Advanced Synthesis of High-Purity Neurokinin-1 Receptor Antagonist Intermediates for Commercial Scale-Up
Introduction to Advanced Pharmaceutical Intermediate Synthesis
The development of efficient synthetic routes for complex heterocyclic compounds is a cornerstone of modern pharmaceutical manufacturing, particularly for intermediates used in the production of potent therapeutic agents. Patent CN1182125C discloses a novel and highly effective process for the preparation of (2R,2-alpha-R,3a)-2-[1-(3,5-bis(trifluoromethyl)phenyl)ethoxy]-3-(4-fluorophenyl)-1,4-oxazine, a critical intermediate in the synthesis of Substance P (neurokinin-1) receptor antagonists. These antagonists are invaluable for treating neurological disorders, inflammation, and emesis, representing a significant market sector in the global pharmaceutical industry. The technical breakthrough presented in this patent addresses long-standing challenges regarding yield stability and product purity that have plagued previous methodologies. By leveraging a streamlined sequence involving organometallic addition followed by catalytic hydrogenation, this invention offers a pathway that is not only chemically robust but also economically viable for large-scale production. For R&D directors and procurement specialists, understanding the nuances of this patented technology is essential for securing a reliable supply chain of high-quality active pharmaceutical ingredient (API) precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in US Patent No. 5,719,147, have historically suffered from significant drawbacks that hinder their utility in commercial manufacturing environments. The conventional synthesis of related oxazine derivatives often resulted in low and unstable yields of the target product, creating unpredictability in production planning and inventory management. These older processes frequently required harsh reaction conditions or multi-step sequences that accumulated impurities, necessitating complex and costly purification protocols to meet the stringent quality standards required for pharmaceutical intermediates. Furthermore, the instability of the yield meant that scaling up these reactions from laboratory benchtop to pilot plant often resulted in drastic drops in efficiency, leading to substantial material waste and increased cost of goods sold (COGS). The reliance on less efficient coupling strategies also limited the throughput capacity, making it difficult for suppliers to meet the growing demand for neurokinin-1 receptor antagonists without compromising on delivery timelines or product consistency.
The Novel Approach
In stark contrast to the limitations of the prior art, the methodology disclosed in CN1182125C introduces a remarkably practical and economical synthetic route that significantly enhances both yield and purity. The core innovation lies in the direct reaction of a benzyl-protected oxazinone precursor with a 4-fluorophenyl organometallic reagent, followed by a straightforward hydrogenation step. This approach eliminates several intermediate isolation steps, thereby reducing the overall processing time and minimizing the potential for product degradation. The use of readily available reagents, such as 4-fluorobromobenzene derived Grignard reagents, ensures that the supply chain remains resilient and cost-effective. By optimizing solvent systems, specifically utilizing tetrahydrofuran (THF) for the Grignard addition, the process achieves superior reaction kinetics and selectivity. The subsequent hydrogenation under mild conditions further simplifies the workflow, allowing for the direct formation of the desired oxazine structure with high fidelity. This novel approach represents a paradigm shift in how these complex intermediates are manufactured, offering a scalable solution that aligns perfectly with the needs of modern cGMP facilities.
