Advanced NHC-Catalyzed Synthesis of Rolipram Intermediates for Commercial Scale-Up
Advanced NHC-Catalyzed Synthesis of Rolipram Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for producing complex active pharmaceutical ingredients (APIs) and their precursors. A groundbreaking approach detailed in patent CN112479966A introduces a novel method for synthesizing rolipram, a selective phosphodiesterase inhibitor with significant potential in treating autoimmune and neurodegenerative diseases. This technology leverages the power of N-heterocyclic carbene (NHC) organocatalysis to achieve high stereoselectivity under remarkably mild conditions. Unlike traditional methods that rely on stoichiometric chiral auxiliaries or expensive transition metals, this process utilizes a desymmetrization strategy that is both atom-economical and operationally simple. For procurement and supply chain leaders, this represents a pivotal shift towards more reliable rolipram intermediate supplier networks that can deliver high-purity materials with reduced environmental impact.
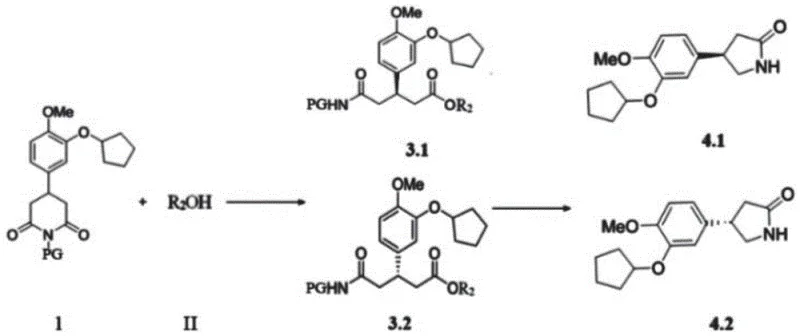
The core innovation lies in the reaction between a substituted glutarimide compound and an alcohol derivative, catalyzed by specific NHC structures. This transformation generates chiral intermediates with exceptional enantiomeric purity, setting the stage for the final assembly of the rolipram scaffold. The versatility of this method allows for a wide range of substituents, ensuring that diverse analogues can be accessed efficiently. By mastering this chemistry, manufacturers can secure a robust supply chain for high-purity pharmaceutical intermediates, mitigating risks associated with volatile metal prices and complex waste disposal protocols inherent in older synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral rolipram and its analogues has been plagued by significant technical and economic hurdles. Traditional routes often necessitate the use of quantitative chiral auxiliary reagents, which add substantial mass and cost to the process without becoming part of the final product. Furthermore, many established methods depend heavily on precious transition metal catalysts, such as rhodium or palladium complexes, for asymmetric hydrogenation or coupling reactions. These metals are not only expensive and subject to market fluctuations but also pose severe challenges regarding residual metal limits in the final API. The removal of trace metals requires additional purification steps, such as scavenging or recrystallization, which inevitably reduce overall yield and extend production timelines. Additionally, some conventional pathways involve harsh reaction conditions or hazardous reagents that complicate safety management and increase the burden on waste treatment facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN112479966A offers a streamlined alternative that addresses these pain points directly. By employing an organic small-molecule N-heterocyclic carbene catalyst, specifically the indane-fused NHC B structure, the process achieves high levels of stereocontrol without the need for stoichiometric chiral sources. The reaction proceeds smoothly at temperatures ranging from minus 30 °C to 30 °C, utilizing common bases like potassium carbonate and solvents such as dichloromethane. This mildness preserves sensitive functional groups and minimizes energy consumption. The subsequent steps involve a palladium-catalyzed deprotection followed by oxidation with hypervalent iodine reagents, leading to the final cyclized product. This sequence not only simplifies the operational workflow but also enhances the overall atom economy, making it an ideal candidate for cost reduction in pharmaceutical manufacturing.
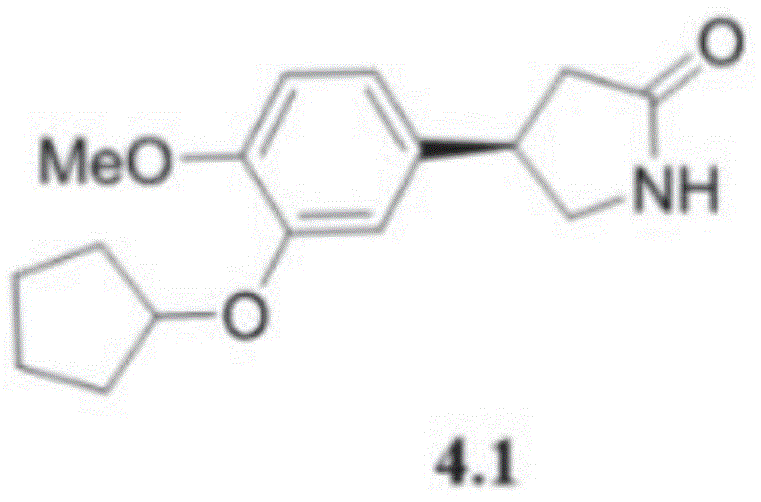
Mechanistic Insights into NHC-Catalyzed Desymmetrization
The success of this synthetic route hinges on the unique ability of the NHC catalyst to differentiate between enantiotopic groups within the prochiral glutarimide substrate. The catalyst, typically featuring a bulky mesityl group, creates a highly defined chiral environment that directs the nucleophilic attack of the alcohol to one specific face of the molecule. This desymmetrization event is the critical stereochemistry-determining step, establishing the chiral center that defines the biological activity of the final drug. The mechanism likely involves the formation of a transient acyl azolium intermediate, which activates the carbonyl group towards nucleophilic substitution while simultaneously shielding one side of the molecule sterically. This precise control ensures that the resulting ester intermediates possess high enantiomeric excess, often exceeding 90% ee, which is crucial for meeting regulatory standards for chiral drugs.
