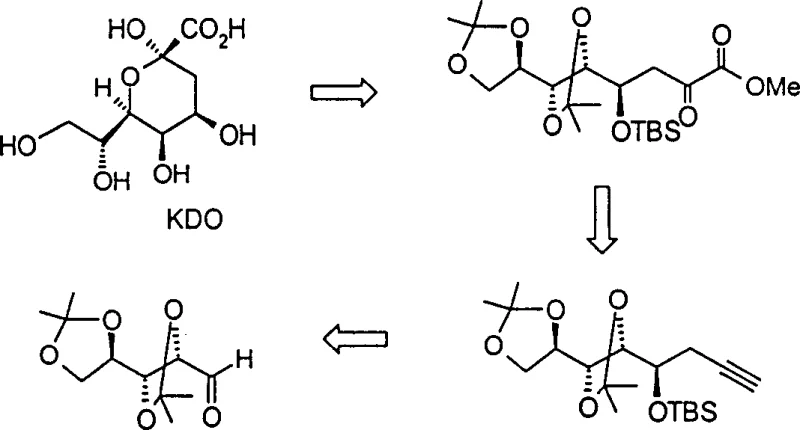
Advanced Synthesis of Alpha-Carbonyl Esters for High-Carbon Sugar Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex molecular architectures, particularly those serving as critical building blocks for bioactive compounds. Patent CN1286799C introduces a transformative methodology for the preparation of alpha-carbonyl ester compounds, which serve as indispensable precursors in the total synthesis of high-carbon sugars such as KDO (3-deoxy-D-manno-2-octulosonic acid) and sialic acid analogues like Zanamivir. Traditionally, the acquisition of these high-value intermediates relied heavily on the extraction of expensive natural products or convoluted multi-step syntheses that suffered from poor atom economy and low overall yields. This patent delineates a robust, two-step protocol involving the halogenation of terminal alkynes followed by a controlled oxidation reaction, offering a scalable alternative that addresses the longstanding challenges of cost and availability in carbohydrate chemistry. By shifting the paradigm from biological extraction to concise chemical synthesis, this technology provides a reliable foundation for the commercial production of antiviral and antibacterial agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-carbon sugars and their derivatives has been plagued by significant economic and technical hurdles that hinder large-scale manufacturing. Conventional routes often depend on chiral pool starting materials derived from nature, such as sialic acid, which commands an exorbitant price due to the complexity of isolation and purification from biological sources. Furthermore, traditional chemical approaches frequently necessitate harsh reaction conditions, including strong acids or bases and extreme temperatures, which can lead to the epimerization of sensitive stereocenters or the degradation of labile protecting groups essential for sugar chemistry. These inefficiencies result in prolonged production timelines, increased waste generation, and a supply chain that is vulnerable to fluctuations in the availability of natural raw materials. For procurement managers and supply chain heads, these factors translate into unpredictable lead times and inflated costs that make the final therapeutic products less accessible to the global market.
The Novel Approach
In stark contrast, the methodology disclosed in CN1286799C offers a streamlined solution that bypasses these traditional bottlenecks through a clever manipulation of terminal alkyne functionality. The core innovation involves converting a terminal alkyne into an alkynyl halide intermediate, which is subsequently oxidized to yield the desired alpha-carbonyl ester with high fidelity. This approach is characterized by its operational simplicity and mild reaction conditions, typically proceeding at temperatures ranging from -78°C to room temperature, thereby preserving the integrity of complex molecular scaffolds. The versatility of this method is evident in its compatibility with a wide array of substrates, including those bearing aryl groups or multi-hydroxyl substituted alkyl chains protected as ethers, esters, or acetals. By decoupling the synthesis from expensive natural precursors and utilizing readily available chemical reagents, this novel route significantly enhances the feasibility of cost reduction in high-carbon sugar manufacturing, making it an attractive option for industrial adoption.
