherichia coli (MREC).
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes standard organic solvents like dichloromethane and straightforward workup procedures involving concentration and column chromatography, making it highly suitable for scale-up from laboratory to commercial manufacturing levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonamide Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, positioning us as an ideal partner for bringing these advanced antibacterial intermediates to market. Our state-of-the-art facilities are equipped to handle the specific requirements of ionic liquid chemistry and phosphonate handling, ensuring stringent purity specifications are met for every batch. With rigorous QC labs employing advanced analytical techniques, we guarantee the structural integrity and biological consistency of our products, providing our partners with the confidence needed to advance their drug candidates through clinical trials.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for transitioning from legacy synthesis routes or need specific COA data and route feasibility assessments for novel analogues, our experts are ready to collaborate. Let us help you secure a stable, cost-effective supply of these critical high-purity antibacterial intermediates to accelerate your fight against drug-resistant infections.
