Advanced Two-Step Synthesis of Substituted Aryl Fluorene Derivatives for Commercial Scale Production
Advanced Two-Step Synthesis of Substituted Aryl Fluorene Derivatives for Commercial Scale Production
The rapid evolution of the organic electronics and pharmaceutical sectors has intensified the demand for high-performance functional materials, specifically substituted aryl fluorene derivatives which serve as critical building blocks for OLED emitters and bioactive agents. Patent CN114804994A, published in July 2022, introduces a robust and scalable methodology for preparing these complex scaffolds with exceptional efficiency. This technical disclosure outlines a streamlined two-step synthetic route that begins with the lithiation of 2-bromobiphenyl and concludes with an acid-catalyzed cyclization, effectively addressing the longstanding challenges of yield optimization and impurity control in fluorene chemistry. By leveraging low-temperature organometallic coupling followed by a mild dehydration step, this innovation provides a reliable substituted aryl fluorene supplier pathway that aligns with modern green chemistry principles while maintaining rigorous quality standards required for electronic grade materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9,9-disubstituted fluorenes and their aryl analogues has been plagued by inefficient multi-step sequences that often require harsh reaction conditions and expensive transition metal catalysts. Traditional Friedel-Crafts alkylations or cyclizations frequently suffer from poor regioselectivity, leading to difficult-to-separate isomeric mixtures that compromise the optical and electronic properties of the final material. Furthermore, conventional routes often necessitate the use of stoichiometric amounts of corrosive Lewis acids like aluminum chloride, which generate substantial quantities of hazardous waste and complicate the workup procedure. The accumulation of heavy metal residues from palladium-catalyzed cross-couplings, often employed in alternative strategies, poses significant risks for downstream applications in sensitive optoelectronic devices where trace impurities can act as quenching sites. These legacy methods result in prolonged production cycles, elevated operational costs, and inconsistent batch-to-batch reproducibility, creating a bottleneck for the commercial scale-up of complex electronic chemicals.
The Novel Approach
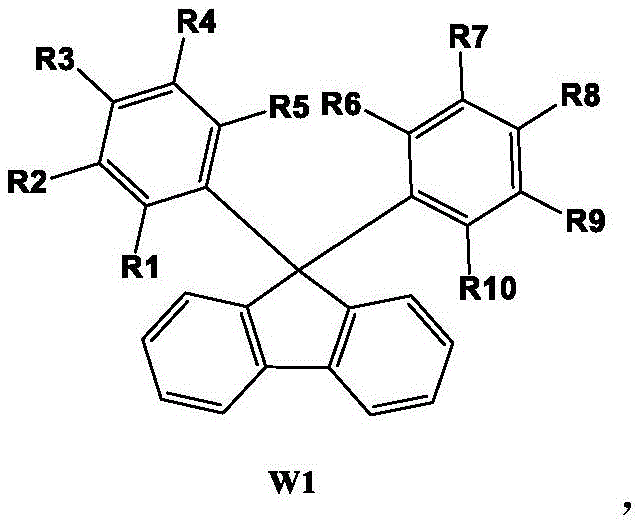
In stark contrast to these archaic techniques, the methodology disclosed in CN114804994A utilizes a direct nucleophilic addition strategy that bypasses the need for transition metal catalysis entirely. The process initiates with the generation of a highly reactive biphenyl lithium species at cryogenic temperatures, which attacks a broad range of substituted aryl ketones to form a stable tertiary alcohol intermediate. This intermediate is subsequently subjected to an intramolecular cyclization under acidic conditions that are remarkably mild compared to traditional superacid media. The elegance of this approach lies in its simplicity; the reaction proceeds with high atom economy and generates minimal byproducts, allowing for isolation of the target fluorene derivative through straightforward filtration and washing protocols. As illustrated in the diverse library of accessible structures, this platform technology enables the rapid exploration of structure-property relationships by accommodating various electron-donating and electron-withdrawing substituents without compromising the core reaction efficiency.
Mechanistic Insights into Sec-Butyl Lithium Mediated Cyclization
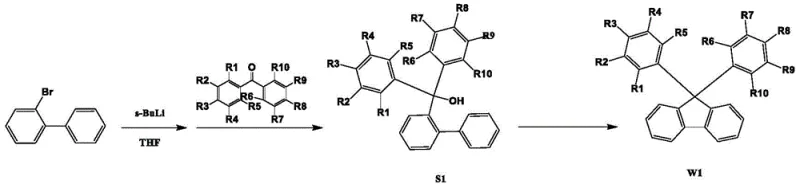
The cornerstone of this synthetic breakthrough is the precise control of organolithium reactivity during the initial carbon-carbon bond formation. At -70°C in anhydrous tetrahydrofuran, sec-butyl lithium effects a rapid halogen-lithium exchange with 2-bromobiphenyl, generating a transient aryl lithium species that is immediately trapped by the electrophilic carbonyl carbon of the substituted aryl ketone. This low-temperature regime is critical for suppressing competing side reactions such as benzyne formation or nucleophilic attack on the solvent, ensuring that the nucleophilic addition proceeds with near-quantitative conversion to the tertiary alcohol intermediate S1. The subsequent cyclization step relies on the activation of the hydroxyl group by strong protic acids, facilitating the loss of water and the formation of a resonance-stabilized carbocation that undergoes intramolecular electrophilic aromatic substitution. This mechanism effectively closes the five-membered ring of the fluorene core, driven by the restoration of aromaticity and the thermodynamic stability of the conjugated system.
