Advanced Catalytic Hydrogenation for Commercial Tetrahydropyran-4-one Production
The chemical landscape for heterocyclic ketones is undergoing a significant transformation driven by the innovations disclosed in patent CN1898229A, which outlines a robust preparation method for tetrahydropyran-4-one and pyran-4-one. This technology addresses critical bottlenecks in the synthesis of these valuable compounds, which serve as essential building blocks for a wide array of pharmaceutical and agrochemical active ingredients. Traditional manufacturing pathways have long been plagued by operational complexities, including the necessity of isolating unstable intermediate salts and utilizing hazardous reagents that complicate safety protocols and increase waste generation. The novel approach detailed in this patent leverages a streamlined cyclization reaction followed by a highly efficient catalytic hydrogenation step, offering a pathway that is not only chemically elegant but also industrially viable for large-scale production. By integrating this methodology, manufacturers can achieve superior yield consistency while mitigating the environmental and safety risks associated with legacy processes, positioning this technology as a cornerstone for modern fine chemical synthesis strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
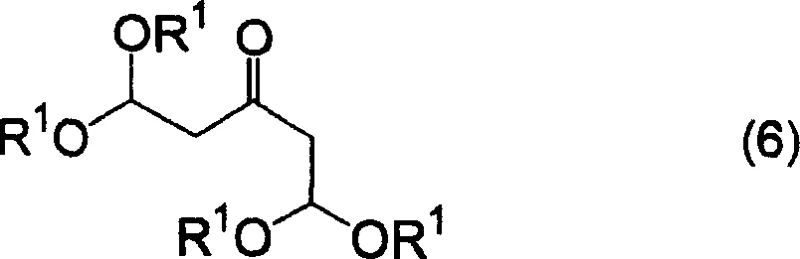
Historically, the industrial preparation of pyran-4-one derivatives has relied on cumbersome multi-step sequences that involve the precipitation of sodium salts of formyl derivatives as key intermediates. These conventional routes often necessitate the use of methanol solutions of hydrogen chloride, which are notoriously difficult to handle due to their corrosive nature and the challenges associated with precise stoichiometric control during neutralization. Furthermore, the reaction times associated with these legacy methods are excessively long, often requiring overnight standing periods in concentrated acidic conditions to drive the cyclization to completion, which severely limits throughput capacity in a commercial setting. The need to isolate and purify intermediate salts adds significant unit operations to the process flow, increasing both capital expenditure on equipment and operational expenditure on labor and energy consumption. Additionally, the variability in yield, often hovering around modest percentages in prior art, creates unpredictability in supply planning and complicates the economic modeling required for viable commercial production of these high-value intermediates.
The Novel Approach
In stark contrast to the convoluted legacy pathways, the method disclosed in CN1898229A introduces a direct and efficient cyclization strategy utilizing 1,1,5,5-tetraalkoxypentan-3-one or its equivalents as the starting material. This innovative route bypasses the need for isolating unstable sodium salts, allowing the reaction to proceed directly to the crude pyran-4-one product through a simplified acid-catalyzed mechanism. The process is characterized by its operational simplicity, where the cyclization can be conducted under controlled temperature conditions that significantly reduce the overall reaction time compared to the overnight protocols of the past. By eliminating the intermediate isolation steps, the novel approach reduces the number of unit operations, thereby minimizing material loss and solvent consumption throughout the manufacturing campaign. This streamlining not only enhances the overall mass balance of the process but also facilitates a more continuous manufacturing flow, which is essential for meeting the rigorous demand schedules of downstream pharmaceutical clients who require reliable and consistent supply chains for their drug development programs.