Mechanistic Insights into Grignard Addition and Catalytic Hydrogenation
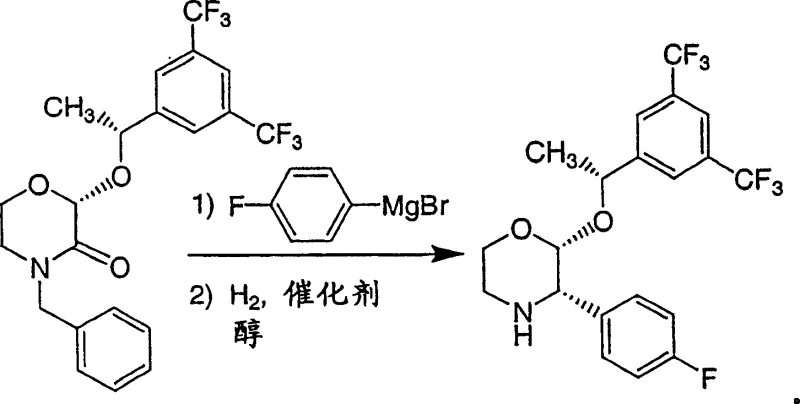
The chemical elegance of this synthesis is rooted in the precise control of reaction parameters during the organometallic addition phase. The reaction between the (2R,2-alpha-R)-4-benzyl-2-[1-(3,5-bis(trifluoromethyl)phenyl)ethoxy]-1,4-oxazin-3-one and the 4-fluorophenyl Grignard reagent is highly sensitive to temperature and solvent choice. The patent specifies a broad operational window from approximately -70°C to +70°C, with a preferred range of 20°C to 25°C, which is advantageous for industrial applications as it avoids the energy costs associated with cryogenic cooling. The mechanism involves the nucleophilic attack of the Grignard reagent on the carbonyl carbon of the oxazinone ring, forming a transient hemiaminal or alkoxide intermediate. The choice of THF as the primary solvent is critical here, as it effectively solvates the magnesium cation, enhancing the nucleophilicity of the organometallic species while maintaining the stability of the sensitive fluorinated aromatic rings. This step is pivotal for establishing the correct regiochemistry, ensuring that the 4-fluorophenyl group is installed at the 3-position of the oxazine ring without disrupting the existing chiral centers.
Following the addition, the process employs a catalytic hydrogenation step that serves a dual purpose: removing the benzyl protecting group and stabilizing the newly formed structure. The use of palladium catalysts, particularly palladium-on-carbon (Pd/C), in a methanol solvent system facilitates the cleavage of the benzyl group under relatively low hydrogen pressures (5-50 psi). This mild condition is crucial for preserving the stereochemical integrity of the molecule, preventing racemization which could occur under more vigorous acidic or thermal conditions. The mechanistic pathway likely involves the adsorption of the benzyl group onto the palladium surface, followed by hydrogenolysis to release toluene and the free amine. The presence of an acid, such as p-toluenesulfonic acid or hydrochloric acid, during or prior to this step aids in the protonation of the amine, driving the equilibrium towards the salt form which often crystallizes more readily. This combination of nucleophilic addition and catalytic deprotection creates a highly efficient cascade that minimizes side reactions and maximizes the recovery of the pharmacologically active stereoisomer.
How to Synthesize (2R,2-alpha-R,3a)-2-[1-(3,5-bis(trifluoromethyl)phenyl)ethoxy]-3-(4-fluorophenyl)-1,4-oxazine Efficiently
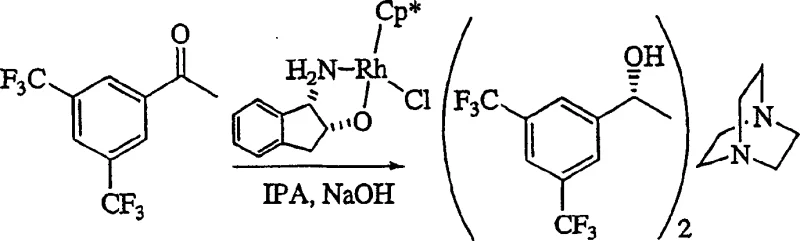
Executing this synthesis requires strict adherence to the optimized conditions outlined in the patent to ensure reproducibility and high quality. The process begins with the preparation of the chiral alcohol precursor, often achieved via asymmetric transfer hydrogenation using rhodium catalysts, which sets the absolute stereochemistry early in the sequence. Once the protected oxazinone starting material is secured, the key transformation involves the controlled addition of the Grignard reagent. Operators must monitor the exotherm carefully, although the preferred ambient temperature range mitigates extreme thermal spikes. After the addition is complete and the intermediate is formed, the reaction mixture is typically quenched with an alcohol and acid before being subjected to hydrogenation. The final isolation involves crystallization, potentially as a hydrochloride salt, which offers superior thermal stability and handling characteristics compared to the free base. Detailed standardized operating procedures for each unit operation are essential for maintaining batch-to-batch consistency.
- React (2R,2-alpha-R)-4-benzyl-2-[1-(3,5-bis(trifluoromethyl)phenyl)ethoxy]-1,4-oxazin-3-one with 4-fluorophenyl organometallic reagent (e.g., Grignard) in THF at controlled temperatures.