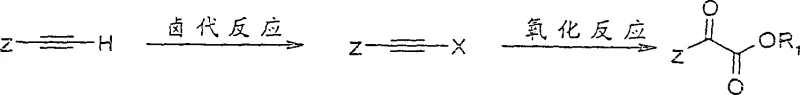
Mechanistic Insights into Halogenation-Oxidation Cascade
The chemical elegance of this process lies in the precise control exerted during the two distinct transformation stages, ensuring high selectivity and yield. The first stage, halogenation, activates the terminal alkyne by introducing a halogen atom (chlorine, bromine, or iodine) at the terminal position. This can be achieved through various reagent systems, such as the reaction with N-bromosuccinimide (NBS) catalyzed by silver nitrate in acetone, or the use of iodine and morpholine in benzene at elevated temperatures. The formation of the alkynyl halide is a critical activation step that renders the triple bond susceptible to the subsequent oxidative cleavage. The second stage involves a carefully buffered oxidation reaction where the alkynyl halide is treated with a heptavalent inorganic manganese compound, such as potassium permanganate, in a mixed solvent system of water and a low-carbon alcohol. Crucially, the reaction pH is maintained between 6 and 8 using monovalent metal bases and magnesium salts, which prevents the aggressive over-oxidation that typically leads to carboxylic acid formation or carbon chain scission. This mechanistic nuance allows for the isolation of the alpha-keto ester functionality rather than complete degradation, representing a significant advancement in oxidative synthetic methodology.
From an impurity control perspective, this mechanism offers distinct advantages over unbuffered oxidation protocols. The inclusion of magnesium salts and specific base ratios helps to moderate the reactivity of the permanganate species, minimizing the formation of side products such as cleaved carboxylic acids or polymerized byproducts. The patent data indicates that this controlled environment facilitates the production of intermediates with high stereochemical purity, which is paramount for the biological activity of the final sugar derivatives. For R&D directors focused on process robustness, the ability to tune the reaction outcome through pH adjustment and reagent stoichiometry provides a powerful tool for optimizing the impurity profile. Furthermore, the compatibility of this oxidative step with diverse protecting groups ensures that the stereochemical information encoded in the starting alkyne is retained throughout the synthesis, thereby reducing the need for costly downstream purification or recrystallization steps.
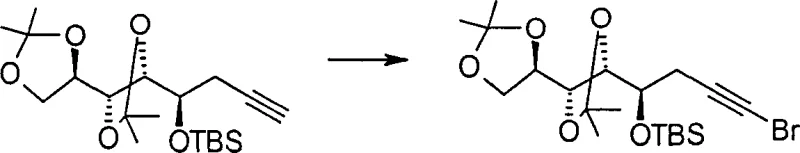
How to Synthesize Alpha-Carbonyl Esters Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize yield and safety. The process begins with the selection of an appropriate terminal alkyne substrate, which may be derived from cheap gluconolactone precursors via established propargylation reactions. The halogenation step should be conducted under anhydrous conditions for chlorination or bromination to prevent premature hydrolysis, whereas iodination may proceed in organic solvents like benzene. Following the isolation of the alkynyl halide, the oxidation step demands careful temperature control, typically initiating at 0°C to manage the exotherm associated with permanganate addition. Detailed standardized operating procedures regarding reagent addition rates, stirring speeds, and quenching protocols are essential to ensure reproducibility across different batches. For a comprehensive guide on the specific molar ratios, solvent choices, and workup techniques validated in the patent examples, please refer to the structured synthesis protocol below.
- Perform halogenation of the terminal alkyne using reagents like N-bromosuccinimide (NBS) with silver nitrate catalysis or iodine with morpholine to generate the alkynyl halide intermediate.
- Conduct the oxidation reaction in a mixed solvent system of water and low-carbon alcohol, utilizing potassium permanganate (KMnO4) with magnesium salts and monovalent metal bases to adjust pH.
