Scalable Aminoborane Production Using Novel Disilylamine Rare Earth Catalysis
The landscape of organoboron chemistry is undergoing a significant transformation driven by the need for more efficient and sustainable synthetic methodologies, particularly for the production of aminoborane compounds which serve as critical building blocks in modern medicinal chemistry and materials science. Patent CN110818733B introduces a groundbreaking advancement in this field by disclosing a novel method for preparing boric acid esters through the hydroboration reaction of imines and boranes, catalyzed by a specialized disilylamine rare earth complex. This technology represents a paradigm shift from traditional catalytic systems, offering a robust solution that combines high catalytic activity with exceptional operational simplicity. By leveraging the unique electronic properties of rare earth metals such as ytterbium, europium, and samarium coordinated with bulky silylamide ligands, this invention achieves reaction efficiencies that were previously difficult to attain without harsh conditions. For R&D directors and process chemists seeking to optimize their synthetic routes for high-purity pharmaceutical intermediates, this patent provides a compelling alternative that promises to streamline production workflows while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aminoboranes via the hydroboration of imines has been plagued by significant inefficiencies that hinder large-scale industrial adoption. Conventional catalytic systems often rely on transition metals such as rhenium, calcium, zinc, magnesium, or nickel, as well as small molecule catalysts like sodium hydroxide or homoleptic boron complexes. While these methods can achieve the desired transformation, they frequently suffer from severe drawbacks including the requirement for elevated reaction temperatures and prolonged reaction times that can extend beyond three days. Such harsh conditions not only increase energy consumption and operational costs but also pose risks regarding the thermal stability of sensitive substrates and the formation of unwanted polysubstituted byproducts. Furthermore, many existing protocols demand high catalyst loadings to drive the reaction to completion, leading to increased metal contamination in the final product and necessitating costly and time-consuming purification steps to meet the stringent purity specifications required for reliable agrochemical intermediate supplier networks and pharmaceutical applications.
The Novel Approach
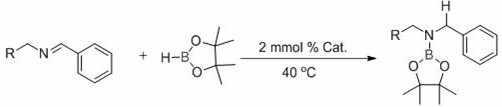
In stark contrast to these legacy methods, the technology disclosed in CN110818733B utilizes a disilylamine rare earth complex to catalyze the hydroboration reaction with unprecedented efficiency and selectivity. This novel approach enables the reaction to proceed under remarkably mild conditions, typically at temperatures around 40°C, completing the transformation in merely 3 hours with yields consistently exceeding 80%. The core innovation lies in the specific coordination environment of the rare earth center, which activates the boron-hydrogen bond effectively without the need for thermal forcing. This drastic reduction in reaction time and severity translates directly into enhanced process safety and reduced energy overhead. Moreover, the system exhibits excellent atom economy, minimizing waste generation and simplifying the downstream isolation of the target aminoborane ester. The general reaction scheme illustrates the seamless conversion of imine substrates into valuable aminoborane products using pinacolborane as the boron source.
Mechanistic Insights into Disilylamine Rare Earth Complex Catalysis
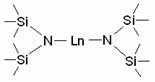
The exceptional performance of this catalytic system can be attributed to the unique structural and electronic characteristics of the disilylamine rare earth complex, specifically formulated as Ln{N(SiMe3)2}3 where Ln represents a lanthanide metal such as Ytterbium (Yb), Europium (Eu), or Samarium (Sm). The steric bulk provided by the trimethylsilyl groups on the nitrogen ligands creates a protected coordination sphere around the metal center, preventing catalyst deactivation through aggregation while still allowing substrate access. This structural integrity ensures that the catalyst remains active throughout the reaction cycle, facilitating the activation of the pinacolborane B-H bond and its subsequent addition across the carbon-nitrogen double bond of the imine. The Lewis acidic nature of the rare earth ion plays a pivotal role in polarizing the imine substrate, thereby lowering the activation energy barrier for the hydroboration step. Understanding this mechanism is crucial for cost reduction in fine chemical intermediates manufacturing, as it allows for the precise tuning of reaction parameters to maximize turnover numbers and minimize catalyst loading to as low as 2 mol%.
Beyond mere catalytic activity, the mechanism inherently supports superior impurity control, a critical factor for R&D teams focused on regulatory compliance. The high selectivity of the rare earth complex minimizes the formation of polysubstituted side products that are common in less controlled hydroboration reactions. By operating under mild thermal conditions, the risk of thermal decomposition of the imine substrate or the aminoborane product is significantly mitigated, preserving the chemical integrity of the molecule. The compatibility of this catalyst with a wide range of electronic environments on the imine substrate—accommodating electron-donating groups like methoxy and electron-withdrawing groups like fluoro—demonstrates its robustness. This versatility ensures that the process can be applied to the commercial scale-up of complex polymer additives and pharmaceutical precursors without requiring extensive re-optimization for each new derivative, thereby accelerating the development timeline for new drug candidates.
