Advanced Synthesis of Garcinia Acid Derivatives for Commercial Scale-Up and Oncology Applications
The pharmaceutical landscape for oncology treatments is constantly evolving, driven by the need for agents that selectively target cancer cells while sparing healthy tissue. Patent CN1563014A, published in early 2005, introduces a groundbreaking series of chemically modified Garcinia acid derivatives, specifically focusing on the isomers Morellic acid, Neogambogic acid, and Allogambogic acid. These novel compounds address critical limitations associated with the natural extraction of Gamboge resin, which has historically been used in traditional medicine but suffers from poor water solubility and significant toxicity issues such as vascular irritation. By strategically modifying the core molecular framework, this technology enables the creation of pharmaceutically acceptable salts and derivatives that exhibit superior stability, enhanced partition ratios between fat and aqueous phases, and reduced side effects. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the chemical versatility of these derivatives is essential for developing next-generation anticancer formulations that maintain efficacy while improving patient safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional utilization of Gamboge resin and its primary active constituent, Morellic acid, has been hindered by inherent physicochemical instabilities that complicate drug formulation and administration. Natural Morellic acid, while possessing potent anticancer properties, is notoriously unstable in alkaline environments, leading to rapid hydrolysis that degrades the active ingredient before it can exert its therapeutic effect. Furthermore, clinical reports indicate that injections of total cambogic acid extracts can cause severe local irritant effects on blood vessels, leading to superficial phlebitis and pain if the concentration is too high or the infusion rate is too fast. The reliance on natural extraction also introduces batch-to-batch variability in impurity profiles, making it difficult to meet the stringent quality control standards required for modern regulatory approval. These factors collectively increase the cost reduction in API manufacturing challenges, as extensive purification and stabilization efforts are required to make the natural product viable for systemic administration.
The Novel Approach
The innovative strategy outlined in the patent data involves precise chemical modifications of the Garcinia acid scaffold to overcome these physiological and chemical barriers without compromising the core pharmacophore responsible for anticancer activity. By converting ketone carbonyl groups into hydroxyls or introducing ester and amide functionalities at the C30 carboxyl position, the new derivatives achieve a much more favorable balance of lipophilicity and hydrophilicity. This structural tuning not only improves water solubility, allowing for easier formulation into injectables or oral dosage forms, but also significantly enhances chemical stability against hydrolysis. The ability to synthesize specific halogenated or reduced analogs provides a robust library of candidates for structure-activity relationship (SAR) studies, enabling medicinal chemists to optimize potency and minimize toxicity. This approach transforms a problematic natural extract into a versatile platform for high-purity OLED material or pharmaceutical intermediate development, ensuring consistent supply and performance.
Mechanistic Insights into Chemical Modification of Garcinia Acid Scaffold
The core of this technological advancement lies in the selective functionalization of the polyprenylated xanthone structure common to Morellic, Neogambogic, and Allogambogic acids. As illustrated in the structural diagrams, the molecule possesses multiple reactive sites, including ketone carbonyls at positions C8 and C12, a carboxylic acid at C30, and several conjugated double bonds in the side chains. The patent details a systematic approach where reducing agents like sodium borohydride or lithium aluminum hydride are employed to selectively reduce the ketone groups to hydroxyls, thereby altering the electronic distribution and hydrogen bonding capacity of the molecule. This reduction is critical because it removes the electrophilic character of the carbonyls, potentially reducing non-specific binding to plasma proteins and improving metabolic stability. Furthermore, the carboxyl group at C30 serves as a handle for amidation or esterification using coupling reagents such as EDC and DMAP, allowing for the attachment of various alkyl or amino chains that can drastically alter the biodistribution and cellular uptake of the drug candidate.
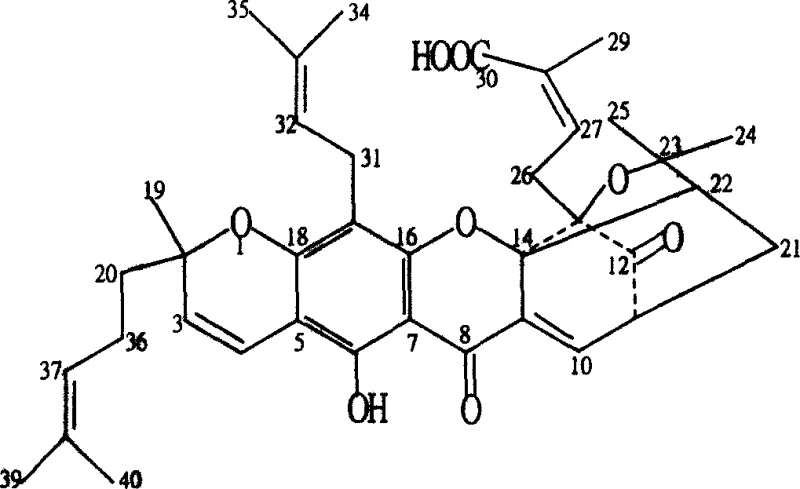
In addition to reduction and acylation, the patent describes halogenation strategies that introduce chlorine or bromine atoms across the double bonds or via substitution reactions. For instance, reacting the acid with liquid bromine or hydrogen bromide under controlled low-temperature conditions yields di-halogenated or mono-halogenated derivatives. These halogenated species are not merely inert variants; they serve as crucial intermediates for further nucleophilic substitution reactions, such as replacing halogen atoms with amino groups to create water-soluble amine salts. The mechanistic precision required to control these reactions—ensuring that the sensitive lactone rings or other functional groups remain intact while modifying specific sites—demonstrates a high level of synthetic sophistication. For a reliable agrochemical intermediate supplier or pharma partner, mastering these selective transformations is key to producing materials that meet the rigorous specifications for clinical trials and commercial launch.
