Advanced Palladium-Catalyzed Synthesis of Piperidine-3-ylcarbamate Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks robust manufacturing routes for complex heterocyclic intermediates, particularly those serving as key building blocks for metabolic disease treatments. Patent CN101977899A introduces a transformative manufacturing method for piperidine-3-ylcarbamate compounds, which are critical precursors in the synthesis of next-generation diabetes therapeutics. This technology addresses significant economic and technical bottlenecks by replacing expensive noble metal catalysts with more accessible alternatives while simultaneously solving long-standing challenges in optical purity. The core innovation lies in a palladium-catalyzed hydrogenation process that operates under strictly controlled acidic conditions, ensuring high conversion rates without the prohibitive costs associated with traditional rhodium-based systems. For global procurement teams and R&D directors, this represents a pivotal shift towards more sustainable and cost-efficient supply chains for high-value pharmaceutical intermediates.
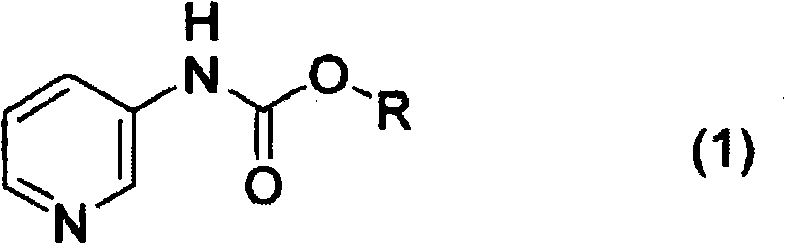
Historically, the synthesis of piperidine-3-ylcarbamate derivatives relied heavily on rhodium catalysts to facilitate the reduction of the pyridine ring. While chemically effective, the reliance on rhodium introduced severe economic constraints due to the metal's volatility in price and scarcity in the global market. Furthermore, conventional methods often struggled to achieve satisfactory optical purity in the final 3-aminopiperidine products, necessitating complex and yield-loss-prone purification steps. The background art indicates that previous optical resolution techniques were industrially unsatisfactory, often failing to meet the stringent enantiomeric excess requirements demanded by modern regulatory bodies for chiral drug substances. These limitations created a pressing need for a manufacturing protocol that could decouple high performance from high cost, enabling scalable production without compromising on the stereochemical integrity of the molecule.
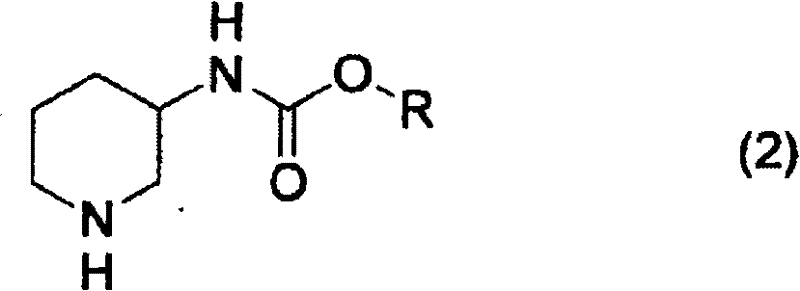
The novel approach detailed in the patent fundamentally reengineers the reduction step by utilizing palladium on carbon (Pd/C) in the presence of specific acid additives. Unlike prior art methods that might operate under neutral or uncontrolled conditions, this process mandates the adjustment of the reaction system's pH to a range of 1 to 7, with an optimal window between 2 and 6. This is achieved through the addition of carboxylic acid compounds, such as acetic acid, or phosphoric acid. The presence of these acids not only accelerates the reaction kinetics but also stabilizes the intermediate species, preventing the formation of undesirable by-products. By shifting from rhodium to palladium, the method drastically lowers the catalyst cost burden, while the pH control mechanism ensures that the yield remains consistently high, often exceeding 90% in optimized examples. This dual optimization of cost and yield makes the process exceptionally attractive for commercial scale-up.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and Optical Resolution
The mechanistic advantage of this process stems from the synergistic interaction between the palladium surface and the protonated substrate environment. In the absence of acid, the basic nitrogen of the pyridine ring can potentially coordinate too strongly with the metal surface or cause catalyst deactivation over time. By introducing acetic acid or phosphoric acid, the reaction medium maintains a protonated state that facilitates smoother hydrogen uptake and ring saturation. The patent data demonstrates that using acetic acid at roughly 1.5 to 6 molar equivalents relative to the substrate creates an ideal environment for the Pd/C catalyst to function at peak efficiency. This careful modulation of the reaction milieu ensures that the reduction proceeds cleanly to the piperidine stage without over-reduction or ring-opening side reactions, which are common pitfalls in heterogeneous hydrogenation of nitrogen-containing heterocycles.
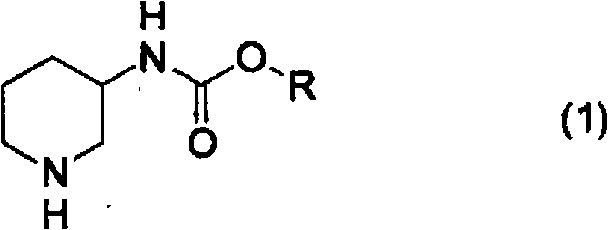
Following the hydrogenation, the resulting racemic piperidine-3-ylcarbamate undergoes a sophisticated optical resolution process to isolate the desired enantiomer. The patent identifies optically active mandelic acid, specifically R-mandelic acid, as the resolving agent of choice for substrates where the protecting group R is ethyl or tert-butyl. The mechanism involves the formation of diastereomeric salts between the racemic amine and the chiral acid. Due to the differences in solubility between the (R)-amine/(R)-acid salt and the (S)-amine/(R)-acid salt, selective crystallization can be achieved. The data shows that by carefully selecting solvents such as ethanol, methanol, or mixed alcohol systems, and controlling the cooling profile, optical purities of up to 99.8% ee can be attained after recrystallization. This level of stereocontrol is vital for ensuring the biological efficacy and safety profile of the final API.
