Advanced Cetrorelix Synthesis via Novel MOBHA Resin for Commercial Scale-Up
The pharmaceutical landscape for gonadotropin-releasing hormone (GnRH) antagonists is undergoing a significant technical evolution, driven by the urgent need for higher purity intermediates and more sustainable manufacturing processes. Patent CN107778355B, published in April 2021, introduces a groundbreaking method for synthesizing cetrorelix that fundamentally restructures the solid-phase peptide synthesis (SPPS) workflow. This innovation addresses critical bottlenecks in the production of this potent ovulation inhibitor, specifically targeting the limitations of traditional resin carriers and cleavage reagents. By adopting a brand-new amino resin carrier known as MOBHA and utilizing protected D-Ornithine as a strategic precursor for D-Citrulline, the disclosed methodology achieves a total yield exceeding 60%, a substantial leap from the historical average of 40-45%. For R&D directors and procurement specialists seeking a reliable cetrorelix supplier, this patent represents a pivotal shift towards cost-effective, high-quality peptide manufacturing that minimizes toxic byproducts and maximizes process efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of cetrorelix has been plagued by inefficient yield profiles and the generation of hazardous impurities that complicate downstream purification. Prior art methods, such as those detailed in patents CN104086632A and others, typically rely on D-Orn(Dde) as the precursor for the critical D-Cit residue. The removal of the Dde protecting group in these conventional routes necessitates the use of hydrazine hydrate, a highly toxic and unstable reagent that poses severe safety risks and environmental compliance challenges for large-scale facilities. Furthermore, traditional cleavage protocols utilizing standard trifluoroacetic acid (TFA) solutions often fail to completely remove side-chain protecting groups, leading to the persistent formation of the toxic impurity [D-Cit(Ac)]-cetrorelix. These technical deficiencies not only cap the total yield at approximately 45% but also impose rigorous and costly purification burdens to meet stringent pharmacopeial standards for single impurities, which must remain below 0.15%.
The Novel Approach
In stark contrast, the novel approach delineated in CN107778355B leverages a structurally distinct carrier resin and a refined deprotection strategy to overcome these legacy hurdles. The core of this innovation lies in the substitution of expensive Rink Amide resins with the newly developed MOBHA resin, which offers superior cost-performance characteristics while maintaining excellent loading capacities. Instead of toxic hydrazine, the process utilizes standard Fmoc or Boc chemistry to protect the D-Ornithine side chain, which is subsequently removed under mild conditions before reacting with tert-butyl isocyanate to form the D-Cit(tBu) moiety directly on the resin. This strategic modification ensures that the subsequent acidolysis step, performed with a specialized TFA solution containing hydrogen bromide, effectively cleaves the peptide while simultaneously maximizing the removal of tBu groups, thereby virtually eliminating the risk of acetylated toxic impurities.
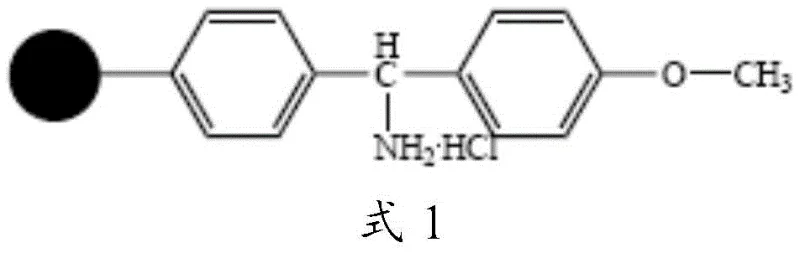
Mechanistic Insights into Solid-Phase Peptide Assembly and Acidolysis
The mechanistic superiority of this synthesis route is rooted in the precise control of side-chain reactivity during the elongation and cleavage phases. The process initiates with the stepwise coupling of protected amino acids onto the MOBHA resin, proceeding from the C-terminus to the N-terminus. A critical mechanistic divergence occurs at the position corresponding to D-Cit; here, a protected D-Orn residue is incorporated into the growing peptide chain. Following the completion of the backbone assembly and N-terminal acetylation, the side-chain protecting group (Boc or Fmoc) on the Ornithine residue is selectively removed. This exposes the delta-amino group, which then undergoes nucleophilic attack on tert-butyl isocyanate in the presence of an organic base like DIPEA. This on-resin transformation generates the D-Cit(tBu) unit in situ, ensuring that the urea linkage is formed with high fidelity before the final cleavage event, thus preventing racemization or incomplete conversion issues common in solution-phase modifications.
The final and perhaps most critical mechanistic step involves the acidolytic cleavage of the peptide from the resin using a trifluoroacetic acid solution fortified with hydrogen bromide (5-10% wt). In conventional TFA cleavages, the steric bulk of the t-butyl group on the Citrulline side chain can sometimes resist complete removal, or worse, participate in side reactions leading to acetylation if acetic acid scavengers are present in high concentrations. The inclusion of HBr acts as a potent scavenger and catalyst that facilitates the rapid and complete cleavage of the t-butyl ether bonds. This ensures that the final cetrorelix product emerges with the native Citrulline side chain fully deprotected, while the harsh acidic conditions simultaneously sever the peptide from the MOBHA linker. This dual-action mechanism is the key to achieving the reported crude purity of over 85% and avoiding the formation of the notorious [D-Cit(Ac)] impurity, streamlining the subsequent HPLC purification stages.
