Revolutionizing Ketimine Production: A Scalable, Green Route for High-Value Pharmaceutical Intermediates
Revolutionizing Ketimine Production: A Scalable, Green Route for High-Value Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust, scalable, and environmentally benign synthetic routes for critical active pharmaceutical ingredient (API) intermediates. Patent CN1325381A introduces a transformative methodology for the preparation of ketimines, specifically targeting precursors for antidepressants like sertraline. This innovation addresses long-standing inefficiencies in traditional imine synthesis by shifting from hazardous aprotic systems to safer, cost-effective protic solvent environments. By leveraging simple alcohols and accessible acid catalysts, this technology offers a compelling alternative to legacy processes that rely on moisture-sensitive reagents and complex purification protocols. For R&D directors and procurement specialists alike, understanding this shift is crucial for optimizing supply chains and reducing the overall cost of goods sold (COGS) in antidepressant manufacturing.
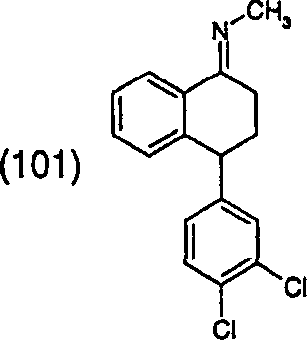
The core of this technological advancement lies in the conversion of tetralone derivatives into their corresponding N-methyl imines. As illustrated in the structural representation of Formula (101), the target molecule serves as a pivotal junction in the synthesis of sertraline, a widely prescribed SSRI. Traditional approaches to generating this functionality often involve harsh conditions that compromise safety and yield. In contrast, the patented method described in CN1325381A utilizes a reaction between a ketone of Formula (2) and methylamine within a protic solvent matrix. This fundamental change in reaction media not only mitigates the risks associated with flammable ethers and chlorinated solvents but also streamlines the isolation process through direct crystallization, thereby enhancing the operational efficiency of the entire production line.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical methods for synthesizing ketimines, such as those disclosed in US-A-4536518 and US-A-4855500, present significant logistical and economic hurdles for modern manufacturing. These legacy processes typically mandate the use of anhydrous aprotic solvents like tetrahydrofuran (THF) or dichloromethane, which are not only flammable and potentially toxic but also require rigorous drying protocols to prevent hydrolysis of the sensitive imine bond. Furthermore, these methods frequently employ titanium tetrachloride (TiCl4) or molecular sieves as promoters or water scavengers. TiCl4 is a corrosive, moisture-sensitive Lewis acid that generates substantial hazardous waste upon quenching, while molecular sieves introduce additional unit operations for filtration and regeneration. The necessity for low-temperature conditions in these traditional routes further escalates energy consumption and capital expenditure, rendering them less attractive for large-scale commercial deployment where margin compression is a constant concern.
The Novel Approach
The novel approach detailed in the patent data fundamentally reimagines the reaction environment by embracing protic solvents, specifically alcohols such as ethanol, isopropanol, or ethylene glycol. This shift eliminates the strict requirement for anhydrous conditions, allowing the reaction to proceed efficiently even in the presence of the water generated during imine formation. By operating at mild temperatures ranging from 20°C to 100°C, the process avoids the energy-intensive cooling steps associated with prior art. Moreover, the compatibility of this system with a wide array of inexpensive catalysts, including sulfonic acids and solid acid clays, removes the dependency on exotic or hazardous reagents. The ability to isolate the product directly via crystallization from the reaction mixture simplifies downstream processing, reducing solvent usage and waste generation while simultaneously improving the overall yield and purity profile of the intermediate.
Mechanistic Insights into Acid-Catalyzed Condensation in Protic Media
The mechanistic elegance of this synthesis relies on the acid-catalyzed condensation of the ketone with methylamine within a hydrogen-bonding solvent network. In this system, the protic solvent facilitates proton transfer events that are critical for the dehydration step of imine formation. Unlike aprotic systems where water must be physically scavenged to drive equilibrium, the protic environment stabilizes the transition states and intermediates through solvation, allowing the reaction to reach completion without aggressive water removal agents. The addition of catalysts such as p-toluenesulfonic acid or scandium triflate further lowers the activation energy, accelerating the rate of nucleophilic attack by the amine on the carbonyl carbon. This catalytic enhancement ensures high conversion rates even at lower temperatures, minimizing the formation of thermal degradation byproducts that often plague high-temperature syntheses.
