Advanced Enzymatic Synthesis of Factor Xa Inhibitor Intermediates for Commercial Scale
The pharmaceutical industry continuously seeks robust manufacturing pathways for potent anticoagulants, particularly Factor Xa inhibitors which play a critical role in preventing thrombotic disorders. Patent CN101611026A introduces a groundbreaking chemoenzymatic strategy for preparing pyrrolidine-3,4-dicarboxamide derivatives, specifically targeting the (3R,4R) stereoisomer essential for biological activity. This intellectual property addresses significant limitations found in prior art, such as WO2005/092881, by replacing hazardous reagents and low-yield steps with safer, more efficient enzymatic transformations. For R&D directors and procurement specialists, this patent represents a pivotal shift towards sustainable and scalable production of high-purity pharmaceutical intermediates. The disclosed methods not only improve overall yield but also mitigate severe safety risks associated with traditional chemical synthesis, ensuring a more reliable supply chain for critical cardiovascular medications. By leveraging specific lipases and optimized cycloaddition conditions, the process achieves superior stereocontrol without relying on costly chiral separation technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Previous synthetic routes for this class of compounds suffered from substantial drawbacks that hindered commercial viability and operational safety. The prior art often relied on the use of pyrophoric reagents like trimethylaluminum for Weinreb amidation, which poses significant fire hazards and requires specialized handling equipment in a manufacturing plant. Furthermore, the N-arylation steps in older methods frequently utilized potassium carbonate in dimethyl sulfoxide at elevated temperatures, a combination known to carry a high risk of thermal runaway events. 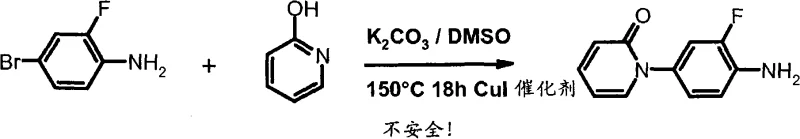 These safety concerns make scale-up dangerous and expensive, as extensive engineering controls are needed to manage exothermic potentials. Additionally, the overall yields were often mediocre due to non-selective reactions that generated complex impurity profiles, necessitating multiple purification steps that erode profit margins and extend production lead times. The reliance on stoichiometric chiral auxiliaries or preparative chromatography further inflated the cost of goods, making the final API economically challenging to produce at a multi-ton scale.
These safety concerns make scale-up dangerous and expensive, as extensive engineering controls are needed to manage exothermic potentials. Additionally, the overall yields were often mediocre due to non-selective reactions that generated complex impurity profiles, necessitating multiple purification steps that erode profit margins and extend production lead times. The reliance on stoichiometric chiral auxiliaries or preparative chromatography further inflated the cost of goods, making the final API economically challenging to produce at a multi-ton scale.
The Novel Approach
The methodology described in CN101611026A fundamentally reengineers the synthesis to prioritize safety and selectivity through biocatalysis and optimized chemical conditions. Instead of hazardous aluminum reagents, the new process employs enzymatic resolution using commercially available lipases, which operate under mild aqueous conditions and offer exceptional enantioselectivity. This shift eliminates the need for cryogenic temperatures and pyrophoric materials, drastically reducing the safety footprint of the manufacturing facility. The N-arylation step is reimagined using copper-catalyzed conditions in safer solvents like DMA or pyridine, avoiding the thermal instability associated with DMSO bases at high heat. By integrating a [3+2] cycloaddition that tolerates aqueous formaldehyde, the route simplifies the construction of the pyrrolidine core, avoiding the isolation of unstable ylide precursors. This holistic approach ensures that each step is amenable to large-scale processing, with crystallization serving as the primary purification method rather than chromatography, thereby enhancing throughput and consistency.
