Advanced Synthesis of Hexaphenoxycyclotriphosphazene for High-Performance Flame Retardants
The development of high-performance flame retardant materials is critical for the safety and reliability of modern engineering plastics, particularly in the electronics and automotive sectors. Patent CN103539820A introduces a groundbreaking preparation method for hexaphenoxycyclotriphosphazene, a key phosphazene derivative known for its exceptional thermal stability and flame-retardant efficiency. This technology addresses the longstanding challenges of low conversion rates and difficult industrialization associated with traditional synthesis routes. By utilizing phosphorus pentachloride and ammonium chloride as starting materials in a chlorobenzene solvent system, the process achieves a high-selectivity synthetic reaction that yields a highly purified intermediate solution. The subsequent reaction with potassium phenate ensures the final product meets stringent purity specifications of greater than 99% with chlorine content below 20ppm. This innovation represents a significant leap forward for any reliable plastic additives supplier aiming to deliver superior fire safety solutions for polycarbonate and PC/ABS alloys.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of hexaphenoxycyclotriphosphazene has been plagued by complex processing steps that hinder efficiency and product quality. Traditional techniques typically involve synthesizing the hexachlorocyclotriphosphazene intermediate, followed by a rigorous isolation process that includes suction filtration, vacuum distillation of chlorobenzene, and crystallization using petroleum ether or normal heptane. These energy-intensive separation steps not only increase operational costs but also expose the thermally sensitive intermediate to conditions that promote unwanted cross-linking reactions. Consequently, the resulting crude product often exhibits poor color, appearing grey or faint yellow, which fails to meet the aesthetic and performance requirements of high-end applications. Furthermore, conventional purification often necessitates washing with sulfuric acid, generating substantial amounts of hazardous acidic wastewater that complicates environmental compliance and waste management protocols.
The Novel Approach
The patented methodology revolutionizes this landscape by implementing a telescoped synthesis strategy that eliminates the need for isolating the hexachlorocyclotriphosphazene intermediate. Instead of distilling off the solvent and crystallizing the intermediate, the process directly utilizes the filtered chlorobenzene solution of hexachlorocyclotriphosphazene for the subsequent substitution reaction. This seamless integration significantly simplifies the technical process, reduces raw material consumption, and minimizes energy usage by avoiding repeated heating and cooling cycles. Crucially, by bypassing the concentration and heating of the intermediate solution, the method effectively prevents the cross-linking reactions that typically degrade yield and product color. The implementation of a composite catalyst system alongside a nitrogen protection atmosphere further enhances reaction selectivity and safety, ensuring a robust and reproducible manufacturing pathway.
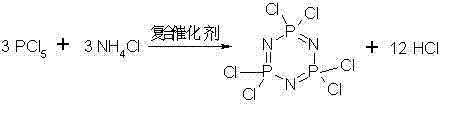
Mechanistic Insights into Composite Catalyzed Cyclization and Substitution
The core of this technological advancement lies in the sophisticated use of a composite catalyst system comprising zinc chloride, iron(III) chloride, and magnesium chloride during the initial cyclization step. This specific combination of Lewis acids creates a synergistic catalytic environment that facilitates the condensation of phosphorus pentachloride and ammonium chloride into the cyclic trimer structure with high fidelity. The presence of nitrogen protection throughout the reaction serves a dual purpose: it prevents the hydrolysis of the moisture-sensitive phosphorus pentachloride and aids in the rapid removal of hydrogen chloride gas generated during the reaction. This continuous removal of byproduct gas drives the equilibrium forward, promoting higher conversion rates and minimizing the formation of linear polymeric byproducts that often contaminate the final product. The precise control of temperature, ramping from 120°C to 130°C, ensures optimal kinetic conditions for ring closure while maintaining the stability of the reactive species involved in the complex inorganic-organic transformation.
Following the formation of the hexachlorocyclotriphosphazene solution, the mechanism shifts to a nucleophilic substitution where phenoxy groups replace the active chlorine atoms on the phosphorus-nitrogen ring. The use of potassium phenolate, prepared in situ from phenol and potassium hydroxide, provides a strong nucleophile that efficiently attacks the electrophilic phosphorus centers. A critical aspect of impurity control in this stage is the avoidance of acidic workups; instead, the process employs a联合 decolorization method using activated carbon and attapulgite clay. This physical adsorption technique effectively removes trace organic impurities and colored bodies without introducing ionic contaminants or requiring aqueous washes that could lead to emulsion formation. The final crystallization from ethanol ensures that any remaining soluble impurities are left in the mother liquor, resulting in a product with exceptional whiteness and a chlorine content reduced to trace levels of less than 20ppm, which is vital for preventing corrosion in downstream polymer processing.
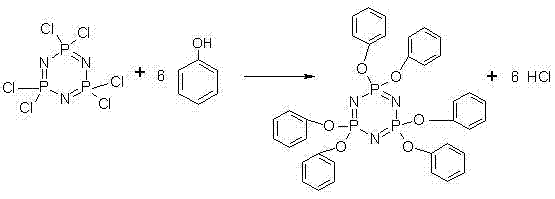
How to Synthesize Hexaphenoxycyclotriphosphazene Efficiently
The synthesis protocol outlined in the patent offers a clear roadmap for manufacturers seeking to optimize their production lines for flame retardant intermediates. The process begins with the careful preparation of raw materials, ensuring strict mass ratios between ammonium chloride, the composite catalyst, pyridine, and phosphorus pentachloride to maintain stoichiometric balance. Reaction conditions are tightly controlled, with an oil bath heating system used to manage the exothermic nature of the cyclization and maintain the reflux temperature within the narrow window of 120°C to 130°C. Following the completion of the first step, the reaction mixture is cooled and filtered to remove insoluble salts, yielding a clear solution ready for the second stage without further purification. The detailed standardized synthesis steps provided below guide operators through the precise addition rates, temperature controls, and decolorization procedures necessary to achieve the reported 76.5% total yield and high purity specifications consistently.
