Advanced Solid-Liquid Phase Synthesis of Alarelin Acetate for Commercial Scale-Up
Introduction to Advanced Peptide Manufacturing Technologies
The pharmaceutical industry continuously seeks robust manufacturing routes for complex peptide therapeutics, particularly for Gonadotropin-Releasing Hormone (GnRH) analogues like Alarelin Acetate. A pivotal development in this domain is documented in patent CN102702327A, which details a novel solid-liquid phase synthesis method designed to overcome the historical bottlenecks of low yield and severe reaction conditions. This technology represents a significant leap forward for any reliable API intermediate supplier aiming to optimize their production lines for high-value nonapeptides. By integrating the purification benefits of solid-phase synthesis with the precise control of solution-phase chemistry, this method addresses critical pain points in the manufacturing of hormone-dependent disease treatments.
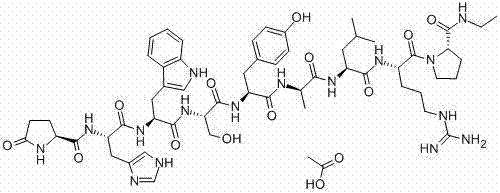
Alarelin Acetate, also known chemically as pGlu-His-Trp-Ser-Tyr-D-Ala-Leu-Arg-Pro-NHC2H5, is a potent synthetic analogue used in treating conditions such as endometriosis. The structural complexity of this molecule, featuring nine amino acid residues and a modified C-terminus, demands a synthesis strategy that ensures high stereochemical purity and minimal epimerization. The patented approach utilizes a fluorenylmethoxycarbonyl (Fmoc) strategy on a specialized proline-dichloro-trityl-chlorine resin, setting the stage for a highly efficient assembly line. This foundational shift allows manufacturers to achieve a total yield exceeding 40 percent, a metric that drastically alters the economic feasibility of large-scale production.
The implications of this technological breakthrough extend beyond mere yield improvements; they encompass a holistic enhancement of process safety and environmental compliance. Traditional liquid-phase syntheses often involve cumbersome isolation steps after each coupling, leading to substantial material loss and solvent waste. In contrast, the hybrid solid-liquid approach described in the patent streamlines the workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing. For R&D directors and procurement managers alike, understanding the nuances of this pathway is essential for securing a stable supply of high-purity peptides in an increasingly competitive market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex peptides like Alarelin Acetate has been plagued by inefficiencies inherent in purely liquid-phase or rigid solid-phase methodologies. In traditional liquid-phase synthesis, the necessity to isolate and purify the intermediate peptide fragments after every single amino acid coupling step results in a cumulative loss of material that can render the process economically unviable for commercial scale-up. Furthermore, the use of harsh reagents required for protection and deprotection in solution often leads to racemization, compromising the optical purity of the final active pharmaceutical ingredient. These severe reaction conditions not only increase the cost of goods sold but also generate significant hazardous waste, posing challenges for environmental compliance and sustainability goals.
On the other hand, conventional solid-phase peptide synthesis (SPPS), while simplifying purification through filtration, faces its own set of hurdles when dealing with C-terminal modifications such as ethylamination. Performing specific amidation reactions directly on the resin can sometimes suffer from incomplete conversion or difficult cleavage conditions that damage the sensitive peptide backbone. The accumulation of deletion sequences and the difficulty in monitoring reaction progress in the later stages of long peptide chains further exacerbate the problem. Consequently, finding a balance between the ease of solid-phase handling and the chemical precision of solution-phase reactions has been a longstanding challenge for process chemists aiming for commercial scale-up of complex polymer additives and peptide drugs.
