Advanced Synthesis of N-Thioaryl Benzenesulfonylimine for Commercial Pharmaceutical Intermediate Production
Advanced Synthesis of N-Thioaryl Benzenesulfonylimine for Commercial Pharmaceutical Intermediate Production
The landscape of organic synthesis, particularly within the realm of pharmaceutical intermediate manufacturing, is constantly evolving to address the critical need for stable, high-performance reagents. A significant breakthrough in this domain is detailed in Chinese Patent CN111848460B, which introduces a novel class of N-thioaryl benzenesulfonylimines. These compounds represent a paradigm shift from traditional electrophilic sulfur aryl reagents, offering unprecedented stability against environmental factors such as moisture and oxygen. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such innovations is paramount for maintaining a competitive edge in the global supply chain. The patent outlines a robust preparation method that transforms bisbenzenesulfonimide into a highly active electrophilic thioarylation reagent, thereby opening new avenues for the construction of complex organic sulfur compounds essential in modern drug discovery.
The core innovation lies in the unique structural architecture of the N-thioaryl benzenesulfonylimine, depicted in the general formula (I). This structure incorporates a nitrogen atom bonded to a sulfur atom, which is further connected to an aryl group, all stabilized by two benzenesulfonyl groups attached to the nitrogen. 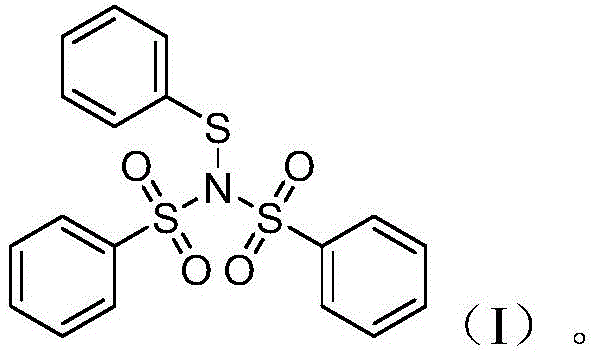 . This specific arrangement mitigates the inherent instability found in simpler sulfenyl chlorides, where the sulfur-chlorine bond is prone to hydrolysis. By replacing the labile chlorine atom with a robust nitrogen-sulfur linkage, the resulting reagent exhibits superior shelf-life and handling characteristics, making it an ideal candidate for large-scale commercial applications where reagent consistency is non-negotiable.
. This specific arrangement mitigates the inherent instability found in simpler sulfenyl chlorides, where the sulfur-chlorine bond is prone to hydrolysis. By replacing the labile chlorine atom with a robust nitrogen-sulfur linkage, the resulting reagent exhibits superior shelf-life and handling characteristics, making it an ideal candidate for large-scale commercial applications where reagent consistency is non-negotiable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organic sulfur compounds has relied heavily on direct thioarylation using reagents like benzene sulfenyl chloride. However, these conventional methods suffer from significant drawbacks that hinder their scalability and operational safety in an industrial environment. The primary limitation is the intrinsic instability of the sulfur-chlorine (S-Cl) bond, which renders these reagents highly sensitive to atmospheric moisture and oxygen. This sensitivity necessitates stringent anhydrous and anaerobic conditions, often requiring specialized equipment such as gloveboxes or rigorous Schlenk line techniques, which drastically increases the capital expenditure and operational complexity for manufacturing facilities. Furthermore, many traditional sulfenylating agents possess potent, unpleasant odors and can decompose rapidly upon exposure to ambient conditions, leading to inconsistent reaction outcomes and potential safety hazards for personnel. The lack of regioselectivity in some linear sulfur aryl reagents also poses challenges in synthesizing complex molecules where precise control over the reaction site is critical for biological activity.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a bisbenzenesulfonimide scaffold to generate a reagent that is both chemically robust and highly reactive. The synthesis begins with the deprotonation of bisbenzenesulfonimide using sodium hydride to form a stable sodium salt, which subsequently reacts with various benzene sulfenyl chlorides. 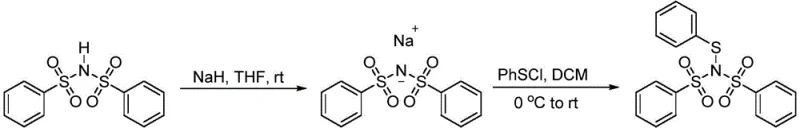 . This two-step process effectively masks the reactivity of the sulfur center until it is needed, providing a 'protected' form of the electrophile that is insensitive to air and water. The resulting N-thioaryl benzenesulfonylimine retains high electrophilicity due to the electron-withdrawing nature of the sulfonyl groups, yet it avoids the volatility and instability associated with free sulfenyl chlorides. This approach not only simplifies the logistics of reagent storage and transport but also enhances the reproducibility of downstream synthetic transformations, a key factor for cost reduction in fine chemical manufacturing.