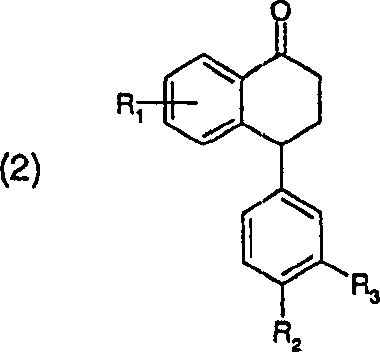
Impurity control is intrinsically built into this mechanism through the physical properties of the product in the chosen solvent. The ketimine product exhibits distinct solubility characteristics in alcohols compared to the starting ketone and potential side products. As the reaction progresses, the product often crystallizes directly from the medium, effectively pulling the equilibrium forward according to Le Chatelier's principle and physically separating the desired molecule from soluble impurities. Subsequent recrystallization steps, performed in the same or similar alcoholic solvents, leverage these solubility differences to exclude residual starting materials (often referred to as 'Sertralon' in the context of sertraline synthesis) and catalyst residues. This dual function of the solvent—as both a reaction medium and a purification agent—significantly reduces the complexity of the impurity profile, ensuring that the final intermediate meets the stringent quality standards required for subsequent hydrogenation and resolution steps in API synthesis.
How to Synthesize Sertraline Imine Efficiently
Implementing this synthesis route requires careful attention to solvent selection and catalyst loading to maximize yield and purity. The process begins by dissolving the tetralone precursor in a selected alcohol, followed by the introduction of methylamine, either as a gas or a solution. The reaction mixture is then heated to a temperature between 25°C and 65°C, optionally in the presence of a catalytic amount of acid. Detailed standard operating procedures regarding specific molar ratios, agitation speeds, and crystallization cooling gradients are essential for reproducibility. For a comprehensive guide on the precise execution of this protocol, including specific examples of catalyst screening and solvent optimization, please refer to the standardized synthesis steps outlined below.
- React the tetralone ketone precursor with methylamine in a protic solvent such as ethanol or isopropanol.
- Optionally add a catalyst such as p-toluenesulfonic acid or a Lewis acid to accelerate the condensation.
- Purify the resulting crude imine through recrystallization from the same alcoholic solvent to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this protic solvent methodology offers profound advantages for procurement managers and supply chain directors focused on cost reduction and reliability. The elimination of specialized reagents like titanium tetrachloride and molecular sieves directly translates to a simplified bill of materials (BOM), reducing raw material costs and minimizing the logistical burden of sourcing hazardous chemicals. Furthermore, the use of commodity solvents like ethanol and isopropanol ensures a stable and abundant supply chain, insulating production schedules from the volatility often seen in the market for specialized aprotic solvents. The simplified workup procedure, which avoids complex aqueous quenches and multiple extraction steps, significantly reduces utility consumption and waste disposal costs, contributing to a leaner and more sustainable manufacturing operation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with inexpensive, commodity-grade alternatives. By removing the need for titanium tetrachloride, manufacturers avoid the costs associated with handling corrosive materials and treating the resulting acidic waste streams. Additionally, the ability to recycle the filtrate for subsequent batches, as demonstrated in the patent examples, drastically reduces solvent consumption. This closed-loop capability minimizes raw material input per kilogram of product, leading to substantial operational savings without compromising on the quality or yield of the final intermediate.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of robust, non-sensitive chemistry that tolerates minor variations in feedstock quality better than moisture-sensitive legacy methods. Since the process does not require strictly anhydrous conditions, the risk of batch failure due to ambient humidity or solvent water content is significantly mitigated. This robustness ensures consistent production output and reduces the likelihood of costly delays or reworks. Moreover, the reliance on widely available alcohols and common acid catalysts means that supply disruptions for any single component are unlikely to halt production, providing a resilient foundation for long-term supply agreements.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of extreme operating conditions. The reaction proceeds safely at near-atmospheric pressure and moderate temperatures, eliminating the need for specialized high-pressure or cryogenic reactors. From an environmental standpoint, the reduction in hazardous waste generation and the use of greener solvents align with increasingly strict global regulatory frameworks. This compliance reduces the administrative and financial burden of environmental permitting and waste management, facilitating smoother regulatory approvals and enhancing the corporate sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ketimine synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is vital for making informed decisions about process adoption and technology transfer.
Q: What are the primary advantages of using protic solvents over aprotic solvents for ketimine synthesis?
A: Using protic solvents like ethanol eliminates the need for hazardous aprotic solvents such as THF or dichloromethane and removes the requirement for expensive drying agents like molecular sieves or titanium tetrachloride, significantly simplifying the workup and reducing environmental impact.
Q: How does this method improve the purity profile of the final API intermediate?
A: The process allows for direct crystallization of the product from the reaction mixture, and subsequent recrystallization steps effectively remove residual starting materials (ketones) and catalyst traces, ensuring stringent purity specifications suitable for pharmaceutical applications.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the method operates at mild temperatures (20-100°C) and atmospheric or slightly elevated pressure, utilizing common industrial solvents and catalysts, making it highly adaptable for continuous processing and large-scale production without specialized cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sertraline Imine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the broader context of API manufacturing. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By integrating advanced methodologies like the protic solvent ketimine synthesis into our portfolio, we offer our partners a competitive edge through superior quality and consistent supply.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your existing supply chain or require specific COA data and route feasibility assessments for new developments, we are ready to provide the support you need. Contact us today to explore a partnership that combines technical excellence with commercial reliability, ensuring your drug development pipeline remains robust and cost-effective.