How to Synthesize Aminoborane Efficiently
The practical implementation of this patented technology involves a straightforward protocol that is highly amenable to standard laboratory and plant equipment. The process begins with the preparation of an anhydrous and oxygen-free environment, typically achieved by purging the reaction vessel with nitrogen gas, which is essential to protect the sensitive rare earth catalyst from moisture and oxidation. Following this, the imine substrate, the disilylamine rare earth complex catalyst, and pinacolborane are mixed sequentially in the reactor. The reaction is then allowed to proceed at a controlled temperature, preferably around 40°C, for a duration of approximately 3 hours. Upon completion, the reaction mixture can be quenched and analyzed, with the product yield determined via nuclear magnetic resonance spectroscopy. For detailed operational parameters and safety guidelines, the standardized synthesis steps are outlined below.
- Under an inert nitrogen atmosphere, sequentially mix the imine substrate, the disilylamine rare earth complex catalyst, and pinacolborane in an anhydrous reaction vessel.
- Maintain the reaction mixture at a mild temperature range of 30-80°C, preferably around 40°C, ensuring uniform mixing throughout the process.
- Allow the hydroboration reaction to proceed for approximately 3 hours, followed by standard workup procedures to isolate the high-purity aminoborane product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this disilylamine rare earth catalyzed hydroboration process offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the manufacturing process, which eliminates the need for prolonged heating cycles and expensive high-pressure equipment associated with older technologies. This reduction in process complexity directly correlates to lower capital expenditure requirements and reduced maintenance costs for production facilities. Furthermore, the ability to achieve high conversion rates in a short timeframe significantly enhances plant throughput, allowing manufacturers to respond more agilely to market demands and reduce inventory holding times. The mild reaction conditions also imply a safer working environment, potentially lowering insurance premiums and regulatory compliance burdens related to hazardous operations.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the exceptionally low catalyst loading requirement of only 2 mol%. In traditional processes, high catalyst loads often necessitate expensive metal recovery units or result in significant loss of precious metals in the waste stream. By minimizing the amount of rare earth complex needed, the raw material cost per kilogram of product is substantially decreased. Additionally, the elimination of harsh reaction conditions reduces energy consumption for heating and cooling, contributing to further operational savings. The high atom economy of the reaction ensures that the majority of the starting materials are incorporated into the final product, minimizing waste disposal costs and maximizing the value derived from every batch of reagents purchased.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on complex, multi-step syntheses that are prone to bottlenecks. This streamlined one-pot hydroboration method reduces the number of unit operations required, thereby decreasing the probability of equipment failure or process deviation. The robustness of the catalyst across a wide range of substrates means that a single catalytic system can be used to produce a diverse portfolio of aminoborane derivatives, simplifying inventory management and reducing the need for multiple specialized catalyst stocks. This flexibility allows suppliers to maintain consistent lead times even when switching between different product grades, ensuring a steady flow of high-purity OLED material precursors or pharmaceutical intermediates to downstream customers without interruption.
- Scalability and Environmental Compliance: As the industry moves towards greener manufacturing practices, the environmental profile of this process offers a distinct competitive advantage. The mild conditions and high selectivity result in a cleaner crude product, which simplifies purification and reduces the volume of solvent and auxiliary chemicals required for workup. This reduction in chemical usage aligns with global sustainability goals and helps manufacturers meet increasingly strict environmental regulations regarding waste discharge. The process is inherently scalable, having been demonstrated to work efficiently from gram-scale laboratory experiments to potential multi-ton production runs, ensuring that supply can be ramped up rapidly to meet surging demand without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled a set of frequently asked questions based on the specific technical disclosures within the patent documentation. These answers address common concerns regarding catalyst stability, substrate compatibility, and process safety, providing a clear picture of what can be expected during technology transfer. Understanding these nuances is essential for making informed decisions about process adoption and for anticipating any necessary adjustments to current manufacturing infrastructure.
Q: What are the primary advantages of the disilylamine rare earth complex catalyst over traditional systems?
A: The disilylamine rare earth complex offers superior catalytic activity under significantly milder conditions (40°C) compared to traditional systems that often require high temperatures or extended reaction times up to three days. Furthermore, it achieves high yields exceeding 80% with a remarkably low catalyst loading of only 2 mol%, enhancing atom economy and reducing metal waste.
Q: Which substrates are compatible with this hydroboration methodology?
A: This catalytic system demonstrates broad substrate scope, effectively processing various imines including benzylidene aniline derivatives with substituents such as methyl, phenyl, p-fluorophenyl, and p-methoxyphenyl. This versatility allows for the synthesis of diverse aminoborane structures required for complex pharmaceutical intermediates.
Q: How does this process impact industrial scalability and safety?
A: The process operates under mild thermal conditions and utilizes a simple mixing protocol without the need for extreme pressures or hazardous reagents, significantly improving operational safety. The short reaction time of 3 hours and easy post-treatment facilitate rapid throughput, making it highly suitable for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminoborane Supplier
The technological breakthroughs detailed in CN110818733B underscore the immense potential of rare earth catalysis in modernizing the production of critical chemical intermediates. At NINGBO INNO PHARMCHEM, we recognize the value of such innovations and have positioned ourselves as a leader in translating advanced academic and patent research into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aminoborane or related intermediate delivered meets the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite forward-thinking organizations to collaborate with us to leverage this cutting-edge hydroboration technology for their specific product lines. By partnering with NINGBO INNO PHARMCHEM, you gain access not just to a supplier, but to a technical ally dedicated to optimizing your supply chain. We encourage you to contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. Let us provide you with specific COA data and comprehensive route feasibility assessments to demonstrate how we can drive efficiency and value for your organization.