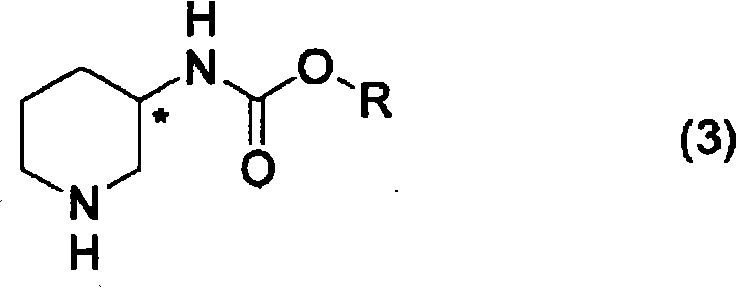
How to Synthesize Piperidine-3-ylcarbamate Efficiently
The synthesis pathway outlined in the patent provides a clear, step-by-step protocol for producing high-purity piperidine-3-ylcarbamate intermediates suitable for GMP manufacturing. The process begins with the carbamation of 3-aminopyridine, followed by the critical acidic hydrogenation step, and concludes with the chiral resolution. Each stage has been optimized to minimize waste and maximize throughput, making it an ideal candidate for technology transfer. The detailed standardized synthesis steps below provide the specific operational parameters required to replicate these results in a pilot or production plant setting.
- Carbamate 3-aminopyridine using alkyl chloroformate or di-tert-butyl dicarbonate to form the pyridine-3-ylcarbamate precursor.
- Perform catalytic hydrogenation using Palladium on Carbon (Pd/C) in the presence of acetic acid or phosphoric acid to maintain pH between 1 and 7.
- Resolve the racemic piperidine-3-ylcarbamate mixture using optically active mandelic acid to crystallize high-purity diastereomeric salts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this manufacturing method offers substantial strategic advantages beyond mere technical feasibility. The primary benefit is the drastic reduction in raw material costs associated with the catalyst system. By eliminating the dependency on rhodium, a metal subject to extreme market fluctuations and supply constraints, manufacturers can stabilize their cost of goods sold (COGS) and improve margin predictability. Furthermore, the use of palladium on carbon, a standard and widely available industrial catalyst, simplifies the sourcing process and reduces lead times for critical reagents. This shift enhances supply chain resilience, ensuring that production schedules are not disrupted by geopolitical or mining-related shortages of precious metals.
- Cost Reduction in Manufacturing: The replacement of expensive rhodium catalysts with palladium on carbon directly translates to significant savings in catalyst expenditure. Additionally, the high yields achieved through pH-controlled hydrogenation reduce the amount of starting material required per kilogram of final product, further lowering the overall material cost. The process also minimizes the need for complex purification steps post-reaction, as the acidic conditions suppress by-product formation, thereby reducing solvent consumption and waste disposal costs associated with downstream processing.
- Enhanced Supply Chain Reliability: Utilizing common reagents like acetic acid and phosphoric acid, alongside widely available palladium catalysts, mitigates the risk of supply bottlenecks. Unlike specialized ligands or rare metals that may have single-source suppliers, the inputs for this process are commodity chemicals with robust global supply networks. This availability ensures consistent production continuity and allows for flexible scaling of operations to meet fluctuating market demand without the fear of reagent scarcity impacting delivery timelines.
- Scalability and Environmental Compliance: The heterogeneous nature of the Pd/C catalyst allows for easy separation via filtration, facilitating a cleaner work-up procedure that is easily scalable from kilogram to tonne quantities. The ability to recycle the optically active mandelic acid from the mother liquor, as described in the patent, further contributes to a greener manufacturing footprint by reducing chemical waste. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, making the process not only economically viable but also environmentally responsible for long-term commercial operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the operational nuances and benefits of the method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the palladium catalyst preferred over rhodium for this synthesis?
A: The patent highlights that traditional rhodium catalysts are prohibitively expensive for large-scale manufacturing. Switching to palladium on carbon (Pd/C) significantly reduces raw material costs while maintaining high conversion rates when combined with specific pH controls.
Q: How does pH control impact the hydrogenation yield?
A: Maintaining the aqueous layer pH between 1 and 7, preferably 2 to 6, using carboxylic acids like acetic acid or phosphoric acid is critical. This acidic environment prevents catalyst poisoning and side reactions, leading to substantially higher yields of the piperidine derivative compared to neutral conditions.
Q: What resolving agent ensures high optical purity?
A: Optically active mandelic acid, particularly R-mandelic acid, is identified as the superior resolving agent. It forms diastereomeric salts with the piperidine-3-ylcarbamate that can be crystallized to achieve optical purities exceeding 99% ee after recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperidine-3-ylcarbamate Supplier
The technological advancements detailed in patent CN101977899A underscore the potential for producing high-quality piperidine-3-ylcarbamate intermediates with exceptional efficiency and purity. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical pharmaceutical building blocks. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for API synthesis. We are committed to leveraging such innovative chemistries to deliver superior value to our global partners.
We invite you to discuss how this optimized manufacturing route can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral intermediate synthesis can accelerate your drug development timeline while optimizing your overall production budget.