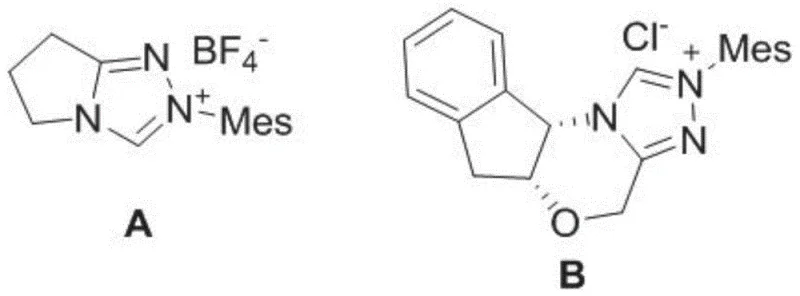
Furthermore, the choice of catalyst structure plays a pivotal role in impurity control. The patent highlights that catalyst A fails to promote the reaction effectively, whereas catalyst B and its enantiomer ent-NHC B provide excellent results. This specificity suggests that the rigid indane backbone of catalyst B is essential for maintaining the correct geometry during the catalytic cycle. By minimizing side reactions and racemization, this mechanism inherently reduces the formation of diastereomeric impurities that are notoriously difficult to separate. For R&D directors, understanding this mechanistic nuance is vital for troubleshooting and optimizing the process during scale-up, ensuring that the impurity profile remains within acceptable limits throughout the production lifecycle.
How to Synthesize Chiral Rolipram Intermediates Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable intermediates with high fidelity. The process begins with the preparation of the glutarimide substrate, followed by the key NHC-catalyzed ring-opening esterification. Detailed standard operating procedures for this transformation, including precise molar ratios and workup techniques, are essential for reproducibility. Following the initial catalytic step, the intermediate undergoes a series of transformations including hydrogenolysis and oxidative cyclization to yield the final lactam structure. For a comprehensive guide on executing these steps with maximum efficiency and safety, please refer to the standardized synthesis protocol below.
- React glutarimide substrate with alcohol using NHC B catalyst and K2CO3 base in DCM at temperatures between -30°C and 30°C.
- Perform hydrogenation or ammonium acetate treatment with palladium catalyst to remove protecting groups.
- Oxidize using hypervalent iodine reagents followed by base-mediated cyclization to yield the final rolipram scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this NHC-catalyzed synthesis route offers profound strategic benefits for procurement and supply chain management teams looking to optimize their sourcing strategies. The shift from metal-dependent catalysis to organocatalysis fundamentally alters the cost structure and risk profile of the supply chain. By removing the dependency on scarce and volatile precious metals for the key stereoselective step, manufacturers can stabilize their raw material costs and reduce exposure to geopolitical supply disruptions. Moreover, the simplified purification requirements translate directly into shorter cycle times and higher throughput, allowing suppliers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive chiral auxiliaries and the reduced loading of precious metal catalysts lead to significant savings in raw material expenditures. Since the NHC catalyst is used in sub-stoichiometric amounts and can potentially be recovered or synthesized from cheaper precursors, the overall cost of goods sold is drastically lowered. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further contributing to operational cost efficiencies without compromising on yield or quality.
- Enhanced Supply Chain Reliability: The substrates required for this reaction, such as substituted glutarimides and simple alcohols, are readily available from multiple global vendors, reducing the risk of single-source bottlenecks. The robustness of the reaction across a wide temperature range and with various bases means that production is less susceptible to minor variations in utility availability or environmental conditions. This resilience ensures a consistent and uninterrupted flow of high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The use of common organic solvents like dichloromethane and tetrahydrofuran facilitates easy scale-up using existing infrastructure, avoiding the need for specialized reactor modifications. Furthermore, the organocatalytic nature of the key step aligns with green chemistry principles by reducing heavy metal waste generation. This simplifies effluent treatment processes and helps manufacturers meet increasingly stringent environmental regulations, thereby avoiding potential fines and reputational damage associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this advanced synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios.
Q: What are the advantages of using NHC catalysts over transition metals for rolipram synthesis?
A: NHC catalysts eliminate the need for expensive and toxic transition metals in the critical stereoselective step, significantly reducing heavy metal impurities and lowering raw material costs while maintaining high enantiomeric excess.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process utilizes mild reaction conditions (-30°C to 30°C) and common organic solvents like DCM and THF, making it highly suitable for large-scale commercial manufacturing without requiring specialized cryogenic equipment.
Q: What level of stereocontrol does this method achieve?
A: The method demonstrates excellent stereoselectivity, achieving enantiomeric excess (ee) values up to 95% for key intermediates, which simplifies downstream purification and ensures high-quality API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rolipram Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this NHC-catalyzed technology for the production of high-value neurological therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of rolipram intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this cutting-edge synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