How to Synthesize Garcinia Acid Derivatives Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction conditions and purification to ensure the integrity of the complex polycyclic structure. The process generally begins with the isolation of highly purified starting material, often achieved through pyridinium salt crystallization followed by acidification, ensuring an HPLC purity greater than 97% before derivatization begins. Subsequent steps involve dissolving the purified acid in appropriate solvents like methanol or dichloromethane and adding stoichiometric amounts of reagents under strict temperature control, typically ranging from 0°C to room temperature to prevent degradation. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, reaction times, and workup procedures necessary to achieve optimal yields and purity.
- Dissolve purified Morellic acid in an organic solvent like methanol and reduce ketone carbonyls at C8 and C12 positions using sodium borohydride or lithium aluminum hydride to form hydroxyl derivatives.
- Perform esterification or amidation at the C30 carboxyl group using coupling agents like EDC and DMAP with corresponding alcohols or amines to enhance solubility profiles.
- Optionally introduce halogen atoms via addition reactions with bromine or hydrogen halides at double bond positions to create novel halogenated derivatives with modified biological activity.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the shift from relying solely on natural extraction to implementing semi-synthetic modification routes offers substantial strategic benefits. Natural products are inherently subject to agricultural variables, seasonal fluctuations, and geopolitical risks that can disrupt the continuity of supply. By establishing a synthetic or semi-synthetic pathway for Garcinia acid derivatives, manufacturers can decouple production from raw material scarcity, ensuring a consistent and reliable flow of intermediates regardless of external environmental factors. This stability is crucial for long-term project planning and inventory management, allowing procurement managers to secure contracts with greater confidence and reduced risk of stockouts. Moreover, the ability to tune the physical properties of the molecule means that downstream processing, such as crystallization and drying, can be optimized for higher throughput and lower energy consumption.
- Cost Reduction in Manufacturing: The synthetic routes described eliminate the need for extensive and costly purification steps often associated with crude natural extracts, which frequently contain complex mixtures of structurally similar impurities. By starting with a defined chemical entity and performing selective transformations, the overall process mass intensity is improved, leading to significant waste reduction and lower solvent usage. The use of common, commercially available reagents like sodium borohydride and EDC further drives down raw material costs compared to specialized biocatalysts or rare earth metals. Additionally, the improved stability of the derivatives reduces losses during storage and transportation, contributing to substantial cost savings across the entire value chain without compromising quality.
- Enhanced Supply Chain Reliability: Implementing a robust chemical synthesis protocol ensures that production schedules are not dictated by the harvest cycles of Gamboge trees, which can be unpredictable. This synthetic reliability allows for just-in-time manufacturing models and reduces the need for large safety stocks of volatile natural materials. The scalability of the reactions, which utilize standard unit operations like stirring, filtration, and distillation, means that production can be rapidly ramped up to meet surging demand without requiring bespoke equipment. This flexibility is a key advantage for supply chain heads looking to mitigate risk and maintain operational agility in a dynamic market environment.
- Scalability and Environmental Compliance: The processes outlined prioritize atom economy and the use of recyclable solvents, aligning with modern green chemistry principles and regulatory expectations for environmental compliance. The elimination of heavy metal catalysts in favor of organic reagents simplifies waste treatment and reduces the burden of hazardous waste disposal. Furthermore, the high selectivity of the reactions minimizes the formation of by-products, streamlining the purification process and reducing the volume of effluent generated. This environmentally conscious approach not only lowers operational costs related to waste management but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized Garcinia acid derivatives. The answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating these intermediates into their own development pipelines or supply networks.
Q: What are the primary advantages of Garcinia acid derivatives over natural extracts?
A: The derivatives described in CN1563014A offer significantly higher stability in aqueous solutions compared to natural Morellic acid, which is prone to hydrolysis in alkaline conditions. Additionally, chemical modification allows for tunable solubility and reduced vascular irritation.
Q: How does the synthesis process ensure high purity for pharmaceutical use?
A: The patent outlines rigorous purification protocols including silica gel column chromatography and recrystallization steps, ensuring HPLC purity levels exceeding 97% for the final active pharmaceutical intermediates.
Q: Can these derivatives be scaled for commercial production?
A: Yes, the synthesis relies on standard organic unit operations such as reduction, esterification, and filtration, which are readily adaptable from laboratory scale to multi-ton commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Garcinia Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of modified natural products in the fight against cancer and infectious diseases. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity Garcinia acid derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific halogenated analogs or large-scale supply of esterified intermediates, our facility is designed to adapt to your unique project requirements while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to unlock the full commercial potential of this technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timeline and bring life-saving therapies to market faster. Let us be your partner in innovation, combining deep technical expertise with a customer-centric approach to drive your success in the competitive pharmaceutical landscape.