- Quench the reaction mixture with acid and alcohol, then proceed immediately to catalytic hydrogenation using Pd/C in methanol.
- Isolate the final product via crystallization, optionally forming stable hydrochloride salts for improved handling and purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the process described in CN1182125C offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the significant reduction of manufacturing complexity, which directly translates to lower operational costs and reduced risk of supply disruption. By utilizing common, commercially available starting materials like 4-fluorobromobenzene and standard catalysts like Pd/C, the process avoids reliance on exotic or single-source reagents that can create bottlenecks. The robustness of the reaction conditions, particularly the ability to run the Grignard addition at near-ambient temperatures, reduces the energy footprint of the manufacturing process and lowers the barrier for scale-up in existing facilities. Furthermore, the high purity of the crude product minimizes the need for extensive chromatographic purification, allowing for simpler work-up procedures like filtration and crystallization. This streamlining of the downstream processing significantly shortens the production cycle time, enabling faster turnaround for customer orders and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The economic viability of this synthetic route is driven by the elimination of expensive transition metal removal steps and the use of cost-effective solvents. Unlike processes that require precious metal scavengers or complex distillation trains, this method relies on standard filtration to remove the heterogeneous palladium catalyst. The high yield reported in the examples implies a substantial reduction in raw material consumption per kilogram of final product, directly impacting the variable cost structure. Additionally, the ability to isolate the product as a stable salt reduces packaging and storage costs associated with unstable free bases. The overall simplicity of the workflow means that labor hours per batch are minimized, contributing to a leaner manufacturing model that can withstand price pressures in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are widely produced and stocked by multiple global vendors. The avoidance of specialized, custom-synthesized reagents reduces the lead time for raw material procurement. The process tolerance to slight variations in reaction conditions, as evidenced by the broad temperature ranges provided in the patent, ensures that manufacturing campaigns are less likely to fail due to minor process deviations. This reliability is critical for maintaining continuous supply to downstream API manufacturers who operate on just-in-time inventory models. Moreover, the scalability of the hydrogenation step means that production volume can be ramped up quickly to meet surges in demand without requiring capital-intensive equipment upgrades, providing a flexible response capability to market dynamics.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with green chemistry principles. The use of methanol and THF, while requiring proper handling, allows for efficient solvent recovery and recycling systems to be implemented. The low-pressure hydrogenation reduces the safety risks associated with high-pressure gas handling, simplifying the regulatory compliance burden for the manufacturing site. The high atom economy of the addition reaction minimizes waste generation, and the crystallization-based purification reduces the volume of organic waste streams compared to chromatography-heavy processes. These factors collectively contribute to a more sustainable manufacturing profile, which is increasingly becoming a key criterion for selection by major pharmaceutical companies aiming to reduce their Scope 3 emissions and meet corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their broader drug development pipelines. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the critical impurities controlled in this synthesis?
A: The process specifically controls diastereomeric impurities through careful temperature regulation during the Grignard addition and utilizes crystallization to remove residual starting materials and byproducts.
Q: Is the hydrogenation step scalable for industrial production?
A: Yes, the hydrogenation operates at low pressures (5-50 psi) using standard Pd/C catalysts in methanol, making it highly suitable for large-scale reactor operations without requiring specialized high-pressure equipment.
Q: How is the stereochemical integrity maintained?
A: Stereochemistry is established early via asymmetric reduction of the ketone precursor using Rhodium catalysts, ensuring high enantiomeric excess (>99% ee) is carried through to the final oxazine intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2R,2-alpha-R,3a)-2-[1-(3,5-bis(trifluoromethyl)phenyl)ethoxy]-3-(4-fluorophenyl)-1,4-oxazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your project from clinical trial phases through to full market launch. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific requirements of fluorinated chemistry and asymmetric synthesis, providing a secure and compliant environment for the manufacture of complex pharmaceutical intermediates like the neurokinin-1 antagonist precursor discussed herein.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall project costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us be your trusted partner in navigating the complexities of pharmaceutical supply chains, delivering excellence in every kilogram.