Mechanistic Insights into Acid-Catalyzed Cyclization and Hydrogenation

The core of this technological advancement lies in the precise mechanistic control of the cyclization and subsequent reduction steps, which are optimized to maximize conversion while minimizing byproduct formation. The cyclization of the tetraalkoxy precursor involves an acid-catalyzed intramolecular condensation that efficiently closes the pyran ring, a transformation that is highly sensitive to the choice of acid and solvent system. The patent specifies the use of inorganic acids such as hydrochloric or sulfuric acid, which provide the necessary proton source to activate the carbonyl and alkoxy groups for nucleophilic attack without introducing extraneous impurities that could complicate downstream purification. Following the formation of the pyran-4-one scaffold, the process employs a sophisticated catalytic hydrogenation step using metal catalysts such as palladium on carbon or platinum, which selectively reduce the unsaturated bonds within the ring structure. This reduction is conducted in a carefully balanced solvent system, often comprising a mixture of aprotic solvents like toluene and alcohol solvents like ethanol, which ensures optimal solubility of the substrate and efficient mass transfer of hydrogen gas to the catalyst surface.
Impurity control is a critical aspect of this synthesis, particularly given the sensitivity of the pyranone ring to over-reduction or polymerization under harsh conditions. The patented method addresses this by strictly controlling the reaction temperature and pressure during the hydrogenation phase, typically maintaining conditions between 0 to 100°C and 0.1 to 10 MPa to prevent the formation of fully saturated side products or ring-opened degradation species. The use of anhydrous metal catalysts, obtained through azeotropic dehydration with hydrophobic organic solvents, further enhances the selectivity of the reaction by removing water that could otherwise poison the catalyst or promote hydrolysis of the sensitive intermediates. This meticulous attention to reaction parameters ensures that the final tetrahydropyran-4-one product meets stringent purity specifications, with the patent examples demonstrating reaction yields as high as 97% for the cyclization step and over 85% for the hydrogenation step. Such high efficiency is indicative of a robust process capable of delivering consistent quality, which is paramount for regulatory compliance in the pharmaceutical industry where impurity profiles must be tightly controlled and documented.
How to Synthesize Tetrahydropyran-4-one Efficiently
The practical implementation of this synthesis route requires a clear understanding of the sequential operations involved, starting from the preparation of the tetraalkoxy precursor to the final isolation of the saturated ketone. The process begins with the condensation of 1,1-dialkoxybutan-3-one with a formate ester in the presence of a base to generate the necessary salt intermediate, which is then subjected to acid hydrolysis to form the pyranone ring. This initial phase sets the foundation for the entire synthesis, as the quality of the crude pyran-4-one directly influences the efficiency of the subsequent hydrogenation step. Operators must ensure precise control over the addition rates of acids and bases to manage the exothermic nature of these reactions, thereby maintaining safety and product integrity throughout the batch cycle. The detailed standardized synthesis steps provided below outline the specific parameters for solvent volumes, catalyst loading, and reaction times that have been validated to achieve optimal results.
- Prepare 1,1,5,5-tetraalkoxypentan-3-one or its equivalent as the cyclization precursor in an organic solvent.
- Perform acid-catalyzed cyclization using inorganic acids like hydrochloric acid to generate crude pyran-4-one.
- Execute catalytic hydrogenation using Pd/C or Pt catalysts in a mixed solvent system to yield tetrahydropyran-4-one.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers substantial advantages for procurement managers and supply chain directors who are tasked with optimizing costs and ensuring material availability. The elimination of complex intermediate isolation steps translates directly into reduced processing time and lower utility consumption, which are key drivers in the overall cost of goods sold for fine chemical intermediates. By simplifying the workflow, manufacturers can achieve higher throughput rates without the need for significant capital investment in new equipment, allowing for a more agile response to market demand fluctuations. Furthermore, the use of readily available raw materials such as simple alkoxy ketones and common metal catalysts reduces the risk of supply chain disruptions that are often associated with specialized or proprietary reagents. This reliability is crucial for maintaining continuous production schedules and avoiding the costly delays that can occur when waiting for scarce starting materials to be delivered from distant suppliers.