The Novel Approach
The innovative method presented in patent CN102702327A elegantly resolves these dichotomies by employing a strategic "solid-liquid" hybrid workflow. The process initiates with the assembly of the peptide chain on a solid support, specifically using H-Pro-2Cl-Trt-Cl resin, which allows for the sequential coupling of Fmoc-protected amino acids under mild conditions. This solid-phase segment leverages the "wash-and-react" simplicity to build the bulk of the nonapeptide sequence with high fidelity, effectively minimizing the formation of deletion impurities that are common in solution phase. Once the full sequence is assembled on the resin, the strategy pivots to a solution-phase finish, which is the key differentiator of this technology.
By cleaving the fully protected peptide fragment (pGluP-9) from the resin using mild acidic conditions, the method preserves the side-chain protecting groups while freeing the C-terminus for specific modification. This allows the critical ethylamination step to occur in a homogeneous solution environment, where reaction kinetics can be tightly controlled to ensure quantitative conversion to the ethylamide derivative. This hybrid model effectively decouples the chain assembly from the terminal modification, optimizing both steps for maximum efficiency. For a reliable agrochemical intermediate supplier or pharma partner, this translates to a process that is not only easier to operate but also delivers a superior impurity profile, facilitating downstream purification and ensuring consistent batch-to-batch quality.
Mechanistic Insights into Fmoc Solid-Liquid Phase Synthesis
The core of this synthesis lies in the meticulous selection of the resin and the orthogonal protection strategy. The process begins with the swelling of H-Pro-2Cl-Trt-Cl resin, which serves as the anchor for the C-terminal proline. The trityl-based linker is acid-labile but stable to the basic conditions used for Fmoc deprotection (typically 20% piperidine in DMF). This orthogonality is crucial; it ensures that the peptide chain remains attached to the solid support during the repetitive cycles of deprotection and coupling, yet can be released cleanly when needed. The coupling reactions utilize potent activating agents such as HBTU, TBTU, or HATU in combination with HOBT or HOAt, which suppress racemization and drive the amide bond formation to completion. The use of bases like DIEA or NMM facilitates the activation of the carboxyl group, ensuring rapid acylation of the growing peptide chain.
A critical mechanistic advantage of this route is the "partial cleavage" step. Unlike standard SPPS where the final cleavage removes all protecting groups simultaneously, this method employs a very dilute trifluoroacetic acid (TFA) solution (1-5% in DCM) to cleave the peptide from the resin while leaving side-chain protectors like Boc, Trt, and Pbf intact. This generates the fully protected fragment pGluP-9 as a soluble species. In the subsequent solution phase, this fragment undergoes ethylamination using ethylamine hydrochloride and coupling reagents like PyBOP. The final step involves a global deprotection using a strong acid cocktail (e.g., TFA:Water:TIS), which removes all remaining side-chain groups to yield the crude Alarelin Acetate. This staged deprotection strategy minimizes side reactions and ensures that the delicate ethylamide bond formed in the previous step remains stable.
Impurity control is inherently built into this mechanism. By performing the bulk of the synthesis on a solid support, excess reagents and soluble byproducts are simply washed away, preventing them from participating in side reactions in subsequent steps. The transition to solution phase for the final modification allows for rigorous analytical monitoring (such as HPLC) before the final deprotection, ensuring that only the correctly modified intermediate proceeds to the final step. This level of control is vital for meeting the stringent purity specifications required for regulatory approval of peptide APIs. The result is a crude product with a purity profile that significantly reduces the burden on final purification columns, thereby increasing overall recovery and process throughput.
How to Synthesize Alarelin Acetate Efficiently
The synthesis of Alarelin Acetate via this patented solid-liquid phase method offers a streamlined pathway that balances operational simplicity with chemical precision. The process is divided into four distinct stages: resin loading and chain elongation, partial cleavage to obtain the protected fragment, solution-phase C-terminal modification, and final global deprotection. Each stage is optimized to maximize yield and minimize the generation of hazardous waste, making it suitable for industrial application. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for process implementation.
- Couple protected amino acids sequentially onto H-Pro-2Cl-Trt-Cl resin using Fmoc chemistry to form the side-chain protected peptide resin.
