Scalable Batch Production of Sodium Taurodeoxycholate: A Technical Breakthrough for Pharmaceutical Manufacturing
Scalable Batch Production of Sodium Taurodeoxycholate: A Technical Breakthrough for Pharmaceutical Manufacturing
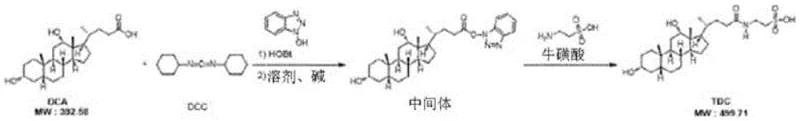
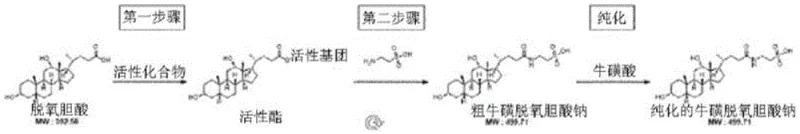
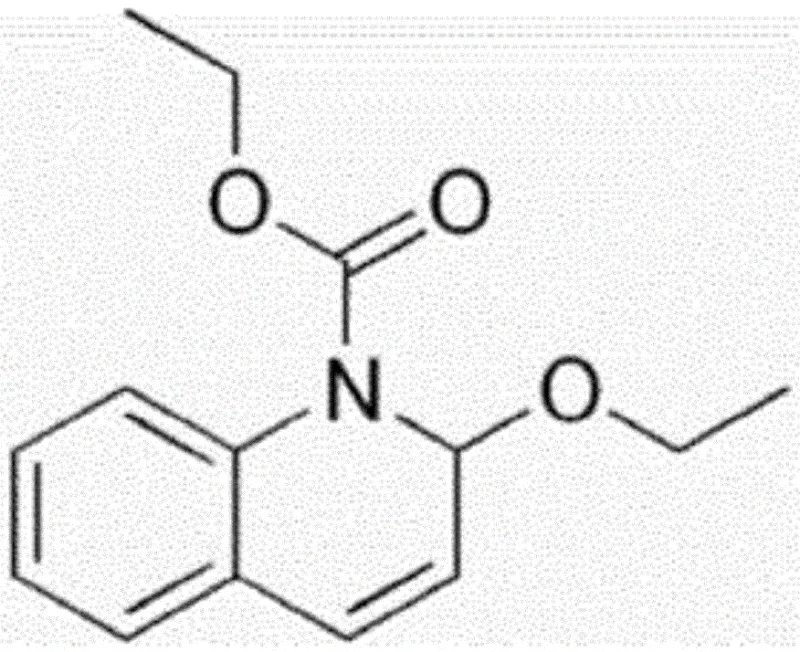
The pharmaceutical industry constantly seeks robust, scalable pathways for complex bile acid derivatives, particularly for applications in metabolic and cognitive health therapeutics. Patent CN116249536A introduces a transformative batch production method for sodium taurodeoxycholate, addressing long-standing challenges in purification and yield consistency. This technology shifts the paradigm from labor-intensive, low-yield laboratory syntheses to a streamlined industrial process capable of delivering high-purity intermediates. By utilizing N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline (EEDQ) as a coupling agent and implementing a novel isopropanol-based batch washing protocol, the method significantly reduces impurity profiles while maintaining operational simplicity. For procurement and supply chain leaders, this represents a critical advancement in securing reliable sources of high-quality pharmaceutical intermediates without the bottlenecks associated with traditional chromatographic separations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sodium taurodeoxycholate has been plagued by inefficient activation strategies and arduous purification requirements. Conventional routes often rely on reagents such as isobutyl chloroformate, pivaloyl chloride, or carbodiimides like DCC and HATU. While these reagents can drive the amide coupling reaction, they frequently generate substantial quantities of side products and urea derivatives that possess solubility characteristics nearly identical to the target molecule. For instance, methods utilizing DCC/HOBt often result in conversion rates that, while high, leave behind stubborn impurities requiring complex work-up procedures. Furthermore, techniques involving triphenylphosphine (TPP) and DTBT suffer from incomplete conversions and the retention of phosphine oxide salts, which are notoriously difficult to separate from the final bile acid salt. These inefficiencies not only depress overall yields, often keeping them below commercially viable thresholds, but also necessitate the use of expensive and time-consuming column chromatography, rendering them unsuitable for multi-kilogram scale manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN116249536A offers a refined pathway that prioritizes both chemical efficiency and downstream processability. The core innovation lies in the strategic combination of EEDQ-mediated coupling followed by a sophisticated recrystallization regimen using isopropanol and water mixtures. This approach bypasses the formation of tenacious by-products common in chloroformate activations. Instead of relying on silica gel columns to scrub impurities, the process exploits subtle solubility differentials through repeated batch washing and recrystallization cycles. The protocol allows for the direct processing of wet cakes between purification steps, eliminating energy-intensive drying phases between cycles. This continuous liquid-phase handling not only accelerates the production timeline but also ensures that the crystal lattice of the sodium taurodeoxycholate is formed under optimal thermodynamic conditions, resulting in a final product with purity exceeding 99% and single impurity levels well below 0.1%. This represents a significant leap forward for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into EEDQ-Mediated Coupling and Purification
The chemical elegance of this process begins with the activation of deoxycholic acid. EEDQ acts as a zero-length crosslinker that converts the carboxylic acid group of the bile acid into a highly reactive mixed anhydride or active ester intermediate in situ. Unlike harsher activating agents, EEDQ operates under mild thermal conditions, typically around 40°C, which preserves the stereochemical integrity of the steroid backbone. The subsequent nucleophilic attack by sodium taurate proceeds efficiently in ethanol, a green solvent that facilitates easy removal post-reaction. The mechanistic advantage here is the generation of quinoline as a by-product, which is significantly more soluble in the wash solvents than the target amphiphilic bile acid salt, allowing for its facile removal during the initial filtration steps. This intrinsic difference in physicochemical properties is the cornerstone of the method's success, ensuring that the crude material entering the purification stage is already of relatively high quality.
