Advanced Clean Preparation of Aminothiazolyloxime for Cephalosporin Manufacturing
Introduction to Patent CN112661719B: A Paradigm Shift in Beta-Lactam Intermediate Synthesis
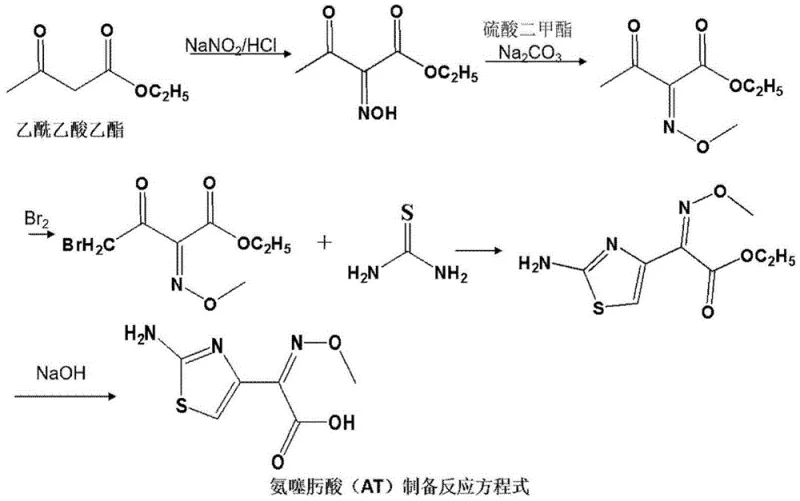
The pharmaceutical industry is constantly seeking sustainable methodologies to produce critical antibiotic intermediates, and the clean preparation process of aminothiazolyloxime disclosed in patent CN112661719B represents a significant technological leap forward for manufacturers of third-generation cephalosporins. This patent addresses the longstanding environmental and economic burdens associated with the traditional synthesis of aminothiazolyloxime, a pivotal building block for drugs like cefotaxime and ceftriaxone. By fundamentally altering the oximation reagent from sodium nitrite to calcium nitrite and integrating a closed-loop acid recycling system, the invention offers a robust solution to the high-salt wastewater issues that have plagued this sector for decades. For R&D directors and process engineers, this approach not only simplifies the operational workflow by eliminating multiple solid-liquid separation steps but also enhances the overall atom economy of the production line. The strategic implementation of calcium sulfate precipitation allows for the physical removal of inorganic salts early in the process, preventing them from contaminating downstream reactions and complicating wastewater treatment facilities. Furthermore, the ability to recycle acidic quench water for final product crystallization demonstrates a sophisticated understanding of process integration that aligns perfectly with modern green chemistry principles. As global regulatory pressures on pharmaceutical effluent tighten, adopting such a clean technology becomes not just an option but a necessity for maintaining a competitive and compliant supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of aminothiazolyloxime has relied heavily on sodium-based reagents, which inadvertently create massive volumes of difficult-to-treat saline wastewater. In the traditional oximation step, sodium nitrite is dissolved in large quantities of pure water, and upon acidification, it generates a mother liquor saturated with sodium salts that cannot be easily separated or reused. Similarly, the subsequent etherification step requires substantial amounts of sodium carbonate, further exacerbating the salt load in the effluent stream. Perhaps most critically, the hydrolysis stage traditionally involves dissolving the intermediate ester in alkali and then neutralizing with fresh acid to precipitate the product, generating yet another stream of high-concentration sodium salt wastewater. Additionally, the halogenation step produces strong acidic wastewater upon quenching, which is typically neutralized with base and discharged, representing a total loss of valuable acidic resources. These cumulative inefficiencies result in a process where more than 30 tons of high-salt wastewater can be generated for every single ton of product produced, creating a severe bottleneck for environmental compliance and cost efficiency. The multiple solid isolation steps required for the intermediate ester also introduce opportunities for product loss, yield reduction, and increased labor intensity, making the conventional route increasingly unsustainable in a high-volume commercial environment.
The Novel Approach
The innovative process described in CN112661719B dismantles these inefficiencies by substituting sodium nitrite with calcium nitrite and sulfuric acid, a change that fundamentally alters the physical state of the byproduct salt. Instead of remaining dissolved in the wastewater, the resulting calcium sulfate precipitates as a solid during the oximation reaction, allowing it to be filtered off and removed from the process stream entirely. This simple yet profound modification means that the subsequent etherification can proceed in a low-salt environment, drastically reducing the volume and toxicity of the wastewater generated. Moreover, the novel approach eliminates the need to isolate the ethyl aminothiazolyloximate intermediate as a solid; instead, it is extracted into an organic phase (chloroform) and directly hydrolyzed in situ. This continuous liquid-phase processing avoids the yield losses associated with filtering and washing solids. Most impressively, the strong acidic water generated during the halogenation quench is not discarded but is captured and repurposed to adjust the pH during the final crystallization of the aminothiazolyloxime acid. This closed-loop strategy not only neutralizes the waste acid problem but also eliminates the need for purchasing fresh acid for the final step, creating a synergistic effect that lowers both raw material costs and waste disposal fees significantly.