From an impurity control perspective, the choice of quenching agent and washing solvent plays a pivotal role in defining the final purity profile. The patent specifies quenching the lithiation mixture with dilute hydrochloric acid, which not only neutralizes excess organolithium reagent but also helps to solubilize lithium salts, allowing them to be removed during the filtration stage. The use of petroleum ether for boiling and washing the solid intermediate is a strategic decision that exploits the differential solubility of the polar alcohol intermediate versus non-polar organic impurities. This physical purification step, combined with the high selectivity of the acid-catalyzed cyclization, ensures that the final product achieves HPLC purity levels exceeding 99% without the need for column chromatography. Such rigorous control over the reaction trajectory minimizes the presence of homologous impurities and unreacted starting materials, which is paramount for applications in high-performance organic light-emitting diodes where spectral purity is non-negotiable.
How to Synthesize Substituted Aryl Fluorene Efficiently
The implementation of this synthesis route in a pilot or production plant requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure safety and reproducibility. The process is divided into two distinct operational stages: the cryogenic lithiation and addition phase, followed by the thermal cyclization and isolation phase. Operators must maintain an inert atmosphere throughout to prevent moisture ingress, which could deactivate the lithium reagent, and carefully manage the exotherm during the quenching step. The following guide summarizes the critical operational windows derived from the experimental examples, providing a roadmap for translating this laboratory-scale innovation into a robust manufacturing protocol suitable for a reliable agrochemical intermediate supplier or electronic material producer.
- Perform lithiation of 2-bromobiphenyl at -70°C using sec-butyl lithium in THF, followed by nucleophilic addition to a substituted aryl ketone to form intermediate S1.
- Quench the reaction with dilute hydrochloric acid, filter, and wash with petroleum ether to isolate the high-purity intermediate.
- React intermediate S1 with glacial acetic acid and hydrochloric acid at 80°C for 4 hours to effect cyclization, then cool and filter to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield, directly impacting the bottom line through operational simplification and risk mitigation. The elimination of transition metal catalysts removes the necessity for expensive scavenging resins and complex metal removal validation studies, which are often cost-prohibitive in large-scale campaigns. Furthermore, the reliance on commodity chemicals such as tetrahydrofuran, hydrochloric acid, and acetic acid ensures a stable and resilient supply chain, insulating the manufacturing process from the volatility associated with specialized reagents. The simplified workup procedure, characterized by filtration and washing rather than energy-intensive distillation or chromatography, drastically reduces utility consumption and processing time, thereby enhancing the overall throughput of the production facility.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high atom efficiency and the avoidance of precious metal catalysts, which significantly lowers the raw material cost per kilogram of finished product. By achieving high conversion rates in the lithiation step and excellent yields in the cyclization step, the process minimizes the loss of valuable starting materials, effectively reducing the cost of goods sold. Additionally, the ability to isolate intermediates and final products via simple filtration eliminates the need for large volumes of organic solvents typically required for extraction and purification, leading to substantial savings in solvent procurement and waste disposal fees.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for flexible scheduling and reduced sensitivity to minor variations in raw material quality, ensuring consistent delivery timelines even in fluctuating market conditions. Since the key reagents like 2-bromobiphenyl and substituted benzophenones are widely available from multiple global vendors, the risk of single-source dependency is mitigated, securing the continuity of supply for critical downstream applications. The short reaction times and straightforward isolation steps further compress the manufacturing cycle time, enabling faster response to urgent customer demands and reducing the inventory holding costs associated with long lead times.
- Scalability and Environmental Compliance: The process design inherently supports scale-up from gram to tonnage quantities without requiring fundamental changes to the reaction engineering, as the heat transfer and mixing requirements are manageable with standard glass-lined or stainless steel reactors. The generation of less wastewater and the absence of heavy metal contaminants simplify the effluent treatment process, ensuring compliance with increasingly stringent environmental regulations and reducing the burden on waste management infrastructure. This environmentally friendly profile not only lowers regulatory risk but also enhances the corporate sustainability credentials of the manufacturing entity, aligning with the ESG goals of major multinational clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, drawing directly from the experimental data and process descriptions provided in the intellectual property documentation. These insights are intended to clarify the operational feasibility and quality attributes of the resulting fluorene derivatives for potential partners and technical evaluators.
Q: What is the purity level achievable with this synthesis method?
A: The patent data indicates that the described method consistently yields products with HPLC purity exceeding 99%, significantly reducing the need for extensive downstream purification processes.
Q: Which solvents are compatible with the lithiation step?
A: While tetrahydrofuran (THF) is the preferred solvent due to its solvation properties for organolithium species, the method also supports diethyl ether and 2-methyltetrahydrofuran as viable alternatives.
Q: How does this process address environmental concerns in fluorene production?
A: The process is designed to generate less wastewater compared to traditional methods, and the use of simple filtration and washing steps minimizes the generation of hazardous organic waste streams.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Aryl Fluorene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity fluorene derivatives play in the advancement of next-generation display technologies and pharmaceutical formulations. Our team of process chemists has extensively evaluated the route disclosed in CN114804994A and confirmed its potential for seamless translation from benchtop to commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of substituted aryl fluorene meets the exacting standards required for high-performance electronic applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this efficient synthetic strategy. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