Furthermore, the purification mechanism relies on a precise manipulation of solvent polarity. The use of isopropanol mixed with water in specific volume ratios (ranging from 1:1 to 1:10) creates a solvent environment where the sodium taurodeoxycholate exhibits temperature-dependent solubility ideal for recrystallization. By heating the mixture to dissolve the solid completely and then slowly cooling it over several hours, the system encourages the growth of large, pure crystals while excluding structurally similar impurities like unreacted deoxycholic acid or taurocholic analogs. The repetition of this cycle two to three times acts as a multiplier for purity, progressively stripping away trace contaminants without the need for additional chemical reagents. This physical purification strategy is far more robust and scalable than chemical scavenging methods, providing a consistent quality profile that is essential for regulatory compliance in API production.
How to Synthesize Sodium Taurodeoxycholate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from bench-scale experimentation to pilot plant operations. The process is designed to be modular, allowing for adjustments in solvent volumes and temperature ramps based on the specific reactor geometry and batch size. The key to success lies in strict adherence to the temperature profiles during the crystallization phases, as rapid cooling can trap impurities within the crystal matrix. Operators must ensure that the dissolution step reaches complete clarity before initiating the cooling ramp to guarantee homogeneity. The following guide summarizes the critical operational parameters required to replicate the high yields and purity reported in the intellectual property documentation.
- Synthesize crude sodium taurodeoxycholate by reacting deoxycholic acid and sodium taurate with EEDQ in ethanol at controlled temperatures.
- Wash the crude reaction mixture with organic solvents such as ethanol and acetone to remove unreacted starting materials and by-products.
- Purify the filter cake through repeated recrystallization using isopropanol and water mixtures, followed by final drying to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this batch production technology translates directly into enhanced operational resilience and cost efficiency. The elimination of column chromatography is perhaps the most significant economic driver, as it removes the need for large volumes of silica gel and the associated disposal costs, while also drastically reducing solvent consumption per kilogram of product. The ability to process wet cakes without intermediate drying steps further compresses the manufacturing cycle time, allowing for higher throughput in existing facility footprints. This streamlined workflow reduces the dependency on specialized separation equipment, lowering capital expenditure barriers for scaling production. Consequently, this method supports a more agile supply chain capable of responding rapidly to fluctuations in market demand for bile acid derivatives.
- Cost Reduction in Manufacturing: The substitution of expensive coupling reagents and chromatographic media with recyclable solvents like ethanol and isopropanol drives down the variable cost of goods sold. By avoiding the use of precious metal catalysts or complex scavengers, the raw material bill is optimized. Additionally, the high recovery rates achieved through the multi-stage recrystallization process minimize waste generation, ensuring that a greater proportion of input materials are converted into saleable product. This efficiency creates substantial margin improvements that can be passed down the supply chain or reinvested into further process optimization.
- Enhanced Supply Chain Reliability: The robustness of the EEDQ coupling chemistry ensures consistent batch-to-batch reproducibility, a critical factor for maintaining uninterrupted supply to downstream API manufacturers. The reliance on commodity chemicals and standard unit operations like filtration and crystallization mitigates the risk of supply disruptions associated with specialty reagents. Furthermore, the scalability of the process means that production volumes can be increased simply by running larger batches or adding parallel reactors, rather than redesigning the entire synthetic route. This flexibility provides a secure foundation for long-term supply agreements.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process is inherently greener due to the reduced solvent intensity and the absence of toxic heavy metal residues. The use of isopropanol and ethanol aligns with modern sustainability goals, facilitating easier regulatory approval and waste management. The simplified work-up procedure also reduces the operator exposure time to potentially hazardous intermediates, enhancing workplace safety. These factors collectively position the manufacturing process as compliant with stringent international environmental standards, reducing the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this batch production technology. The answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering transparency into the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What distinguishes this batch production method from conventional synthesis routes?
A: Unlike conventional methods using chloroformates or carbodiimides which generate difficult-to-remove impurities, this method utilizes EEDQ for cleaner activation and employs a unique isopropanol batch washing technique that eliminates the need for complex column chromatography.
Q: How does the purification process ensure high purity levels?
A: The process leverages specific solubility differences in isopropanol-water mixtures. By repeating the dissolution and recrystallization cycle two to three times without intermediate drying, impurities are effectively washed away, achieving purity levels exceeding 99%.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly demonstrates batch production capabilities exceeding 1kg per run with yields over 25%. The avoidance of specialized separation equipment like preparative HPLC makes it highly scalable for industrial API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Taurodeoxycholate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation therapeutics. Our technical team has extensively analyzed the batch production methodology described in CN116249536A and possesses the expertise to implement this advanced route at commercial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of sodium taurodeoxycholate meets the exacting standards required for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify the superior quality and consistency of our offerings. Together, we can drive forward the availability of essential bile acid derivatives for the global healthcare market.
