Advanced Manufacturing Of Chiral Benzoxazine Intermediates For Global Antibiotic Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for high-value antibiotics, particularly fluoroquinolones like levofloxacin. Patent CN100432060C presents a significant technological advancement in the preparation of benzoxazine derivatives, which serve as critical chiral intermediates in this therapeutic class. The invention addresses longstanding challenges in stereoselectivity and process efficiency by offering multiple distinct pathways, labeled Method A through Method J, to construct the core benzoxazine scaffold. These methodologies provide manufacturers with flexible options to optimize production based on available infrastructure and raw material sourcing strategies. By integrating novel condensation techniques and improved resolution protocols, this technology enables the consistent production of high-purity intermediates essential for downstream drug synthesis.
Historically, the synthesis of optically active benzoxazine derivatives has been plagued by low yields and complex purification requirements. Conventional approaches, such as those cited in Japanese Patent No. 2,612,327, often suffer from epimerization under acidic or basic conditions, leading to a significant reduction in the yield of the desired optically active isomer. Other traditional methods rely on microbial reduction, which introduces variability due to biological factors and complicates product isolation because the physical properties of the product closely resemble the starting materials. Furthermore, processes utilizing expensive asymmetric acyloxyborate alkali metal hydrides as reducing agents impose substantial cost burdens and safety concerns regarding reagent handling and disposal in a commercial setting.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing routes for levofloxacin intermediates frequently depend on optical resolution of racemates, which theoretically limits the maximum yield to 50% unless efficient racemization and recycling loops are implemented. Methods reported in earlier literature often require additional protection and deprotection steps, such as the removal of p-toluenesulfonyloxy groups, which add unnecessary unit operations and increase the overall environmental footprint of the synthesis. The reliance on specific chiral starting materials from the natural chiral pool can also create supply chain bottlenecks, as the availability and price of these precursors fluctuate with agricultural and fermentation outputs. Additionally, the use of harsh reaction conditions in older protocols can lead to the formation of difficult-to-remove impurities, necessitating extensive chromatographic purification that is impractical for large-scale industrial applications.
The Novel Approach
The methodology disclosed in CN100432060C overcomes these hurdles by introducing versatile synthetic pathways that allow for the direct construction of the chiral center or its efficient resolution using accessible reagents. As illustrated in the general reaction scheme below, the process facilitates the conversion of simple aniline derivatives into complex benzoxazine structures through a series of controlled condensation and cyclization reactions. This approach minimizes the number of isolation steps and allows for the continuous processing of intermediates, thereby enhancing throughput and reducing solvent consumption. The patent specifically highlights the utility of phase transfer catalysts and specific inorganic bases that promote high yields without compromising optical purity, making the process economically viable for mass production.
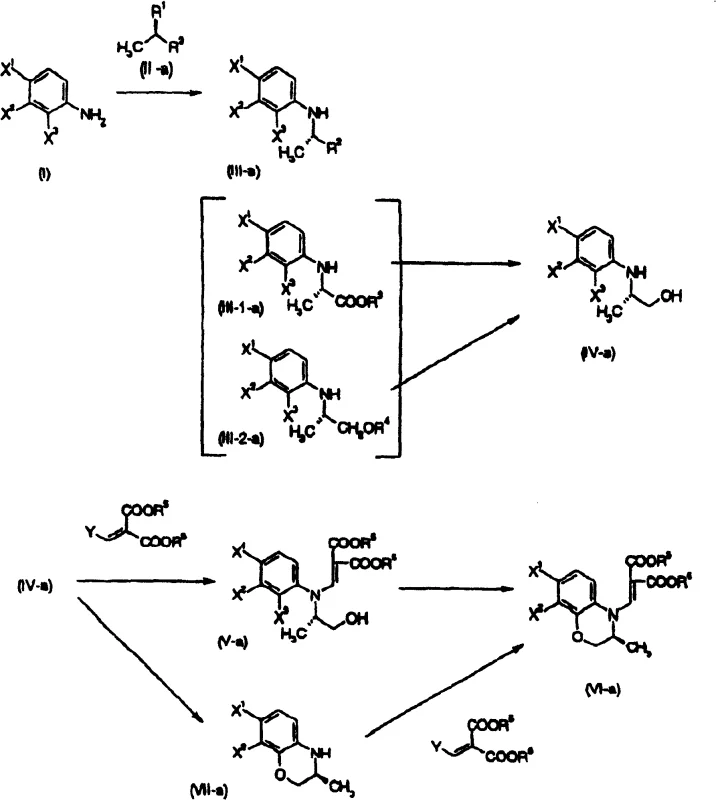