- React phosphorus pentachloride and ammonium chloride in chlorobenzene with a ZnCl2/FeCl3/MgCl2 composite catalyst at 120-130°C to form hexachlorocyclotriphosphazene solution.
- Directly add the crude hexachlorocyclotriphosphazene solution to a prepared potassium phenolate mixture without distillation or crystallization of the intermediate.
- Perform decolorization using activated carbon and attapulgite, followed by solvent recovery and ethanol recrystallization to obtain >99% purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers profound strategic benefits that extend beyond simple chemical yield improvements. The elimination of vacuum distillation and intermediate crystallization steps translates directly into a drastically simplified operational workflow, reducing the equipment footprint and maintenance requirements associated with complex separation units. By removing the need for sulfuric acid washing, the process significantly lowers the costs related to hazardous waste treatment and environmental compliance, thereby reducing the total cost of ownership for the manufacturing facility. The improved reproducibility and color consistency of the product mean fewer batch rejections and less variability in the final polymer formulations, ensuring a more stable supply of high-quality materials for downstream customers. This reliability is essential for maintaining long-term contracts with major polymer producers who demand consistent material performance.
- Cost Reduction in Manufacturing: The streamlined process architecture eliminates several energy-intensive unit operations, such as solvent recovery via vacuum distillation and the heating required for intermediate crystallization. By avoiding the thermal stress on the hexachlorocyclotriphosphazene intermediate, the process prevents yield losses associated with cross-linking and polymerization side reactions, thereby maximizing the output from every kilogram of raw material input. Furthermore, the substitution of expensive and hazardous acid washing steps with solid-phase decolorization agents reduces the consumption of auxiliary chemicals and lowers the burden on wastewater treatment infrastructure. These cumulative efficiencies result in substantial cost savings in polymer additives manufacturing, allowing for more competitive pricing structures without compromising on margin.
- Enhanced Supply Chain Reliability: The robustness of the composite catalyst system and the tolerance of the reaction conditions contribute to a highly stable production process that is less prone to deviations and shutdowns. The ability to telescope the synthesis steps reduces the overall cycle time per batch, enabling manufacturers to respond more quickly to fluctuations in market demand and reducing lead time for high-purity plastic additives. Additionally, the use of common and readily available solvents like chlorobenzene and ethanol minimizes the risk of supply disruptions associated with specialty reagents. This operational resilience ensures a continuous flow of materials, which is critical for just-in-time manufacturing environments in the automotive and electronics industries.
- Scalability and Environmental Compliance: The simplicity of the reaction setup, which avoids complex pressure vessels or extreme cryogenic conditions, makes the commercial scale-up of complex flame retardants straightforward and predictable. The reduction in hazardous waste generation, particularly the absence of acidic effluents, aligns perfectly with increasingly stringent global environmental regulations and corporate sustainability goals. This green chemistry approach not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible partner in the value chain. The process is designed to be easily adaptable from pilot scale to multi-ton production, ensuring that capacity can be expanded seamlessly as market adoption of phosphazene-based flame retardants grows.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of hexaphenoxycyclotriphosphazene based on the patented technology. These insights are derived directly from the experimental data and process descriptions found in the intellectual property documentation, providing clarity on yield optimization, purity control, and environmental impact. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this material into their existing supply chains or product formulations. The answers reflect the specific advantages of the composite catalyst system and the telescoped synthesis approach over legacy methods.
Q: How does the new process improve product color compared to traditional methods?
A: Traditional methods often result in grey or faint yellow crystals due to cross-linking reactions during the heating and concentration of the intermediate. The novel process avoids vacuum distillation of the hexachlorocyclotriphosphazene intermediate, preventing thermal degradation. Furthermore, the use of a combined activated carbon and attapulgite decolorization system effectively adsorbs colored impurities, ensuring a white crystal product with high optical purity.
Q: What are the environmental benefits of avoiding sulfuric acid washing?
A: Conventional synthesis frequently relies on sulfuric acid scrubbing to purify the organic phase, which generates significant volumes of acidic wastewater requiring neutralization and treatment. The patented method eliminates this step entirely by utilizing a solid-phase decolorization and filtration strategy. This drastically reduces the chemical oxygen demand (COD) of the effluent and lowers the operational burden associated with hazardous waste disposal and environmental compliance.
Q: Why is the composite catalyst system superior for cyclization?
A: The use of a composite catalyst comprising zinc chloride, iron(III) chloride, and magnesium chloride creates a synergistic effect that enhances the selectivity of the cyclization reaction. This specific combination promotes the formation of the trimeric hexachlorocyclotriphosphazene structure while suppressing the formation of linear polymers or higher-order cyclic byproducts. This results in a cleaner reaction profile, simplifying downstream purification and improving the overall mass balance of the synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexaphenoxycyclotriphosphazene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced flame retardants play in ensuring the safety and performance of next-generation engineering plastics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify parameters such as chlorine content and thermal stability before shipment. Our capability to handle complex organophosphorus chemistry allows us to offer a level of technical support and customization that goes beyond simple transactional supply, making us a true partner in your material development journey.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages inherent in this process compared to your current sourcing strategies. We encourage potential partners to reach out for specific COA data and route feasibility assessments to validate the compatibility of our hexaphenoxycyclotriphosphazene with your polymer matrices. Let us collaborate to drive innovation and safety in the global plastics industry through superior chemical solutions.
