Advanced One-Pot Synthesis of Rasagiline Racemic Intermediate for Commercial Scale-up
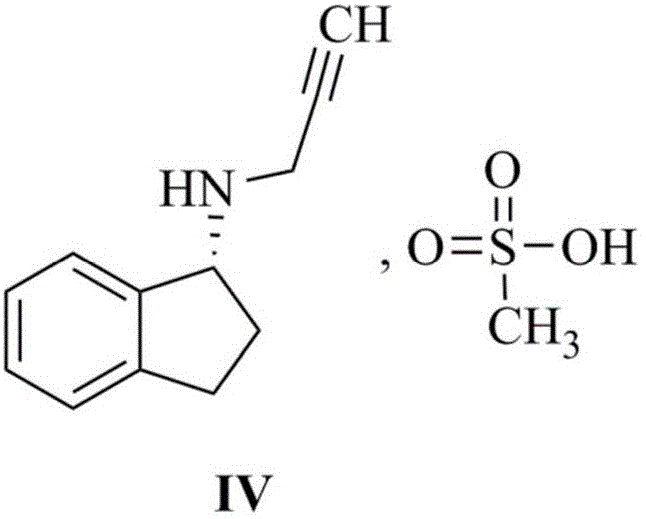
The pharmaceutical landscape for Parkinson's disease treatment continues to evolve, with Rasagiline Mesylate standing out as a critical selective MAO-B inhibitor. Patent CN110776429A introduces a transformative approach to synthesizing the key racemic intermediate, N-(2-propargyl)-2,3-dihydro-1H-inden-1-amine, which serves as the foundational building block for this vital medication. This innovation addresses long-standing inefficiencies in reductive amination processes, shifting away from cumbersome multi-step isolations toward a streamlined, high-efficiency one-pot methodology. For global procurement leaders and R&D directors, this patent represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates. By leveraging titanium-based dehydration chemistry combined with controlled borohydride reduction, the process achieves exceptional yields and purity profiles that were previously difficult to attain consistently. This technical breakthrough not only enhances the economic viability of producing Rasagiline but also ensures a more reliable supply of this essential neuroprotective agent for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
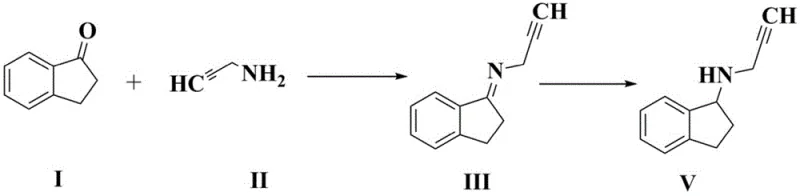
Historically, the synthesis of N-(2-propargyl)-2,3-dihydro-1H-inden-1-amine has been plagued by significant chemical and operational challenges that hinder efficient commercial manufacturing. Traditional two-step methods involve the initial formation of an imine intermediate, specifically N-(2,3-dihydro-1H-inden-ylidene)-2-propargyl-1-amine, followed by a separate reduction step. However, this imine intermediate is chemically unstable and highly susceptible to hydrolysis, reverting back to the starting materials of 1-indanone and propargylamine under standard conditions. This instability leads to notoriously low yields and generates a complex mixture of by-products that complicate downstream purification. Furthermore, existing one-pot variations reported in prior art, such as those utilizing acetic acid and sodium borohydride without effective dehydration control, often result in products with deep, undesirable coloration that is extremely difficult to remove. These factors collectively render conventional methods unsuitable for large-scale amplification, as they incur high material costs and fail to meet the stringent purity specifications required for pharmaceutical grade intermediates.
The Novel Approach
The methodology disclosed in Patent CN110776429A fundamentally reengineers the reaction pathway by integrating a specialized dehydrating agent directly into the reductive amination sequence. By employing titanium alkoxides, specifically tetraisopropyl titanate, the process effectively scavenges water generated during imine formation, driving the equilibrium forward without the need to isolate the unstable intermediate. This allows the reduction to proceed immediately in the same vessel using a sodium borohydride and acetic acid system, creating a seamless transition from ketone to amine. The result is a robust one-pot synthesis that operates under mild temperatures, typically between 0°C and 50°C, minimizing thermal degradation and side reactions. This novel approach not only simplifies the operational workflow by eliminating intermediate filtration and drying steps but also dramatically improves the physical appearance of the crude product, yielding a reddish-brown liquid rather than the dark tars associated with older techniques. Consequently, this method lays a superior foundation for the subsequent resolution and salt formation steps required to produce the final active pharmaceutical ingredient.