A key innovation in this novel approach is the flexibility to choose between chemical and enzymatic resolution strategies depending on the specific stereochemical requirements of the final product. The patent details the use of specific enzymes, such as lipases and proteases derived from various microbial sources, to achieve asymmetric hydrolysis of ester intermediates with exceptional enantioselectivity. This biocatalytic option provides a green chemistry alternative to traditional chemical resolution, operating under mild pH and temperature conditions that preserve the integrity of sensitive fluorine substituents on the aromatic ring. Furthermore, the ability to racemize the unwanted enantiomer and feed it back into the resolution loop ensures that the theoretical yield limitation of 50% is effectively overcome, maximizing atom economy and reducing raw material waste significantly.
Mechanistic Insights into Enzymatic and Chemical Resolution
The core of the stereocontrol mechanism in this patent relies on the precise interaction between the substrate and the chiral selector, whether it be a biological enzyme or a chiral organic base. In the enzymatic pathway, microorganisms such as Bacillus subtilis or Zygoascus hellenicus are employed to selectively hydrolyze one enantiomer of the ester intermediate while leaving the other intact. This kinetic resolution exploits the subtle steric differences between the (R) and (S) configurations at the chiral center adjacent to the carbonyl group. The enzyme's active site accommodates only one specific spatial arrangement, catalyzing the hydrolysis to the corresponding carboxylic acid which can then be separated from the unreacted ester via extraction due to their differing solubility profiles in aqueous and organic phases.
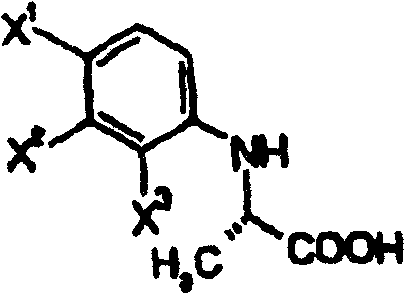
Alternatively, the chemical resolution pathway utilizes optically active organic bases, such as 1-phenylethylamine derivatives, to form diastereomeric salts with the racemic carboxylic acid intermediate. These salts exhibit different crystallization behaviors, allowing for the selective precipitation of the desired isomer through careful control of solvent composition and temperature. The patent emphasizes the importance of recrystallization to enhance the optical purity of the salt, often achieving levels exceeding 99% ee before the free acid is regenerated. This chemical method is particularly advantageous for large-scale operations where the cost and stability of enzymes might be a concern, providing a robust and reproducible means of securing high-quality chiral intermediates for subsequent cyclization steps.
How to Synthesize Benzoxazine Derivative Efficiently
The synthesis of these high-value intermediates involves a sequence of well-defined chemical transformations that begin with the condensation of a substituted aniline with a chiral lactate or pyruvate derivative. Following the initial bond formation, the process requires careful management of reaction parameters such as temperature, solvent polarity, and base strength to prevent racemization and ensure high conversion rates. The subsequent reduction and cyclization steps are critical for closing the oxazine ring, which forms the structural backbone of the final antibiotic molecule. Detailed standardized synthetic steps for implementing these transformations in a GMP-compliant environment are provided in the guide below.
- Condense trifluoroaniline derivatives with lactate esters or pyruvates under basic conditions to form the initial amino acid ester framework.
- Perform asymmetric resolution using specific enzymes like lipases or chiral organic bases to isolate the desired (S)-enantiomer with high optical purity.
