Scalable Synthesis of 4-Oxa-7-Azaspiro[2.5]Octane for Advanced Kinase Inhibitor Manufacturing
The pharmaceutical industry is constantly seeking robust synthetic routes for complex spirocyclic scaffolds, particularly those serving as key intermediates for kinase inhibitors. Patent CN108530375B, published in April 2020, addresses a critical gap in the supply chain for 4-oxa-7-azaspiro[2.5]octane and its salts. This specific chemical architecture has gained immense traction as a pivotal building block for developing therapeutics targeting LRRK2 for Parkinson's disease, GPR43 for metabolic disorders like diabetes and obesity, and IRAK4 for inflammatory conditions. While the biological utility of these compounds is well-documented in international filings such as WO2014137728A1 and WO2016053772A1, the prior art notably lacked an efficient, scalable preparation method for the core spirocyclic amine itself. This patent introduces a ingenious four-step sequence that transforms simple, commercially available starting materials into the high-value target molecule with remarkable efficiency.
By establishing a clear pathway from methyl 1-hydroxy-1-cyclopropanecarboxylate, this technology provides a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates. The methodology described eliminates the need for obscure reagents or hazardous conditions that typically plague early-stage process chemistry. For R&D directors and procurement specialists alike, understanding this route is essential for securing the supply of next-generation neurodegenerative and metabolic disease treatments. The following analysis dissects the technical merits and commercial implications of this novel synthesis strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of spirocyclic amine frameworks like 4-oxa-7-azaspiro[2.5]octane has presented significant challenges in process chemistry. Conventional approaches often rely on multi-step sequences involving expensive chiral auxiliaries or harsh cyclization conditions that compromise the integrity of the strained cyclopropane ring. Many existing methods suffer from poor atom economy and generate substantial quantities of difficult-to-remove impurities, which drastically increases the cost of goods sold (COGS) during purification. Furthermore, traditional routes frequently utilize unstable intermediates that require cryogenic storage or immediate consumption, creating logistical bottlenecks in a manufacturing environment. The lack of a dedicated, optimized synthesis for this specific scaffold meant that drug developers were forced to adapt generic spirocycle methodologies, resulting in suboptimal yields and inconsistent batch quality. These inefficiencies translate directly into higher risks for supply chain continuity and delayed timelines for clinical trial material production.
The Novel Approach
The patented methodology offers a transformative solution by utilizing a linear, four-step progression that maximizes yield while minimizing operational complexity. Starting with the inexpensive methyl 1-hydroxy-1-cyclopropanecarboxylate, the process employs a straightforward nucleophilic substitution followed by a controlled catalytic hydrogenation. This is succeeded by a base-mediated intramolecular cyclization and a final reduction step, collectively achieving a total yield exceeding 50 percent. The brilliance of this design lies in its modularity; each intermediate is stable and isolable, allowing for rigorous quality control checkpoints before proceeding to the next stage. By avoiding extreme temperatures and pressures, the route significantly lowers the barrier to entry for manufacturing partners. This approach not only fills the void in the prior art but also sets a new benchmark for the cost reduction in pharmaceutical intermediate manufacturing by streamlining the entire production workflow.
![Complete four-step synthesis route of 4-oxa-7-azaspiro[2.5]octane from methyl 1-hydroxy-1-cyclopropanecarboxylate](/insights/img/4-oxa-7-azaspiro-octane-synthesis-pharma-supplier-20260307012353-02.webp)
Mechanistic Insights into the Four-Step Cascade Synthesis
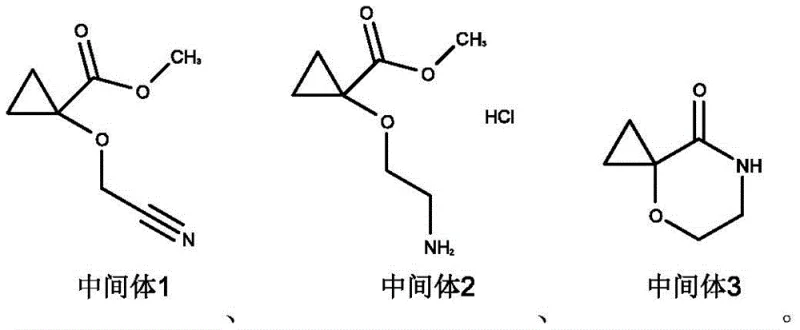
The chemical elegance of this process is rooted in the careful selection of reagents that preserve the sensitive spirocyclic geometry throughout the transformation. The initial step involves the deprotonation of the hydroxyl group using sodium hydride in tetrahydrofuran at mild temperatures ranging from -10°C to 10°C, followed by alkylation with bromoacetonitrile. This generates Intermediate 1, a nitrile ether, with high fidelity and minimal side reactions. Subsequently, the nitrile group is selectively reduced to a primary amine via catalytic hydrogenation using palladium on carbon or Raney nickel in the presence of hydrochloric acid. This acidic environment is crucial as it forms the stable amine salt, Intermediate 2, preventing unwanted polymerization or degradation of the reactive amine species. The subsequent cyclization relies on the nucleophilic attack of the newly formed amine onto the proximal ester carbonyl, facilitated by organic bases like triethylamine, to close the six-membered lactam ring efficiently.