Mechanistic Insights into Titanium-Mediated Reductive Amination
The core innovation of this synthesis lies in the synergistic interaction between the titanium-based dehydrating agent and the borohydride reducing system within a protic or semi-protic solvent environment. Tetraisopropyl titanate acts as a potent Lewis acid and water scavenger, coordinating with the carbonyl oxygen of 1-indanone and the amine nitrogen of propargylamine to facilitate the elimination of water and the formation of the imine species in situ. Unlike traditional methods where water accumulation reverses the imine formation, the titanium species sequesters the water, effectively locking the equilibrium toward the imine and making it immediately available for reduction. Simultaneously, the sodium borohydride, activated by glacial acetic acid, generates a reactive hydride source capable of reducing the transient imine bond to a stable secondary amine. This dual-action mechanism ensures that the concentration of the free imine remains low enough to prevent polymerization or hydrolysis, yet high enough to sustain the reduction rate. The careful control of stoichiometry, particularly the molar ratio of the dehydrating agent to the ketone, is critical to maximizing conversion while minimizing the formation of titanium-containing sludge that could complicate workup.
Impurity control is another critical aspect where this mechanistic design excels, particularly regarding the suppression of over-reduction or alkyne hydrogenation side reactions. The reaction conditions are meticulously tuned to maintain a temperature range of 0°C to 30°C during the addition of reagents, which kinetically favors the reduction of the C=N bond over the C≡C triple bond present in the propargyl group. In less controlled environments, the alkyne moiety is vulnerable to partial hydrogenation, leading to allyl or propyl impurities that are structurally similar and difficult to separate. By utilizing tetrahydrofuran or ethanol as the solvent and strictly regulating the addition rate of the amine and dehydrating agent, the process maintains a homogeneous reaction phase that promotes selectivity. Furthermore, the quenching procedure using potassium carbonate solution is designed to decompose excess borohydride and hydrolyze titanium complexes into filterable solids, ensuring that the final organic extract is free from metal contaminants. This rigorous control over the reaction micro-environment results in an intermediate with HPLC purity exceeding 97%, significantly reducing the burden on downstream crystallization and purification units.
How to Synthesize N-(2-propargyl)-2,3-dihydro-1H-inden-1-amine Efficiently
Implementing this advanced synthesis route requires precise adherence to the sequential addition of reagents and temperature controls outlined in the patent examples to ensure reproducibility and safety. The process begins with the preparation of the active reducing agent by suspending sodium borohydride in tetrahydrofuran and carefully adding glacial acetic acid under cooling, a step that generates hydrogen gas and requires adequate ventilation and monitoring. Once the reducing system is stabilized, the starting materials, 1-indanone and propargylamine, are introduced along with the tetraisopropyl titanate, initiating the cascade of dehydration and reduction. The reaction mixture is then maintained under agitation for a period of 6 to 10 hours, allowing sufficient time for the complete conversion of the ketone to the desired amine racemate. Following the reaction completion, a standardized workup protocol involving aqueous quenching, pH adjustment, and solvent extraction is employed to isolate the product in high yield. For a detailed breakdown of the specific operational parameters, stoichiometric ratios, and safety precautions, please refer to the standardized synthesis guide below.
- Prepare the reducing system by suspending sodium borohydride in tetrahydrofuran and adding glacial acetic acid, stirring at 0-30°C for 0.5 to 2 hours to generate the active reducing species in situ.
- Add 1-indanone, propargylamine, and tetraisopropyl titanate sequentially to the reaction mixture, maintaining the temperature between 0-50°C to facilitate imine formation and immediate reduction.
- Quench the reaction with potassium carbonate solution, extract the organic phase, adjust pH to alkaline conditions, and purify via solvent evaporation to obtain the high-purity reddish-brown liquid intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this improved synthesis method offers substantial benefits that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The elimination of the intermediate isolation step drastically reduces the consumption of solvents, filtration media, and energy associated with drying and handling solid intermediates, leading to significant cost reduction in pharmaceutical intermediates manufacturing. Moreover, the enhanced stability of the reaction profile minimizes the risk of batch failures due to intermediate hydrolysis, thereby improving overall equipment effectiveness and throughput capacity. The ability to produce the intermediate with superior color and purity right from the crude stage reduces the reliance on expensive decolorization agents like activated carbon, which often adsorb product and lower overall recovery rates. These efficiencies translate into a more predictable production schedule and a lower cost of goods sold, making the final API more competitive in the global marketplace.
