Advanced Synthesis of N-Thiocyanobenzenesulfonimide for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Synthesis of N-Thiocyanobenzenesulfonimide for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The landscape of organic synthesis is constantly evolving, driven by the need for more efficient and versatile reagents that can streamline the production of complex molecules. A significant breakthrough in this domain is documented in patent CN111217733B, which details the preparation and application of N-thiocyanobenzenesulfonimide, a novel electrophilic thiocyanating agent. This compound addresses a critical gap in synthetic chemistry, where active electrophilic thiocyanide reagents have historically been scarce compared to their nucleophilic counterparts. For R&D directors and process chemists, the introduction of such a reagent opens new avenues for constructing organosulfur cyanide compounds, which are pivotal intermediates in the synthesis of bioactive natural products and pharmaceutical agents. The technology described offers a robust pathway to access these valuable structures under remarkably mild conditions, ensuring high purity and yield while minimizing operational hazards associated with harsh reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of organosulfur cyanide compounds has relied heavily on nucleophilic thiocyanide reagents, which function by capturing cationic intermediates. While effective in certain contexts, this approach limits the scope of substrates that can be utilized, particularly when dealing with electron-deficient systems that require an electrophilic attack to initiate the reaction. Furthermore, existing methods for generating electrophilic thiocyanating species often involve unstable precursors, hazardous reaction conditions, or complex multi-step procedures that result in poor atom economy and difficult purification profiles. The lack of stable, high-activity electrophilic reagents has slowed the development of efficient synthetic routes for various sulfur-containing heterocycles and functionalized molecules, creating a bottleneck in the manufacturing of advanced pharmaceutical intermediates and fine chemicals that demand precise structural control.
The Novel Approach
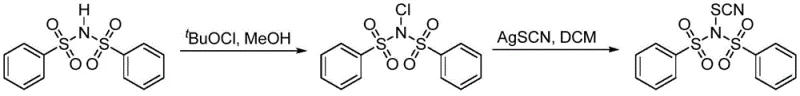
The methodology outlined in the patent presents a transformative solution by introducing a two-step synthesis that converts readily available bis(benzenesulfonyl)imide into the highly active N-thiocyanobenzenesulfonimide. This process leverages the high reactivity of tert-butyl hypochlorite to generate an N-chloro intermediate, which is subsequently displaced by silver thiocyanate to install the thiocyanate group. The elegance of this route lies in its operational simplicity and the driving force provided by the precipitation of silver chloride, which pushes the equilibrium towards the desired product. By utilizing common solvents like methanol and dichloromethane at room temperature, the process eliminates the need for extreme temperatures or pressures, thereby enhancing safety and scalability for industrial applications.
Mechanistic Insights into Electrophilic Thiocyanation and Functionalization
The core innovation of this technology is the generation of a potent electrophilic sulfur center within the sulfonimide framework. In the final product, the nitrogen atom is flanked by two strongly electron-withdrawing sulfonyl groups, which significantly enhances the leaving group ability of the benzenesulfonimide moiety during nucleophilic attack. When employed in reactions such as thiocyanation/lactonization, the reagent facilitates the formation of a thiocyanato group on the substrate while simultaneously promoting cyclization. This dual functionality is particularly valuable for constructing complex ring systems found in drug candidates. The mechanism typically involves the activation of an alkene or nucleophile by a Lewis acid or promoter, followed by the transfer of the SCN+ equivalent from the reagent, resulting in high regioselectivity and stereocontrol that is difficult to achieve with standard nucleophilic salts.
From an impurity control perspective, the reaction design inherently minimizes side products. The use of silver thiocyanate ensures that the chloride leaving group from the intermediate is sequestered as insoluble silver chloride, which is easily removed by filtration. This prevents the accumulation of halogenated impurities that often plague chlorination-based syntheses. Additionally, the mild conditions prevent the decomposition of the sensitive thiocyanate group, which can otherwise degrade into isothiocyanates or other sulfur species under thermal stress. The result is a crude product profile that is exceptionally clean, reducing the burden on downstream purification steps like column chromatography or recrystallization, which is a critical factor for maintaining cost-efficiency in large-scale manufacturing environments.
How to Synthesize N-Thiocyanobenzenesulfonimide Efficiently
The synthesis protocol described in the patent is designed for reproducibility and ease of execution, making it highly suitable for translation from the laboratory to pilot plant scales. The process begins with the dissolution of the starting imide in methanol, followed by the controlled addition of the chlorinating agent. This step is rapid, completing within minutes, and yields a solid intermediate that can be isolated by simple filtration. The second step involves reacting this intermediate with silver thiocyanate in anhydrous dichloromethane under an inert atmosphere to prevent moisture-induced degradation. The reaction proceeds in the dark to avoid photolytic decomposition, a standard precaution for silver salts and sensitive organic halides. Detailed standardized operating procedures for this synthesis are provided below to ensure consistent quality and yield.