Mechanistic Insights into Enzymatic Resolution and Cycloaddition
The core innovation lies in the stereoselective construction of the pyrrolidine ring, achieved through a dipolar [3+2] cycloaddition followed by kinetic resolution. The process begins with the generation of an azomethine ylide from N-benzyl-trimethylsilylmethylamine and formaldehyde, which reacts with diethyl fumarate to form the racemic pyrrolidine diester. 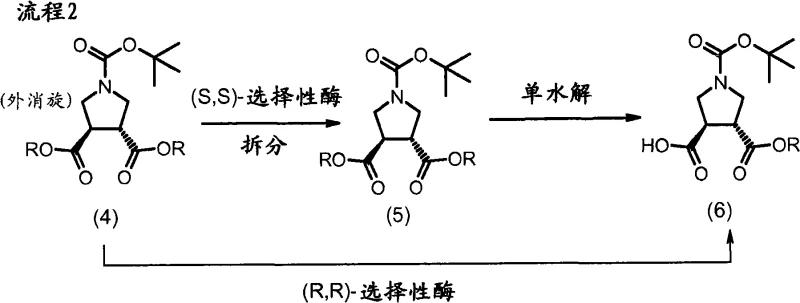 Crucially, the patent details how specific lipases, such as Lipolase from Thermomyces lanuginosus, can distinguish between enantiomers in an emulsified system. The enzyme selectively hydrolyzes the (S,S)-enantiomer, leaving the desired (R,R)-diester intact with high enantiomeric excess. This biocatalytic step is highly sensitive to pH and buffer composition, with phosphate buffers at neutral pH proving optimal for maintaining enzyme activity and selectivity. The mechanism relies on the precise fit of the substrate into the enzyme's active site, where steric hindrance prevents the hydrolysis of the unwanted isomer. Following resolution, a second enzymatic step using Lipase OF performs a selective monohydrolysis, converting one ester group to a carboxylic acid while leaving the other intact. This chemoenzymatic cascade avoids the use of harsh chemical hydrolysis agents that could racemize the sensitive chiral centers, ensuring the final intermediate meets stringent purity specifications required for pharmaceutical applications.
Crucially, the patent details how specific lipases, such as Lipolase from Thermomyces lanuginosus, can distinguish between enantiomers in an emulsified system. The enzyme selectively hydrolyzes the (S,S)-enantiomer, leaving the desired (R,R)-diester intact with high enantiomeric excess. This biocatalytic step is highly sensitive to pH and buffer composition, with phosphate buffers at neutral pH proving optimal for maintaining enzyme activity and selectivity. The mechanism relies on the precise fit of the substrate into the enzyme's active site, where steric hindrance prevents the hydrolysis of the unwanted isomer. Following resolution, a second enzymatic step using Lipase OF performs a selective monohydrolysis, converting one ester group to a carboxylic acid while leaving the other intact. This chemoenzymatic cascade avoids the use of harsh chemical hydrolysis agents that could racemize the sensitive chiral centers, ensuring the final intermediate meets stringent purity specifications required for pharmaceutical applications.
Impurity control is meticulously managed through the choice of reagents and reaction conditions that minimize side reactions. The use of aqueous formaldehyde in the cycloaddition step, previously thought to quench the ylide, is optimized to favor cycloaddition over hydrolysis, reducing the formation of oligomeric byproducts. In the amidation steps, the activation of the carboxylic acid using isobutyl chloroformate in the presence of lithium chloride accelerates the reaction rate at lower temperatures, preventing epimerization. The final alkylation with 2,2-difluoroethyl triflate is conducted under biphasic conditions to sequester acidic byproducts, protecting the acid-sensitive Boc group until the intended deprotection step. This level of mechanistic control ensures that the impurity profile remains simple and predictable, facilitating easier regulatory approval and consistent batch-to-batch quality. The ability to crystallize intermediates at multiple stages further purifies the material, removing trace enzymes and organic impurities without the need for silica gel chromatography.
How to Synthesize Pyrrolidine-3,4-dicarboxamide Efficiently
The synthesis of this complex intermediate requires a precise sequence of chemoenzymatic transformations to ensure high yield and optical purity. The process initiates with the cycloaddition to build the core ring system, followed by enzymatic resolution to establish the correct stereochemistry. Detailed standardized synthesis steps are provided below to guide process development teams in replicating these results.
- Perform [3+2] cycloaddition using N-benzyl-trimethylsilylmethylamine and diethyl fumarate to form the pyrrolidine core.