. This two-step process effectively masks the reactivity of the sulfur center until it is needed, providing a 'protected' form of the electrophile that is insensitive to air and water. The resulting N-thioaryl benzenesulfonylimine retains high electrophilicity due to the electron-withdrawing nature of the sulfonyl groups, yet it avoids the volatility and instability associated with free sulfenyl chlorides. This approach not only simplifies the logistics of reagent storage and transport but also enhances the reproducibility of downstream synthetic transformations, a key factor for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Electrophilic Thioarylation
The mechanistic underpinning of this technology relies on the unique electronic properties of the N-S bond within the benzenesulfonylimide framework. When the bisbenzenesulfonimide is treated with a strong base like sodium hydride in dry tetrahydrofuran, the acidic proton on the nitrogen is abstracted, generating a nucleophilic nitrogen anion stabilized by resonance with the adjacent sulfonyl groups. This anion then attacks the electrophilic sulfur atom of the benzene sulfenyl chloride, displacing the chloride ion and forming the new N-S bond. The presence of the two bulky benzenesulfonyl groups creates a specific steric environment around the nitrogen center, which influences the regioselectivity of subsequent reactions. Unlike linear reagents that may offer little steric differentiation, this cyclic-like constraint (in terms of steric bulk) directs incoming nucleophiles to specific sites on the substrate, enabling the synthesis of isomers that are difficult to access via traditional routes. This level of control is invaluable for R&D directors aiming to optimize the purity profiles of active pharmaceutical ingredients.
Furthermore, the application of this reagent extends beyond simple substitution; it facilitates cascade reactions such as thioarylation followed by lactonization or etherification. For example, in the presence of a Brønsted acid like trifluoroacetic acid, the reagent can initiate a cyclization sequence where the thioaryl group is installed, and a lactone ring is simultaneously formed. 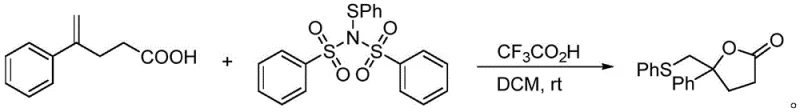 . This dual functionality demonstrates the reagent's versatility in constructing complex heterocyclic cores found in many bioactive molecules. The mechanism likely involves the activation of the alkene by the electrophilic sulfur species, followed by intramolecular nucleophilic attack by the carboxylic acid moiety. The ability to perform such transformations under mild conditions (room temperature) without the need for transition metal catalysts represents a significant advancement in green chemistry principles, reducing the burden of heavy metal removal in the final purification steps.
. This dual functionality demonstrates the reagent's versatility in constructing complex heterocyclic cores found in many bioactive molecules. The mechanism likely involves the activation of the alkene by the electrophilic sulfur species, followed by intramolecular nucleophilic attack by the carboxylic acid moiety. The ability to perform such transformations under mild conditions (room temperature) without the need for transition metal catalysts represents a significant advancement in green chemistry principles, reducing the burden of heavy metal removal in the final purification steps.
How to Synthesize N-Thioaryl Benzenesulfonylimine Efficiently
The preparation of this high-value intermediate is designed for operational simplicity and scalability, utilizing common laboratory solvents and reagents that are readily available in the global chemical market. The process avoids exotic catalysts or extreme pressure conditions, making it highly amenable to transfer from bench scale to pilot plant operations. The protocol emphasizes the importance of anhydrous conditions during the initial salt formation to ensure complete conversion, followed by a controlled addition of the sulfenyl chloride to manage exothermicity.