The final transformation involves the reduction of the lactam carbonyl to a methylene group, converting the cyclic amide into the target cyclic amine. This is achieved using potent reducing agents such as lithium aluminum hydride or borane-tetrahydrofuran complexes, which are capable of cleaving the robust amide bond without opening the cyclopropane ring. The preservation of the spiro-center is paramount, as any ring-opening would render the material useless for its intended biological activity. The patent specifies precise molar ratios and solvent systems to ensure that the reduction proceeds cleanly to the amine without over-reduction or decomposition. This mechanistic precision ensures that the final product, 4-oxa-7-azaspiro[2.5]octane hydrochloride, is obtained with high purity, meeting the stringent specifications required for active pharmaceutical ingredient (API) synthesis.
How to Synthesize 4-Oxa-7-Azaspiro[2.5]Octane Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process begins with the handling of sodium hydride, necessitating anhydrous conditions and inert atmosphere protection to prevent exothermic incidents. Operators must carefully control the addition rate of reagents during the substitution and reduction phases to manage heat evolution effectively. The hydrogenation step, while operating at relatively low pressures of 1 to 10 atmospheres, still requires standard safety protocols for handling hydrogen gas and pyrophoric catalysts like Raney nickel. Detailed standard operating procedures (SOPs) should be established for the workup of each step, particularly the extraction and chromatography processes described in the examples, to maximize recovery. For a comprehensive breakdown of the specific reaction conditions, stoichiometry, and isolation techniques, please refer to the standardized guide below.
- Perform nucleophilic substitution of methyl 1-hydroxy-1-cyclopropanecarboxylate with bromoacetonitrile using sodium hydride in THF to form Intermediate 1.
- Conduct catalytic hydrogenation of the nitrile group in Intermediate 1 using Pd/C or Raney Nickel under acidic conditions to yield the amine salt Intermediate 2.
- Execute base-mediated intramolecular cyclization of Intermediate 2 to form the lactam ring structure of Intermediate 3.
- Reduce the lactam carbonyl of Intermediate 3 using lithium aluminum hydride or borane complexes to obtain the final spirocyclic amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented route offers compelling advantages that directly address the pain points of modern pharmaceutical supply chains. The reliance on commodity chemicals rather than bespoke, high-cost building blocks fundamentally alters the cost structure of the final intermediate. By utilizing a pathway that avoids transition metals in the final steps and uses standard solvents like methanol and ethyl acetate, the process simplifies waste management and solvent recovery operations. This simplicity translates into a more resilient supply chain, as the risk of disruption due to the scarcity of a single exotic reagent is virtually eliminated. Furthermore, the high overall yield means that less raw material is required to produce the same amount of product, driving down the effective cost per kilogram significantly.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and the use of widely available starting materials like methyl 1-hydroxy-1-cyclopropanecarboxylate lead to substantial cost savings. The process avoids complex purification techniques such as preparative HPLC, relying instead on standard column chromatography and crystallization, which are far more economical at scale. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, further lowering the operational expenditure. The high yield of each individual step minimizes material loss, ensuring that the theoretical cost savings are realized in actual production runs.
- Enhanced Supply Chain Reliability: Because the synthesis depends on bulk chemicals that are produced by multiple global suppliers, the risk of supply interruption is drastically reduced. The stability of the intermediates allows for the potential to stockpile key precursors, providing a buffer against market volatility. This reliability is critical for maintaining continuous production schedules for downstream API manufacturing, ensuring that clinical and commercial demands are met without delay. The robustness of the chemistry also means that technology transfer between different manufacturing sites can be executed with minimal friction.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily replicated in large-scale stainless steel reactors. The absence of highly toxic reagents or extreme pressures simplifies the environmental health and safety (EHS) profile of the manufacturing site. Waste streams are primarily composed of standard organic solvents and inorganic salts, which can be treated using conventional wastewater treatment protocols. This alignment with green chemistry principles facilitates regulatory approval and supports the sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation. Understanding these nuances is vital for process chemists and procurement officers evaluating this technology for adoption. The clarity provided here aims to mitigate risks associated with technology transfer and scale-up.
Q: What is the total yield of the patented 4-step synthesis route?
A: The patented process achieves a total yield of more than 50% over the four sequential steps, which is considered highly efficient for constructing complex spirocyclic scaffolds in pharmaceutical manufacturing.
Q: Are the starting materials for this synthesis readily available?
A: Yes, the synthesis begins with methyl 1-hydroxy-1-cyclopropanecarboxylate and bromoacetonitrile, both of which are commercially available commodity chemicals, ensuring a robust and reliable supply chain.
Q: Does this process require specialized high-pressure equipment?
A: No, the hydrogenation step operates at mild pressures ranging from 1 to 10 atmospheres, and other steps occur at ambient or slightly elevated temperatures, making it suitable for standard industrial reactors without exotic equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Oxa-7-Azaspiro[2.5]Octane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team of expert process chemists has extensively evaluated the route described in CN108530375B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities capable of handling the specific hydrogenation and reduction steps required for this synthesis with the utmost safety and precision. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which ensure that every batch of 4-oxa-7-azaspiro[2.5]octane meets the exacting standards required for GMP manufacturing.
We invite you to collaborate with us to optimize your supply chain for LRRK2 and GPR43 inhibitor programs. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to accelerate your drug development journey with reliable, cost-effective, and high-purity chemical solutions.