How to Synthesize Cetrorelix Efficiently
The implementation of this advanced synthesis protocol requires strict adherence to the optimized molar ratios and reaction times specified in the patent to ensure reproducibility at scale. The process is designed to be compatible with standard automated peptide synthesizers, utilizing common solvents like DMF and DCM, which facilitates easy technology transfer from laboratory to pilot plant. The following guide outlines the critical operational phases, emphasizing the specific reagents and conditions necessary to replicate the high-yield results documented in the intellectual property. For detailed technical parameters regarding reagent activation and washing cycles, please refer to the standardized synthesis steps provided below.
- Synthesize peptide resin 1 by coupling protected amino acids (replacing D-Cit with protected D-Orn) onto the novel MOBHA amino resin using condensation reagents.
- Remove the side-chain protecting group from D-Orn and react the free amino group with tert-butyl isocyanate to form D-Cit(tBu) on the resin.
- Perform acidolysis using a special trifluoroacetic acid solution containing hydrogen bromide to cleave the peptide and remove protecting groups, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates into tangible operational efficiencies and risk mitigation strategies that extend far beyond simple yield metrics. The elimination of hydrazine hydrate from the process flow removes a significant regulatory and safety burden, simplifying the storage, handling, and disposal of hazardous materials within the manufacturing facility. This reduction in hazardous reagent usage directly correlates to lower operational expenditures related to environmental compliance and worker safety protocols, making the supply chain more resilient against regulatory tightening. Additionally, the switch to the MOBHA resin, which is explicitly noted to be lower in cost than the industry-standard Rink Amide series, provides an immediate reduction in raw material costs for the solid support, a major cost driver in solid-phase peptide synthesis.
- Cost Reduction in Manufacturing: The documented increase in total yield from the industry baseline of approximately 45% to over 60% represents a massive improvement in raw material efficiency, meaning significantly less starting amino acid and resin are required to produce the same mass of final API. By avoiding the use of expensive and toxic hydrazine hydrate, the process eliminates the need for specialized neutralization and waste treatment procedures associated with hazardous nitrogenous compounds. Furthermore, the higher crude purity (>85%) reduces the load on preparative HPLC columns during purification, extending column life and reducing solvent consumption, which collectively drives down the cost of goods sold (COGS) for high-purity cetrorelix.
- Enhanced Supply Chain Reliability: The reliance on standard Fmoc/Boc protecting group chemistry and commercially available reagents like tert-butyl isocyanate ensures that the supply chain is not vulnerable to the shortages of exotic or highly regulated precursors. The robustness of the MOBHA resin and the mild reaction conditions contribute to a more predictable manufacturing timeline, reducing the risk of batch failures that can disrupt delivery schedules. This stability is crucial for maintaining continuous supply to pharmaceutical partners who require consistent volumes of GnRH antagonists for assisted reproductive technology treatments, ensuring that lead times for high-purity pharmaceutical intermediates remain competitive and reliable.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard solid-phase synthesis equipment and solvents that are already prevalent in GMP manufacturing facilities, allowing for seamless transition from kilogram to multi-ton production scales. The avoidance of toxic hydrazine and the minimization of difficult-to-remove impurities significantly lowers the environmental footprint of the manufacturing process, aligning with modern green chemistry principles and corporate sustainability goals. This environmental compatibility simplifies the permitting process for new production lines and ensures long-term operational continuity in regions with strict environmental regulations, securing the supply chain against future ecological compliance risks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of cetrorelix using the novel MOBHA resin methodology. These answers are derived directly from the experimental data and technical specifications outlined in patent CN107778355B, providing clarity on the process advantages and quality outcomes. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does the new MOBHA resin improve cetrorelix production costs?
A: The novel MOBHA resin utilized in this process is structurally optimized to be lower in cost compared to conventional Rink Amide series resins. Furthermore, the process eliminates the need for toxic hydrazine hydrate, reducing hazardous waste disposal expenses and simplifying safety protocols.
Q: What mechanism prevents the formation of toxic [D-Cit(Ac)]-cetrorelix impurities?
A: The process employs a specialized acidolysis step using a trifluoroacetic acid solution containing hydrogen bromide. This specific reagent combination maximizes the removal of tBu side-chain protecting groups from the generated D-Cit(tBu) intermediate, thereby preventing the acetylation side reactions that typically lead to toxic impurity formation in traditional methods.
Q: What purity and yield levels can be expected from this synthesis method?
A: According to the patent data, this method achieves a crude product purity of over 85% and a final pure product purity exceeding 99.5%. The total yield is significantly improved, reaching more than 60%, compared to the 40-45% typical of prior art methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cetrorelix Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a premier CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the MOBHA resin synthesis route are fully realized in your supply chain. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of cetrorelix meets the >99.5% purity benchmark with single impurities controlled below 0.15%, exactly as validated by the patent data.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for their GnRH antagonist programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-yield route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the highest standards of quality, efficiency, and supply security in the industry.