- Execute enzymatic resolution using Lipolase to separate enantiomers and achieve high optical purity.
- Conduct selective monohydrolysis and subsequent amide coupling to finalize the active pharmaceutical intermediate structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible benefits in terms of cost stability and operational reliability. The elimination of pyrophoric reagents and high-risk thermal conditions reduces the need for specialized containment infrastructure, lowering capital expenditure for manufacturing sites. By replacing expensive chiral chromatography with enzymatic resolution, the cost of raw materials and consumables is significantly reduced, directly impacting the bottom line. The robustness of the enzymatic steps ensures consistent yields, minimizing the risk of batch failures that can disrupt supply schedules. Furthermore, the use of commercially available enzymes from major suppliers ensures a secure supply of biocatalysts, mitigating the risk of single-source dependency. The process is designed to be scalable from kilogram to multi-ton production without significant re-optimization, allowing for flexible response to market demand fluctuations.
- Cost Reduction in Manufacturing: The replacement of stoichiometric chiral reagents with catalytic enzymes drastically lowers material costs per kilogram of product. Eliminating the need for preparative HPLC or chiral column chromatography removes a major bottleneck and expense from the purification workflow. The use of crystallization as the primary isolation technique further reduces solvent consumption and waste disposal costs. Additionally, the safer reaction conditions lower insurance premiums and safety compliance costs associated with handling hazardous chemicals. Overall, the streamlined process flow reduces the number of unit operations, leading to substantial cost savings in labor and utilities.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and widely available biocatalysts ensures that raw material sourcing is not a critical vulnerability. The process avoids reagents that are subject to strict regulatory controls or geopolitical supply constraints. Improved safety profiles mean fewer unplanned shutdowns due to safety incidents, ensuring continuous production capacity. The high selectivity of the enzymatic steps reduces the generation of hard-to-remove impurities, decreasing the likelihood of batch rejection during quality control. This reliability translates to more predictable lead times and the ability to maintain safety stock levels without excessive inventory costs.
- Scalability and Environmental Compliance: The aqueous nature of the enzymatic steps aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. Lower solvent usage and the absence of heavy metal catalysts simplify wastewater treatment and regulatory compliance. The process is inherently scalable, as enzymatic reactions often translate well from lab to plant scale with minimal parameter adjustment. The ability to recycle solvents like ethyl acetate and toluene further enhances the sustainability profile. This environmental advantage supports corporate sustainability goals and facilitates smoother regulatory approvals in regions with strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and examples provided in the patent documentation to ensure accuracy and relevance for decision-makers.
Q: How does this patent improve safety over previous methods?
A: The process eliminates the use of pyrophoric trimethylaluminum and avoids high-temperature K2CO3/DMSO conditions that pose runaway reaction risks, significantly enhancing operational safety.
Q: What is the advantage of the enzymatic resolution step?
A: Using commercial lipases like Lipolase allows for high enantiomeric excess without expensive chiral chromatography, reducing material costs and simplifying downstream processing.
Q: Is this route suitable for large-scale manufacturing?
A: Yes, the methodology replaces hazardous reagents with robust enzymatic steps and crystallization-driven purifications, which are inherently more scalable and controllable in industrial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolidine-3,4-dicarboxamide Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate complex patent methodologies like CN101611026A into commercial reality. Our CDMO team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand the critical importance of stringent purity specifications and rigorous QC labs in the pharmaceutical sector, and our facilities are equipped to handle the specific requirements of enzymatic and chemoenzymatic processes. Our commitment to quality ensures that every batch of pyrrolidine-3,4-dicarboxamide intermediate meets the highest standards for downstream API synthesis. We are dedicated to providing a seamless partnership that supports your drug development timeline from clinical trials to commercial launch.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for this critical intermediate. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to this safer, more efficient route. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project's unique requirements. By collaborating with us, you gain access to a partner who values innovation, safety, and reliability above all else. Let us help you secure a sustainable and cost-effective supply of high-purity pharmaceutical intermediates for your next generation of therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