- Execute intramolecular cyclization and subsequent condensation with ethoxymethylenemalonate to construct the final benzoxazine ring system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain professionals, the adoption of the synthesis methods described in CN100432060C offers tangible strategic benefits regarding cost structure and supply continuity. The process is designed to utilize commodity chemicals and widely available catalysts, reducing exposure to volatile pricing associated with specialized chiral reagents. By streamlining the synthesis into fewer steps and enabling the recycling of unwanted isomers, the overall material cost per kilogram of the active intermediate is substantially lowered. This efficiency translates directly into improved margin potential for downstream drug manufacturers and greater resilience against raw material shortages in the global pharmaceutical supply network.
- Cost Reduction in Manufacturing: The elimination of expensive asymmetric reducing agents and the ability to recycle racemized byproducts significantly lower the direct material costs associated with production. By avoiding complex microbial fermentation steps that require sterile conditions and lengthy incubation times, the process reduces utility consumption and facility overheads. The use of robust inorganic bases and phase transfer catalysts further simplifies the reaction workup, minimizing the volume of solvents and consumables required for purification. These cumulative efficiencies result in a more cost-effective manufacturing profile that enhances competitiveness in the generic antibiotic market.
- Enhanced Supply Chain Reliability: Diversifying the synthetic route to include both chemical and enzymatic options mitigates the risk of supply disruptions caused by reliance on a single technology platform. The starting materials, such as trifluoroanilines and malonate derivatives, are produced by multiple global suppliers, ensuring a stable and competitive sourcing environment. The scalability of the chemical resolution method allows for rapid ramp-up of production capacity to meet sudden spikes in demand without the lead time constraints typical of biological systems. This flexibility ensures consistent delivery schedules and strengthens partnerships with downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are easily managed in standard stainless steel reactors without requiring exotic pressure or temperature controls. The reduction in solvent usage and the ability to recover and reuse valuable chiral auxiliaries contribute to a lower environmental footprint, aligning with increasingly stringent global regulations on pharmaceutical waste. The avoidance of heavy metal catalysts simplifies the regulatory approval process for the intermediate, as residual metal testing becomes less critical. This compliance advantage accelerates time-to-market for new drug filings and reduces the administrative burden on quality assurance teams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. Understanding these details is crucial for R&D teams evaluating process transfer and procurement officers assessing supplier capabilities. The answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance.
Q: What are the primary advantages of the enzymatic resolution method described in CN100432060C?
A: The enzymatic resolution method offers superior stereoselectivity compared to traditional chemical resolution, often achieving optical purities exceeding 98% ee without the need for expensive chiral catalysts. It operates under mild conditions, reducing energy consumption and minimizing the degradation of sensitive functional groups during the separation of enantiomers.
Q: How does this process improve supply chain stability for levofloxacin production?
A: By utilizing readily available starting materials like trifluoroaniline and standard esterification reagents, the process reduces dependency on scarce chiral pool resources. The ability to racemize unwanted isomers and recycle them back into the process significantly enhances overall material efficiency and ensures consistent supply volumes.
Q: Is this synthesis route scalable for industrial commercial production?
A: Yes, the patent explicitly describes methods suitable for industrial application, including the use of phase transfer catalysts and robust inorganic bases that function effectively in large-scale reactors. The elimination of complex purification steps associated with microbial fermentation further simplifies the scale-up process for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazine Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom pharmaceutical manufacturing, leveraging deep expertise in complex organic synthesis to deliver high-quality intermediates like those described in CN100432060C. Our state-of-the-art facilities are equipped to handle diverse synthetic pathways, from enzymatic biocatalysis to advanced asymmetric chemical resolution, ensuring that we can meet the specific technical requirements of your project. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, maintaining stringent purity specifications through our rigorous QC labs. Our commitment to quality ensures that every batch of benzoxazine derivative meets the exacting standards required for the synthesis of potent fluoroquinolone antibiotics.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of these advanced synthetic methods. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your next project. Together, we can drive efficiency and innovation in the production of life-saving antimicrobial therapies.
