Advanced One-Step Cyanation for 2,3,4-Trimethoxy Benzonitrile Commercial Production
Advanced One-Step Cyanation for 2,3,4-Trimethoxy Benzonitrile Commercial Production
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex heterocyclic scaffolds, particularly for oncology and cardiovascular applications. A pivotal advancement in this domain is detailed in patent CN102603570A, which discloses a novel preparation method for 2,3,4-trimethoxy benzonitrile, a critical building block for various bioactive molecules. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot direct cyanation strategy. By leveraging a unique catalytic system comprising silver nitrate, iodine, and copper salts within an ionic liquid medium, the process achieves direct functionalization of 1,2,3-trimethoxybenzene. For R&D directors and procurement specialists, this innovation offers a compelling value proposition: it drastically simplifies the supply chain by reducing unit operations while simultaneously mitigating the environmental hazards associated with legacy halogenation protocols. The ability to produce high-purity pharmaceutical intermediates through such a robust and atom-economical route underscores the potential for significant cost optimization in large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,4-trimethoxy benzonitrile has been plagued by inefficiencies and safety concerns inherent to stepwise functionalization strategies. As illustrated in prior art, such as the work by Chen Xingquan, the conventional route typically initiates with pyrogallol, proceeding through methylation, followed by a hazardous bromination step using N-bromosuccinimide (NBS), and finally a cyanation with cuprous cyanide. 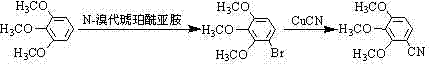 This multi-stage approach introduces substantial logistical burdens, as each intermediate requires isolation and purification, leading to cumulative yield losses and increased solvent consumption. Furthermore, the reliance on NBS introduces significant cost volatility, as this reagent is relatively expensive compared to bulk commodity chemicals. From a safety perspective, the generation of brominated byproducts and the handling of stoichiometric amounts of heavy metal cyanides pose severe occupational health risks and create complex waste streams that require costly treatment before disposal. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for high-purity pharmaceutical intermediates, making the conventional route suboptimal for modern, lean manufacturing environments.
This multi-stage approach introduces substantial logistical burdens, as each intermediate requires isolation and purification, leading to cumulative yield losses and increased solvent consumption. Furthermore, the reliance on NBS introduces significant cost volatility, as this reagent is relatively expensive compared to bulk commodity chemicals. From a safety perspective, the generation of brominated byproducts and the handling of stoichiometric amounts of heavy metal cyanides pose severe occupational health risks and create complex waste streams that require costly treatment before disposal. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for high-purity pharmaceutical intermediates, making the conventional route suboptimal for modern, lean manufacturing environments.
The Novel Approach
In stark contrast, the methodology disclosed in CN102603570A revolutionizes the synthesis by enabling a direct C-H activation and cyanation in a single reaction vessel. This novel approach utilizes 1,2,3-trimethoxybenzene, a commercially abundant and inexpensive feedstock, reacting it directly with a cyanide source in the presence of an iodine/copper/silver catalytic triad dissolved in an ionic liquid. 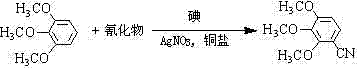 By eliminating the discrete bromination step, the process removes the need for expensive halogenating agents and the associated purification of bromo-intermediates. The reaction operates under sealed conditions at temperatures between 160-240°C, driving the equilibrium towards the desired nitrile product with reported yields reaching up to 72% in optimized examples. This consolidation of steps not only accelerates the overall throughput but also significantly reduces the physical footprint required for production. For supply chain managers, this translates to a more resilient production schedule with fewer points of failure, ensuring a consistent supply of this valuable intermediate for downstream API synthesis without the bottlenecks typical of linear, multi-step processes.
By eliminating the discrete bromination step, the process removes the need for expensive halogenating agents and the associated purification of bromo-intermediates. The reaction operates under sealed conditions at temperatures between 160-240°C, driving the equilibrium towards the desired nitrile product with reported yields reaching up to 72% in optimized examples. This consolidation of steps not only accelerates the overall throughput but also significantly reduces the physical footprint required for production. For supply chain managers, this translates to a more resilient production schedule with fewer points of failure, ensuring a consistent supply of this valuable intermediate for downstream API synthesis without the bottlenecks typical of linear, multi-step processes.