Mechanistic Insights into Calcium-Mediated Oximation and In-Situ Hydrolysis
From a mechanistic perspective, the substitution of sodium with calcium in the oximation step leverages the low solubility product of calcium sulfate to drive the reaction equilibrium and facilitate purification. When ethyl acetoacetate reacts with calcium nitrite in the presence of sulfuric acid, the nitrous acid generated in situ attacks the active methylene group to form the oxime, while the calcium ions immediately bind with sulfate ions to form insoluble calcium sulfate crystals. This precipitation acts as a driving force for the reaction and simultaneously purifies the reaction mixture by removing the inorganic counter-ions that would otherwise accumulate. The filtrate, now largely free of dissolved inorganic salts, provides an ideal medium for the subsequent methylation with dimethyl sulfate, ensuring high conversion rates without the interference of high ionic strength. Following methylation and halogenation, the process utilizes a biphasic system where the halogenated intermediate reacts with thiourea to form the thiazole ring. The addition of chloroform at this stage is critical, as it selectively extracts the organic thiazole derivative into the organic phase, leaving behind the aqueous acidic byproducts. This phase separation is the key enabler for the final innovation: the direct hydrolysis of the ester in the organic phase using liquid alkali. The hydrolysis converts the ester into its water-soluble carboxylate salt, which migrates back into the aqueous phase, leaving organic impurities behind in the chloroform layer.
The control of impurities in this novel route is inherently superior due to the minimization of unit operations where contamination typically occurs. In traditional methods, every filtration and drying step of the intermediate ester introduces risks of thermal degradation or mechanical entrapment of impurities. By maintaining the intermediate in solution until the final hydrolysis, the process minimizes these exposure points. Furthermore, the recycling of the acidic quench water serves a dual purpose: it acts as the precipitating agent for the final product and ensures that any residual basic impurities from the hydrolysis step are neutralized in a controlled manner. The pH adjustment using this recycled acid allows for precise crystallization conditions, promoting the formation of high-purity crystals while excluding soluble organic impurities that remain in the mother liquor. This meticulous control over the physicochemical environment throughout the synthesis ensures that the final aminothiazolyloxime meets the stringent purity specifications required for cephalosporin antibiotic production, thereby reducing the burden on downstream purification processes and enhancing the overall quality profile of the active pharmaceutical ingredient.
How to Synthesize Aminothiazolyloxime Efficiently
The synthesis of aminothiazolyloxime via this clean process involves a carefully orchestrated sequence of reactions that prioritize waste minimization and resource efficiency. The procedure begins with the oximation of ethyl acetoacetate using calcium nitrite and sulfuric acid at controlled low temperatures to ensure selective formation of the oxime and complete precipitation of calcium sulfate. Following filtration, the oxime is methylated in the filtrate, halogenated, and cyclized with thiourea to form the thiazole ring structure. The resulting intermediate is extracted into chloroform, hydrolyzed with sodium hydroxide, and finally precipitated using the recycled acidic water from the earlier halogenation step. For detailed operational parameters, stoichiometric ratios, and safety protocols, please refer to the standardized synthesis guide below.
- Perform oximation of ethyl acetoacetate using calcium nitrite and sulfuric acid to precipitate calcium sulfate, followed by filtration and methylation with dimethyl sulfate.
- Execute halogenation and cyclization with thiourea, extracting the intermediate into chloroform to separate from acidic aqueous byproducts.
- Hydrolyze the organic phase with liquid alkali, then utilize the recycled acidic water from the halogenation step to precipitate the final purified acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the technology described in CN112661719B offers tangible benefits that extend far beyond mere regulatory compliance. The primary advantage lies in the drastic reduction of wastewater treatment costs, as the elimination of high-salt sodium wastewater removes the need for expensive evaporation or advanced oxidation processes typically required to handle such effluents. By converting the salt byproduct into a filterable solid (calcium sulfate), the facility can significantly lower its environmental footprint and avoid the hefty fines associated with saline discharge limits. Moreover, the recycling of acidic water for the final crystallization step reduces the consumption of fresh mineral acids, leading to direct savings in raw material procurement budgets. The simplification of the process flow, particularly the removal of the intermediate solid isolation step, translates to shorter batch cycle times and higher throughput capacity without the need for additional capital investment in equipment. This increased operational efficiency ensures a more reliable supply of aminothiazolyloxime, mitigating the risk of production delays that can ripple through the entire cephalosporin supply chain.