- Cost Reduction in Manufacturing: The streamlined nature of this process significantly lowers manufacturing costs by removing the need for expensive and hazardous reagents like methanol solutions of hydrogen chloride, which require specialized handling and disposal protocols. The reduction in unit operations means less solvent is consumed and less waste is generated, leading to substantial savings in waste treatment and environmental compliance costs. Additionally, the high yields achieved in both the cyclization and hydrogenation steps maximize the utilization of raw materials, ensuring that a greater proportion of the input mass is converted into saleable product rather than being lost as byproducts. This efficiency gain allows for a more competitive pricing structure, enabling suppliers to offer high-purity intermediates at a price point that supports the economic viability of downstream drug development projects without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard catalytic hydrogenation equipment enhances the robustness of the supply chain, as these resources are widely available from multiple global vendors. This diversification of supply sources mitigates the risk of single-source dependency, which is a common vulnerability in the pharmaceutical supply chain that can lead to production stoppages. The simplified process flow also reduces the likelihood of operational failures or batch rejections, as there are fewer critical control points where errors can occur during manufacturing. Consequently, suppliers can provide more accurate lead time estimates and maintain higher inventory levels of finished goods, ensuring that customers receive their orders on schedule and can plan their own production activities with greater confidence and certainty.
- Scalability and Environmental Compliance: The process is inherently scalable, as the reaction conditions do not require extreme pressures or temperatures that would necessitate specialized high-pressure reactors or cryogenic cooling systems. This ease of scale-up allows manufacturers to transition smoothly from pilot plant quantities to multi-ton commercial production without encountering the engineering bottlenecks that often plague complex synthetic routes. Moreover, the reduction in hazardous waste and the use of less toxic solvents align with increasingly stringent environmental regulations, reducing the regulatory burden on the manufacturing facility. This compliance not only avoids potential fines and shutdowns but also enhances the corporate sustainability profile, which is becoming an important factor for pharmaceutical companies when selecting long-term manufacturing partners who share their commitment to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity on its operational feasibility and strategic value. These answers are derived directly from the technical specifications and experimental data presented in the patent documentation, ensuring that the information provided is accurate and relevant to industry professionals. Understanding these details is essential for making informed decisions about integrating this method into existing manufacturing portfolios or sourcing strategies. The insights provided here aim to bridge the gap between theoretical chemical potential and practical industrial application, offering a comprehensive view of what stakeholders can expect.
Q: What are the primary advantages of this new synthesis method over conventional routes?
A: The patented method eliminates the need for precipitating sodium salts of formyl derivatives and avoids handling difficult methanol solutions of hydrogen chloride, resulting in simplified operations and significantly reduced reaction times compared to traditional multi-step processes.
Q: Which catalysts are most effective for the hydrogenation step in this process?
A: The process utilizes metal catalysts containing palladium, platinum, or nickel, such as palladium on carbon or Raney nickel, which allow for high conversion rates under moderate temperature and pressure conditions without requiring complex activation procedures.
Q: How does this method impact the purity profile of the final tetrahydropyran-4-one?
A: By avoiding the formation of difficult-to-remove intermediate salts and utilizing efficient azeotropic dehydration techniques, the method ensures a cleaner crude product that requires less rigorous purification, thereby maintaining high purity specifications suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydropyran-4-one Supplier
The technical potential of this synthesis route is immense, offering a pathway to high-purity heterocyclic ketones that are critical for the development of next-generation therapeutics. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facility is equipped with state-of-the-art hydrogenation reactors and rigorous QC labs that ensure stringent purity specifications are met for every batch produced. We understand the critical nature of pharmaceutical intermediates and the need for absolute consistency in quality, which is why we have invested heavily in process analytical technology to monitor reaction progress in real-time. This commitment to excellence ensures that our clients receive materials that are not only chemically pure but also fully compliant with the regulatory standards required for global drug registration and commercialization.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages of switching to this more efficient manufacturing method. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this approach for your projects. Our team is ready to collaborate with you to tailor the production parameters to your exact needs, ensuring a seamless integration of these high-quality intermediates into your drug development pipeline. Let us partner with you to accelerate your timeline and reduce your costs through superior chemical manufacturing expertise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