Mechanistic Insights into Ag/Cu-Catalyzed Direct Cyanation
The core of this technological breakthrough lies in the synergistic interaction between the silver catalyst, the copper co-catalyst, and the iodine promoter within the ionic liquid matrix. The mechanism likely proceeds via an electrophilic aromatic substitution or a radical-mediated pathway facilitated by the in situ generation of reactive iodine species. The silver nitrate acts as a potent Lewis acid or oxidant, activating the aromatic ring or the cyanide source, while the copper salt facilitates the transfer of the cyano group to the activated position on the benzene ring. The ionic liquid, specifically 1-n-butyl-3-methylimidazolium tetrafluoroborate, serves not merely as a solvent but as a stabilizing medium that enhances the solubility of inorganic salts and stabilizes charged intermediates, thereby lowering the activation energy of the rate-determining step. This unique microenvironment allows the reaction to proceed efficiently even with less toxic cyanide sources like potassium ferricyanide, which are typically inert under standard organic conditions. Understanding this mechanistic nuance is crucial for R&D teams aiming to replicate or scale this process, as the precise molar ratios—such as the optimal 1:0.5:0.8:0.5:0.8 ratio of substrate to cyanide to catalyst to iodine to copper—are critical for maximizing turnover numbers and minimizing side reactions.
Furthermore, the regioselectivity observed in this reaction is a testament to the electronic directing effects of the methoxy substituents combined with the steric constraints imposed by the catalytic complex. The methoxy groups at the 2 and 3 positions strongly activate the ring, directing the incoming cyano group predominantly to the 4-position, which is the least sterically hindered and most electron-rich site available. This high degree of selectivity is paramount for pharmaceutical applications, where isomeric impurities can be difficult to remove and may possess unwanted biological activity. The patent data indicates that by carefully controlling the reaction temperature, preferably around 180°C, and the reaction time (optimally 35 hours in specific examples), the formation of undesired isomers or over-cyanated byproducts is effectively suppressed. This intrinsic control over the impurity profile simplifies the downstream purification process, often allowing for high-purity isolation via simple vacuum distillation or column chromatography, thereby ensuring the final product meets the stringent specifications required for GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize 2,3,4-Trimethoxy Benzonitrile Efficiently
Implementing this synthesis requires precise adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process begins with the careful weighing and mixing of the solid reagents—1,2,3-trimethoxybenzene, the chosen cyanide source (with potassium ferricyanide being the preferred embodiment for safety and efficacy), silver nitrate, iodine, and a copper salt such as copper chloride. These components are introduced into a reaction vessel containing the ionic liquid, which must be thoroughly degassed or handled under an inert atmosphere if specified, although the patent notes operation under sealed conditions. The detailed standardized synthetic steps, including specific heating ramps, stirring rates, and work-up procedures necessary to achieve the reported 72% yield, are critical for successful technology transfer. Operators must ensure that the sealing of the reaction tube is robust to withstand the autogenous pressure generated at 160-240°C, as the integrity of the closed system is vital for maintaining the reaction kinetics and preventing the loss of volatile components. Following the reaction period, the cooling phase must be managed carefully to prevent thermal shock to the glassware or precipitation issues that could comp filtration.
- Dissolve 1,2,3-trimethoxybenzene, a cyanide source (preferably potassium ferricyanide), AgNO3 catalyst, Iodine, and a copper salt in an ionic liquid such as 1-n-butyl-3-methylimidazolium tetrafluoroborate.
- Seal the reaction vessel and heat the mixture to a temperature range of 160-240°C, maintaining these conditions for over 5 hours to ensure complete conversion.