- Cost Reduction in Manufacturing: The shift to a calcium-based oximation process fundamentally alters the cost structure by eliminating the generation of high-salt wastewater, which is notoriously expensive to treat and dispose of in the pharmaceutical industry. By precipitating calcium sulfate as a solid, the process avoids the accumulation of dissolved salts that necessitate energy-intensive evaporation or specialized membrane filtration systems. Additionally, the reuse of acidic quench water for product crystallization negates the need for purchasing fresh acid for this specific unit operation, resulting in compound savings on chemical inputs. The reduction in unit operations, specifically the removal of the intermediate drying and milling steps, also lowers utility consumption (electricity and steam) and reduces labor hours per batch. These cumulative efficiencies drive down the overall cost of goods sold (COGS), providing a competitive pricing advantage in the global market for beta-lactam intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by environmental shutdowns or bottlenecks in waste treatment capacity, both of which are mitigated by this cleaner technology. The reduced volume and toxicity of the wastewater make the manufacturing process more resilient to fluctuating environmental regulations, ensuring uninterrupted production even during periods of strict enforcement. Furthermore, the simplified process flow with fewer solid handling steps reduces the likelihood of mechanical failures or operational errors that can cause batch failures. The ability to recycle process streams internally creates a more self-sufficient production loop, decreasing dependence on external utilities and waste disposal services. This robustness ensures that buyers can rely on consistent delivery schedules and stable inventory levels, which is critical for maintaining the production of life-saving antibiotics like ceftriaxone and cefotaxime.
- Scalability and Environmental Compliance: Scaling up chemical processes often amplifies waste issues, but this technology is inherently designed for scalability due to its reliance on simple filtration and phase separation rather than complex distillation or crystallization of intermediates. The precipitation of calcium sulfate is a scalable unit operation that can be easily managed in large reactors using standard filtration equipment. From an environmental standpoint, the process aligns with the principles of green chemistry by maximizing atom economy and minimizing the use of auxiliary substances. The significant reduction in three wastes (wastewater, waste gas, and solid waste) facilitates easier permitting for new production lines and simplifies the renewal of existing environmental licenses. This forward-looking approach future-proofs the manufacturing asset against tightening global sustainability standards, making it a strategically sound investment for long-term growth in the fine chemicals sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this clean synthesis route for aminothiazolyloxime. These answers are derived directly from the technical disclosures and beneficial effects outlined in patent CN112661719B, providing clarity on how this method outperforms traditional sodium-based routes. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or procurement teams assessing the long-term value proposition of suppliers utilizing this methodology.
Q: How does the calcium nitrite method reduce wastewater compared to traditional sodium nitrite processes?
A: The traditional process generates high-concentration sodium salt wastewater during oximation and etherification. By using calcium nitrite and sulfuric acid, the salt byproduct (calcium sulfate) precipitates as a solid and is filtered off, drastically reducing the dissolved salt load in the wastewater and allowing for easier treatment or reuse of the mother liquor.
Q: What are the purity advantages of avoiding solid isolation of the intermediate ester?
A: Traditional methods require cooling, crystallizing, filtering, and washing the ethyl aminothiazolyloximate intermediate, which often leads to product loss and impurity entrapment. The patented method keeps the intermediate in an organic solution (chloroform) for direct hydrolysis, minimizing mechanical losses and exposure to potential contaminants during solid handling.
Q: Can the acidic wastewater from the halogenation step be safely recycled?
A: Yes, the process is designed to capture the strong acidic water generated during the halogenation quench. Instead of neutralizing and discharging it, this acidic water is strategically reused in the final step to adjust the pH and precipitate the aminothiazolyloxime acid, effectively closing the loop on acid consumption and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminothiazolyloxime Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthesis routes is critical for the future of the pharmaceutical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the calcium nitrite oximation process can be seamlessly translated from the lab to the plant. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required for cephalosporin manufacturing. We understand that consistency is key in API intermediate supply, and our robust infrastructure allows us to maintain high-purity aminothiazolyloxime supplies even amidst fluctuating market demands. By leveraging our technical expertise, we help partners navigate the complexities of process optimization while adhering to the highest environmental and safety standards.
We invite you to collaborate with us to explore how this advanced synthesis technology can benefit your specific supply chain requirements. Our team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential reductions in waste treatment and raw material costs for your organization. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your production needs. Together, we can build a more sustainable and efficient supply chain for next-generation antibiotics, ensuring that life-saving medications remain accessible and affordable globally.
