Advanced Synthetic Route for High-Purity Ezetimibe Intermediates via Dithiolane Protection
Advanced Synthetic Route for High-Purity Ezetimibe Intermediates via Dithiolane Protection
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for the production of critical lipid-lowering agents such as ezetimibe. Patent CN107488138B introduces a transformative synthetic methodology that addresses longstanding challenges in the manufacture of key ezetimibe intermediates. This novel approach leverages a strategic 1,3-dithiolane protecting group to stabilize the carbonyl functionality during the construction of the complex beta-lactam core. Unlike traditional methods that rely on ethylene glycol ketals, which often result in difficult-to-handle fine solids and variable yields, this invention utilizes a dithiolane moiety that offers superior steric protection and cleaner deprotection profiles. The process begins with the conversion of 4-(4-fluoro-benzoyl)-butyric acid into a protected acid derivative, setting the stage for a highly stereoselective assembly of the azetidinone ring system. For procurement specialists and supply chain managers, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials with consistent batch-to-batch reproducibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ezetimibe precursors has been plagued by inefficiencies associated with standard ketal protection strategies. As illustrated in prior art such as Patent WO2007072088, the use of ethylene glycol to protect the ketone functionality often leads to the formation of intermediate compounds that are physically difficult to manage. These intermediates frequently manifest as extremely fine solid substances that resist effective crystallization during post-treatment phases. The physical nature of these solids complicates filtration processes, leading to product loss where small amounts of valuable material flow out with the filtrate. Furthermore, the instability of these conventional protecting groups under certain reaction conditions can lead to premature deprotection or side reactions, generating significant impurities that are costly to remove. The cumulative effect of these issues is a process characterized by low overall yields, poor product quality, and increased production costs, making it less attractive for cost reduction in API manufacturing initiatives.
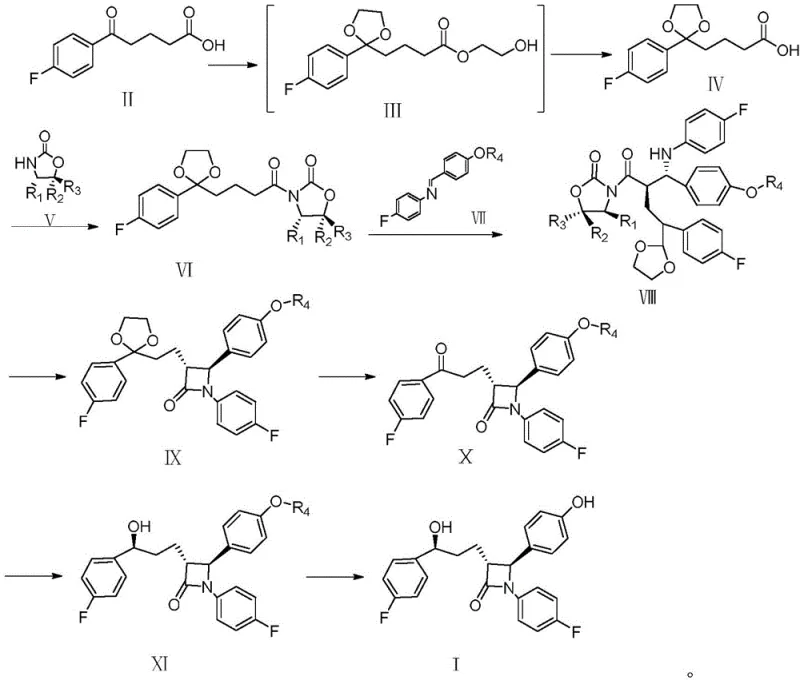
The Novel Approach
In stark contrast to these legacy methods, the route disclosed in CN107488138B employs a 1,3-dithiolane protecting group which fundamentally alters the physical and chemical landscape of the synthesis. This sulfur-based acetal provides robust protection against nucleophilic attack and harsh reaction conditions while remaining stable throughout the critical acylation and cyclization steps. The strategic advantage lies in the ease of removal; the dithiolane group can be selectively hydrolyzed under mild conditions without compromising the integrity of the sensitive beta-lactam ring or the chiral centers. This selectivity ensures that the final intermediate is obtained with exceptional purity, often exceeding 99%, thereby reducing the burden on downstream purification units. By eliminating the formation of fine, uncrystallizable solids, the new method streamlines the isolation process, enhancing throughput and ensuring that the commercial scale-up of complex pharmaceutical intermediates is both technically feasible and economically sound.
Mechanistic Insights into Dithiolane Protection and Evans Auxiliary Cyclization
The core of this synthetic innovation lies in the precise manipulation of functional groups to control stereochemistry and reactivity. The process initiates with the reaction of 4-(4-fluoro-benzoyl)-butyric acid with ethanedithiol in the presence of a Lewis acid catalyst, such as zinc chloride, and a water scavenger like trimethyl orthoformate. This step efficiently converts the ketone into the corresponding 1,3-dithiolane derivative. Mechanistically, the sulfur atoms coordinate with the Lewis acid, facilitating the nucleophilic attack on the carbonyl carbon and stabilizing the resulting cationic intermediate. Following this protection, the carboxylic acid is activated using pivaloyl chloride to form a mixed anhydride, which then reacts with a chiral oxazolidinone auxiliary (specifically 4-phenyl-2-oxazolidinone). This acylation step is crucial as it installs the chiral template necessary for subsequent stereocontrol. The resulting imide is then subjected to a titanium-mediated reaction with a fluorophenyl imine. The titanium tetrachloride acts as a Lewis acid to activate the imine, promoting a highly diastereoselective addition that establishes the critical stereocenters required for the biological activity of the final drug substance.