- React bisbenzenesulfonimide with sodium hydride in dry tetrahydrofuran at room temperature to form the sodium salt.
- Dissolve the sodium salt in dry dichloromethane and react with benzene sulfenyl chloride under inert atmosphere.
- Purify the resulting N-thioaryl benzenesulfonylimine via column chromatography and vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers tangible benefits that directly impact the bottom line and operational resilience. The shift towards reagents that are insensitive to air and water fundamentally alters the risk profile of the manufacturing process, reducing the likelihood of batch failures due to environmental exposure. This reliability translates into more predictable production schedules and reduced waste, which are critical metrics for supply chain heads managing just-in-time inventory systems. Moreover, the elimination of sensitive reagents simplifies the regulatory compliance landscape regarding hazardous material transport and storage, potentially lowering insurance and logistics costs associated with specialized containment.
- Cost Reduction in Manufacturing: The synthetic route described eliminates the need for expensive transition metal catalysts and the associated downstream purification steps required to remove trace metals to ppm levels. By relying on organic reagents and simple acid-base chemistry, the process significantly reduces the cost of goods sold (COGS). Additionally, the mild reaction conditions (room temperature) minimize energy consumption for heating or cooling, contributing to substantial cost savings in utility expenses over long production runs. The high chemical yields reported in the examples further enhance the economic viability by maximizing the output per unit of raw material input.
- Enhanced Supply Chain Reliability: The stability of the N-thioaryl benzenesulfonylimine reagent ensures a longer shelf life compared to traditional sulfenyl chlorides, allowing for larger batch production and strategic stockpiling without the risk of rapid degradation. This stability mitigates the risk of supply disruptions caused by reagent spoilage during transit or storage. Furthermore, the starting materials, such as bisbenzenesulfonimide and various substituted benzene sulfenyl chlorides, are commercially accessible, ensuring a robust upstream supply chain that is less susceptible to single-source bottlenecks.
- Scalability and Environmental Compliance: The process utilizes standard solvents like dichloromethane and tetrahydrofuran, which are well-understood in industrial waste management systems, facilitating easier compliance with environmental regulations. The absence of heavy metals simplifies the wastewater treatment process, reducing the load on effluent treatment plants. The straightforward workup procedure, involving concentration and column chromatography, is easily adaptable to continuous flow chemistry or large-scale batch reactors, supporting the commercial scale-up of complex pharmaceutical intermediates without requiring bespoke engineering solutions.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common inquiries based on the patent specifications. These insights address the practical aspects of handling, reactivity, and scope, ensuring that stakeholders have a clear understanding of the reagent's capabilities and limitations.
Q: What are the stability advantages of N-thioaryl benzenesulfonylimine over traditional reagents?
A: Unlike traditional benzene sulfenyl chlorides which possess unstable S-Cl bonds and are sensitive to moisture, N-thioaryl benzenesulfonylimine features a stable N-S bond. This structural modification renders the reagent insensitive to air and water, significantly simplifying storage and handling protocols in industrial settings.
Q: Can this reagent be used for lactonization reactions?
A: Yes, the patent explicitly demonstrates the utility of this reagent in thioarylation/lactonization reactions. For instance, it successfully facilitates the conversion of 4-phenyl-4-pentenoic acid into lactonization products with high efficiency under mild acidic conditions.
Q: What are the typical reaction conditions for synthesizing this reagent?
A: The synthesis operates under remarkably mild conditions. The initial salt formation occurs at room temperature in THF, and the subsequent substitution reaction proceeds in dichloromethane, transitioning from 0°C to room temperature, avoiding the need for extreme cryogenic or high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Thioaryl Benzenesulfonylimine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced reagents like N-thioaryl benzenesulfonylimine in accelerating drug discovery and process development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory curiosity to commercial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required by global regulatory bodies. We understand that consistency is key in pharmaceutical manufacturing, and our commitment to quality assurance ensures that your supply chain remains uninterrupted.
We invite you to collaborate with us to leverage this innovative chemistry for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific synthesis requirements, identifying opportunities to optimize your current processes. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical intermediates can drive value and efficiency for your organization.