- Upon completion, cool the system to room temperature and purify the resulting crude 2,3,4-trimethoxy benzonitrile using column chromatography or vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that extend far beyond simple yield improvements. The primary driver for cost reduction in pharma intermediates manufacturing here is the drastic simplification of the raw material portfolio. By replacing expensive, specialized reagents like N-bromosuccinimide and cuprous cyanide with commodity chemicals like iodine, copper chloride, and silver nitrate, the direct material costs are significantly lowered. Moreover, the elimination of intermediate isolation steps means that solvents, energy, and labor associated with multiple work-ups and drying processes are virtually eradicated. This streamlining results in a substantially reduced carbon footprint and lower waste disposal costs, aligning perfectly with modern sustainability goals and regulatory pressures. The ability to use cheaper starting materials like 1,2,3-trimethoxybenzene, which can itself be derived from low-cost pyrogallol, further anchors the cost structure, providing a buffer against market volatility in raw material pricing and ensuring long-term economic viability for high-volume production campaigns.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the convergence of several efficiency factors. By condensing a three-step linear synthesis into a single pot, the manufacturer eliminates the capital expenditure and operational overhead associated with multiple reactors and purification units. The removal of corrosive reagents like thionyl chloride, which requires specialized corrosion-resistant equipment and extensive scrubbing systems, further reduces CAPEX requirements. Additionally, the use of ionic liquids, which can potentially be recycled and reused in subsequent batches, minimizes the recurring cost of solvents. This holistic reduction in process complexity translates directly to a lower cost per kilogram of the final API intermediate, allowing pharmaceutical companies to improve their margins or pass savings on to healthcare systems without compromising on quality or supply security.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and stability of raw materials. Traditional routes relying on NBS or specific aldehyde precursors are often subject to supply disruptions due to the limited number of global manufacturers for these specialty chemicals. In contrast, the reagents required for this novel cyanation method—iodine, copper salts, and silver nitrate—are produced on a massive industrial scale for diverse applications, ensuring a robust and diversified supply base. This abundance mitigates the risk of stockouts and price spikes, guaranteeing continuous production flow. Furthermore, the simplified process flow reduces the lead time for high-purity pharmaceutical intermediates, as there are fewer quality control checkpoints and hold times between steps. This agility allows suppliers to respond more rapidly to fluctuating demand from downstream API manufacturers, fostering stronger partnerships and more reliable just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this ionic liquid-based approach is inherently designed for scalability. The homogeneous nature of the reaction mixture in the ionic liquid facilitates efficient heat and mass transfer, which are common challenges in batch processing. The absence of toxic heavy metal waste streams, such as those generated by lead thiocyanate methods mentioned in the background art, simplifies environmental compliance and permitting. The process generates significantly less hazardous waste, reducing the burden on wastewater treatment facilities and lowering the costs associated with environmental audits and remediation. This 'green chemistry' profile not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible partner in the pharmaceutical value chain, appealing to ESG-focused stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding the nuances of catalyst loading, temperature sensitivity, and purification methods is essential for both technical teams evaluating feasibility and commercial teams assessing risk.
Q: What are the primary advantages of this ionic liquid method over traditional bromination routes?
A: The patented ionic liquid method eliminates the need for expensive and hazardous reagents like N-bromosuccinimide (NBS) and cuprous cyanide used in traditional multi-step syntheses. It reduces the process from three distinct steps (methylation, bromination, cyanation) to a single direct cyanation step, significantly lowering operational complexity and waste generation.
Q: Which cyanide sources are compatible with this catalytic system?
A: The patent specifies several viable cyanide sources, including potassium ferricyanide, sodium cyanide, and potassium cyanide. Potassium ferricyanide is highlighted as the preferred non-toxic alternative, offering a safer handling profile compared to traditional alkali metal cyanides while maintaining high reaction efficiency.
Q: How does this process impact the scalability of API intermediate production?
A: By utilizing cheap and readily available 1,2,3-trimethoxybenzene as the starting material and avoiding corrosive reagents like thionyl chloride, the process offers superior scalability. The use of recyclable ionic liquids and simplified purification steps (distillation/chromatography) facilitates easier transition from laboratory scale to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,4-Trimethoxy Benzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the broader context of drug development and commercialization. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2,3,4-trimethoxy benzonitrile meets the exacting standards required for oncology and cardiovascular API synthesis. Our commitment to process excellence means we can leverage innovations like the ionic liquid cyanation method to deliver superior value to our partners, combining technical sophistication with commercial reliability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate the quality and consistency of our production capabilities firsthand. Let us collaborate to drive efficiency and innovation in your pharmaceutical manufacturing operations.