- Cleave the fully protected peptide chain from the resin using mild acid (1-5% TFA/DCM) to obtain the pGluP-9 fragment.
- Perform C-terminal ethylamination on the pGluP-9 fragment in solution phase using ethylamine hydrochloride and coupling agents like PyBOP.
- Remove all side-chain protecting groups using a TFA-based cocktail to yield the crude Alarelin Acetate peptide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solid-liquid phase synthesis method presents a compelling value proposition centered on cost efficiency and supply reliability. The primary driver of cost reduction is the significant improvement in overall yield, which the patent cites as exceeding 40 percent. In peptide manufacturing, where raw material costs for protected amino acids are high, even a marginal increase in yield translates into substantial savings in direct material costs. Furthermore, the elimination of multiple isolation and purification steps during the chain assembly phase reduces solvent consumption and labor hours, directly lowering the variable cost per kilogram of the final product.
Supply chain reliability is enhanced by the robustness of the Fmoc chemistry employed in this method. The reagents used, such as Fmoc-protected amino acids and standard coupling agents like HBTU and PyBOP, are commodity chemicals with stable global supply chains. This reduces the risk of production delays caused by the scarcity of exotic catalysts or specialized reagents. Additionally, the process operates under mild conditions (mostly at room temperature or slightly elevated temperatures like 25°C), which reduces the energy load on manufacturing facilities and minimizes the risk of thermal runaway incidents. This operational stability ensures consistent lead times and fosters a more predictable supply schedule for downstream customers.
From an environmental and scalability perspective, the method aligns well with modern green chemistry initiatives. The solid-phase portion of the synthesis drastically reduces the volume of solvent required for intermediate workups compared to traditional liquid-phase methods. The ability to scale this process from laboratory benchtop to multi-ton production is facilitated by the modular nature of solid-phase reactors, which can be easily expanded. The reduction in hazardous waste generation, particularly through the optimized deprotection cocktails, simplifies waste treatment protocols and lowers compliance costs. These factors collectively contribute to a more sustainable and economically viable manufacturing model for high-purity OLED material precursors and pharmaceutical intermediates alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the solid-liquid phase synthesis of Alarelin Acetate. These answers are derived directly from the technical specifications and experimental data provided in patent CN102702327A. They are intended to clarify the operational benefits and chemical rationale behind this advanced manufacturing route for stakeholders evaluating potential partnerships.
Q: What are the primary advantages of the solid-liquid phase synthesis method for Alarelin Acetate?
A: The method combines the purification ease of solid-phase synthesis with the flexibility of solution-phase modification, resulting in a total yield exceeding 40% and significantly reduced environmental pollution compared to traditional liquid-phase methods.
Q: Why is H-Pro-2Cl-Trt-Cl resin selected for this synthesis?
A: This specific resin allows for mild acid cleavage (using 1-5% TFA), which removes the peptide from the solid support while keeping the side-chain protecting groups intact, enabling subsequent solution-phase modifications.
Q: How is the C-terminal ethylamide group introduced in this process?
A: Unlike standard solid-phase methods, the C-terminal ethylamination is performed in the solution phase on the fully protected fragment pGluP-9 using ethylamine hydrochloride and coupling reagents such as PyBOP or HBTU.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alarelin Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to meet the evolving demands of the global pharmaceutical market. Our technical team has extensively analyzed the solid-liquid phase methodology described in recent patents and possesses the expertise to implement these advanced routes effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and compliant with international regulatory standards. Our facilities are equipped with state-of-the-art rigorous QC labs capable of monitoring every step of the peptide assembly to guarantee stringent purity specifications.
We invite procurement leaders and R&D directors to collaborate with us to leverage these process innovations for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this high-yield synthesis method. We encourage you to contact our technical procurement team to request specific COA data and comprehensive route feasibility assessments. Let us demonstrate how our commitment to technological excellence can drive value and reliability in your Alarelin Acetate supply chain.