- Isolate the final alpha-carbonyl ester product through standard aqueous workup, extraction with ethyl acetate, and purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and logistics, the adoption of this synthetic route presents a compelling value proposition centered on stability and efficiency. By replacing volatile natural extracts with stable, commodity-grade chemical reagents, manufacturers can insulate their supply chains from the biological variability and seasonal constraints that often plague fermentation-based or extraction-based processes. The reliance on common oxidants and halogenating agents means that raw material procurement becomes significantly more predictable, allowing for better inventory management and long-term contracting strategies. Moreover, the simplified two-step nature of the synthesis reduces the number of unit operations required, which directly correlates to lower capital expenditure on equipment and reduced energy consumption per kilogram of product. This streamlining of the manufacturing process not only accelerates time-to-market for new drug candidates but also enhances the overall resilience of the supply network against external disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive natural starting materials like sialic acid serves as the primary driver for cost optimization in this workflow. Instead of purchasing high-cost biological precursors, manufacturers can utilize inexpensive terminal alkynes and bulk chemical reagents, leading to substantial savings in raw material costs. Additionally, the high yields reported in the patent examples minimize the loss of valuable intermediates, further improving the cost-efficiency of the overall process. The reduction in processing steps also lowers labor and utility costs, contributing to a more competitive pricing structure for the final active pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: Sourcing chemicals from a broad base of industrial suppliers reduces the risk of single-source dependency that is common with specialized biological extracts. The reagents used in this protocol, such as potassium permanganate and N-bromosuccinimide, are widely produced and readily available in the global chemical market, ensuring consistent supply continuity. This reliability is crucial for maintaining uninterrupted production schedules, particularly for life-saving medications where supply shortages can have severe consequences. The robustness of the chemical synthesis also allows for easier scaling from gram to ton quantities without the need for complex bioreactor infrastructure.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of standard organic solvents facilitate straightforward scale-up from laboratory to commercial production volumes. The process avoids the generation of hazardous heavy metal waste often associated with other catalytic methods, simplifying waste treatment and disposal protocols. By adhering to greener chemistry principles through efficient atom utilization and reduced step counts, this method supports corporate sustainability goals and helps manufacturers meet increasingly stringent environmental regulations without compromising on output quality or volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-carbonyl ester synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the scope and limitations of the method. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the versatility of the chemistry and its alignment with modern pharmaceutical production standards.
Q: What are the primary advantages of this halogenation-oxidation method over traditional extraction?
A: This method replaces expensive natural extraction processes (like obtaining sialic acid from biological sources) with a concise two-step chemical synthesis. It utilizes readily available terminal alkynes and common oxidants, significantly reducing raw material costs and improving batch-to-batch consistency for industrial scale-up.
Q: Can this synthesis tolerate complex protecting groups found in sugar chemistry?
A: Yes, the patent explicitly demonstrates compatibility with various protecting groups including ethers (TBS, MOM, Bn), esters (Acetyl, Benzoyl), and acetals/ketals (Acetonide). The mild reaction conditions (0°C to room temperature) prevent the degradation of sensitive carbohydrate scaffolds during the transformation.
Q: What represents the key innovation in the oxidation step of this process?
A: The innovation lies in the direct oxidation of the alkynyl halide intermediate rather than the terminal alkyne itself. By using a buffered system with magnesium salts and monovalent metal bases to maintain a pH of 6-8, the process achieves high yields of the alpha-keto ester while minimizing over-oxidation or cleavage of the carbon chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Carbonyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Whether you require custom synthesis of complex alpha-carbonyl esters or large-scale supply of key sugar intermediates, our infrastructure is designed to support your project's specific needs with speed and precision.
We invite you to collaborate with us to leverage this innovative synthetic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us today to request specific COA data for our available intermediates and to discuss route feasibility assessments that can accelerate your path to market. Together, we can drive down costs and enhance the availability of vital pharmaceutical compounds through advanced chemical manufacturing.