- React bis(benzenesulfonyl)imide with tert-butyl hypochlorite in methanol at room temperature to form N-chlorobenzenesulfonimide.
- Filter and dry the N-chloro intermediate, then dissolve in dry dichloromethane under inert atmosphere.
- Add silver thiocyanate and react in the dark at room temperature, followed by filtration and low-temperature concentration to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers tangible benefits in terms of cost structure and operational reliability. The reliance on commercially available starting materials like bis(benzenesulfonyl)imide and tert-butyl hypochlorite ensures a stable supply chain without dependence on exotic or single-source precursors. The high yields reported in the patent examples indicate a highly efficient conversion of raw materials into the final product, which directly correlates to reduced waste generation and lower raw material costs per kilogram of output. Furthermore, the elimination of cryogenic conditions or high-pressure equipment reduces the capital expenditure required for manufacturing infrastructure, allowing for more flexible production scheduling and faster response times to market demands.
- Cost Reduction in Manufacturing: The process significantly lowers manufacturing costs by eliminating the need for expensive transition metal catalysts or complex ligand systems often required in modern C-H functionalization. The precipitation of silver chloride serves as a self-cleaning mechanism, reducing the solvent and energy consumption associated with extensive aqueous workups or chromatographic purification. Additionally, the short reaction times mean that reactor turnover rates can be maximized, allowing facilities to produce larger volumes of the reagent within the same timeframe, effectively spreading fixed operational costs over a greater output and driving down the unit price for downstream users.
- Enhanced Supply Chain Reliability: By utilizing robust chemical transformations that are insensitive to minor fluctuations in ambient temperature, the supply of this critical intermediate becomes more predictable and resilient. The stability of the final product, when stored under recommended anhydrous and oxygen-free conditions, allows for longer shelf-life and easier logistics management compared to unstable liquid reagents. This reliability is crucial for pharmaceutical manufacturers who require consistent quality and uninterrupted supply to maintain their own production schedules for active pharmaceutical ingredients, mitigating the risk of costly delays caused by reagent shortages or quality failures.
- Scalability and Environmental Compliance: The synthesis is inherently scalable due to its exothermic nature being manageable at room temperature and the use of common organic solvents that are easily recovered and recycled. The absence of toxic heavy metal residues in the final organic product simplifies regulatory compliance regarding residual solvents and metal impurities, which is a stringent requirement for pharmaceutical grade materials. Moreover, the solid byproduct (silver chloride) can be collected and processed for silver recovery, adding a potential revenue stream or cost offset that further improves the environmental and economic footprint of the overall manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of N-thiocyanobenzenesulfonimide. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the reagent's capabilities and limitations. Understanding these details is essential for process engineers and buyers evaluating the feasibility of integrating this new chemistry into their existing workflows.
Q: What distinguishes N-thiocyanobenzenesulfonimide from traditional thiocyanation reagents?
A: Unlike traditional nucleophilic thiocyanide reagents, N-thiocyanobenzenesulfonimide acts as a highly active electrophilic thiocyanating agent, enabling reactions with electron-rich substrates that were previously difficult to functionalize efficiently.
Q: What are the key advantages of the synthesis method described in CN111217733B?
A: The method utilizes mild reaction conditions (room temperature), short reaction times (5-10 minutes for the first step), and achieves high chemical yields (up to 95% for the intermediate and 90% for the final product), significantly simplifying purification.
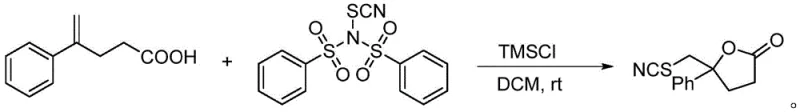
Q: Can this reagent be used for lactonization reactions?
A: Yes, the patent explicitly demonstrates its efficacy in thiocyanation/lactonization tandem reactions, such as converting 4-phenyl-4-pentenoic acid into the corresponding lactone with high efficiency using TMSCl as a promoter.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Thiocyanobenzenesulfonimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-performance reagents like N-thiocyanobenzenesulfonimide in the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the structural integrity and impurity profile of every batch, guaranteeing that the electrophilic activity of the reagent is preserved for your most sensitive synthetic transformations. We are committed to supporting your R&D efforts with reliable supply and technical expertise.
We invite you to contact our technical procurement team to discuss how this innovative reagent can optimize your synthetic routes. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to this electrophilic thiocyanation method might reduce your overall process costs. We are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, helping you accelerate your timeline from discovery to commercial manufacturing with confidence.