- Cost Reduction in Manufacturing: The streamlined one-pot process eliminates the need for separate reaction vessels and unit operations dedicated to imine isolation, which traditionally consume significant time and resources. By combining dehydration and reduction into a single operational window, manufacturers can reduce labor hours and utility consumption, such as heating and cooling loads, across the production cycle. The high yield of over 92% reported in the patent examples means that less raw material is wasted, directly lowering the variable cost per kilogram of the produced intermediate. Additionally, the simplified workup procedure reduces the volume of waste streams generated, lowering disposal costs and environmental compliance burdens associated with hazardous waste management.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent supply of high-quality intermediate, mitigating the risks associated with batch-to-batch variability that often plague complex amine syntheses. Since the raw materials, including 1-indanone and propargylamine, are commodity chemicals with established supply chains, the risk of raw material shortage is minimized compared to routes requiring exotic or custom-synthesized reagents. The scalability demonstrated in the patent, with successful runs in 50L reactors, confirms that the technology can be transferred to multi-ton production scales without losing efficiency or quality. This reliability allows procurement managers to negotiate longer-term contracts with greater confidence, knowing that the supplier can meet demand fluctuations without compromising on delivery timelines.
- Scalability and Environmental Compliance: The process utilizes common organic solvents like tetrahydrofuran and ethanol, which are easily recoverable and recyclable through standard distillation infrastructure, supporting green chemistry initiatives. The absence of heavy metal catalysts, relying instead on titanium alkoxides which hydrolyze to benign titanium dioxide, simplifies the regulatory filing and reduces the burden of residual metal testing in the final drug substance. The high purity of the crude product reduces the need for multiple recrystallizations, which in turn lowers the total solvent intensity of the manufacturing process. This alignment with environmental, social, and governance (ESG) goals makes the supply chain more sustainable and attractive to partners who prioritize eco-friendly manufacturing practices in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology for Rasagiline intermediates. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is crucial for technical teams evaluating the transfer of this technology to their own production facilities or for procurement specialists assessing supplier capabilities. The answers reflect the specific advantages of the titanium-mediated one-pot method over legacy processes, highlighting improvements in yield, purity, and operational simplicity.
Q: What are the key advantages of the one-pot method over the traditional two-step synthesis for Rasagiline intermediates?
A: The one-pot method eliminates the need to isolate the unstable imine intermediate (Formula III), which is prone to hydrolysis and results in low yields in traditional methods. By using a titanium-based dehydrating agent in situ, the reaction drives directly to the amine product with significantly higher yields (over 92%) and superior color quality, reducing purification costs.
Q: How does the new process address the issue of dark coloration often seen in reductive amination?
A: Traditional methods often produce dark-colored by-products that are difficult to decolorize, requiring extensive charcoal treatment which lowers yield. The optimized protocol in Patent CN110776429A utilizes specific molar ratios of tetraisopropyl titanate and controlled low-temperature addition, resulting in a reddish-brown liquid that is much easier to purify to HPLC standards above 97% without complex decolorization steps.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability through examples conducted in 50L glass reactors, achieving consistent yields of approximately 92.6% and optical purity suitable for subsequent resolution. The use of common solvents like THF and ethanol, along with standard workup procedures involving aqueous quenching and extraction, ensures the process is robust and adaptable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rasagiline Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of essential Parkinson's disease medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest international standards. By adopting the advanced one-pot synthesis technology described in Patent CN110776429A, we can offer our partners a superior product profile with improved cost efficiency and supply continuity. Our facility is equipped to handle the specific solvent systems and temperature controls required for this titanium-mediated process, guaranteeing consistent quality and reliability for your long-term projects.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain strategy. Request a Customized Cost-Saving Analysis to understand the specific economic benefits of switching to this high-yield method for your requirements. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your volume needs, ensuring that you have all the necessary information to make informed sourcing decisions. Partner with us to leverage cutting-edge chemical manufacturing capabilities that drive value and security in your pharmaceutical supply network.