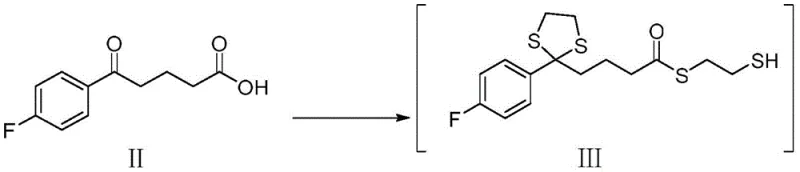
Following the establishment of stereochemistry, the molecule undergoes an intramolecular cyclization to form the four-membered beta-lactam ring, a structural motif essential for ezetimibe's mechanism of action. This cyclization is facilitated by silanization agents like bis(trimethylsilyl)acetamide, which activate the amide nitrogen for nucleophilic attack on the ester carbonyl. The final step involves the hydrolytic removal of the dithiolane protecting group using pyridinium p-toluenesulfonate (PPTS) in a mixture of ethanol and water. This mild acidic condition is specifically chosen to cleave the thioacetal without opening the strained beta-lactam ring. The result is a keto-functionalized intermediate ready for the final chiral reduction to ezetimibe. This mechanistic pathway demonstrates a sophisticated understanding of orthogonal protection strategies, ensuring that each functional group reacts only when intended, thereby minimizing the generation of regioisomers and other process-related impurities.
How to Synthesize Ezetimibe Intermediate Efficiently
The execution of this synthetic route requires careful attention to reaction parameters, particularly temperature control and reagent stoichiometry, to maximize yield and purity. The process is designed to be operationally simple, utilizing common organic solvents such as tetrahydrofuran, dichloromethane, and acetonitrile, which are readily available in most manufacturing facilities. The initial protection step is conducted at ambient temperatures (20-25°C), eliminating the need for energy-intensive cooling or heating systems. Subsequent acylation and cyclization steps utilize standard laboratory equipment and do not require exotic catalysts or high-pressure reactors. For technical teams looking to implement this process, the detailed standardized synthesis steps are outlined below to ensure reproducibility and safety.
- Convert 4-(4-fluoro-benzoyl)-butyric acid to the dithiolane-protected acid using ethanedithiol and Lewis acid catalysis.
- Acylate chiral oxazolidinone with the protected acid using pivaloyl chloride to form the mixed anhydride intermediate.
- React the acylated oxazolidinone with an imine derivative under titanium catalysis to establish stereochemistry, followed by cyclization and deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers compelling advantages for organizations focused on optimizing their supply chain resilience and cost structures. The primary driver of value is the significant improvement in process efficiency, which directly translates to lower manufacturing costs. By replacing problematic ethylene glycol protection with the more robust 1,3-dithiolane system, the process eliminates the bottlenecks associated with difficult filtrations and low-yielding crystallizations. This enhancement in physical handling characteristics means that less material is lost during workup, and the time required for isolation is drastically reduced. Furthermore, the high purity of the intermediates generated reduces the need for extensive chromatographic purification or multiple recrystallizations, which are often the most expensive and time-consuming parts of API production. For a reliable pharmaceutical intermediate supplier, this efficiency allows for more competitive pricing and faster turnaround times.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, such as palladium used in some prior art routes, represents a direct saving in raw material costs. Additionally, the use of mild reaction conditions reduces energy consumption for heating and cooling, while the high yields minimize the cost of goods sold per kilogram of product. The simplified purification protocol further lowers solvent usage and waste disposal costs, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The starting materials for this route, including 4-(4-fluoro-benzoyl)-butyric acid and ethanedithiol, are commodity chemicals with stable global supply chains. This reduces the risk of production delays caused by the scarcity of specialized reagents. Moreover, the robustness of the chemistry means that the process is less sensitive to minor variations in raw material quality or environmental conditions, ensuring consistent output even during large-scale production runs. This reliability is critical for maintaining continuous supply to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard reactor configurations. The avoidance of hazardous reagents and the generation of fewer toxic by-products align with modern green chemistry principles. The ability to recycle solvents like THF and DCM, combined with the reduced waste stream from higher yields, simplifies environmental compliance and wastewater treatment, making the facility more sustainable and reducing regulatory risks associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ezetimibe intermediate synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of the technology. Understanding these details is essential for R&D directors and procurement officers evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What is the primary advantage of using 1,3-dithiolane over ethylene glycol in this synthesis?
A: The 1,3-dithiolane group provides superior steric hindrance and stability during subsequent reactions, preventing side reactions and allowing for easier, cleaner deprotection compared to the fine, difficult-to-crystallize solids often produced by ethylene glycol protection.
Q: How does this route improve impurity profiles for GMP manufacturing?
A: By utilizing selective protecting groups that are easily removed and avoiding harsh conditions that degrade sensitive functional groups, the process minimizes the formation of by-products, resulting in intermediates with purity levels exceeding 99%.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method employs mild reaction temperatures (20-25°C) and readily available reagents like zinc chloride and pivaloyl chloride, avoiding expensive transition metal catalysts and complex cryogenic conditions, which facilitates safe and cost-effective scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and high-quality synthetic routes in the competitive landscape of pharmaceutical manufacturing. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN107488138B and is fully prepared to leverage this advanced dithiolane-protection strategy for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ezetimibe intermediate meets the highest global standards for safety and efficacy.
We invite potential partners to engage with our technical team to discuss how this innovative synthesis can optimize your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a stable, cost-effective supply of high-purity ezetimibe intermediates that will drive the success of your final drug product.
