Revolutionizing Adiponitrile Synthesis: Advanced Ni(0) Catalyst Systems for Industrial Scale
The chemical industry's relentless pursuit of efficient nylon 6,6 precursors has found a significant breakthrough in patent CN1293942C, which details a novel Ni(0)-containing catalyst system for hydrocyanation processes. This technology fundamentally shifts the paradigm of catalyst preparation by utilizing readily available metallic nickel powder instead of expensive, unstable organometallic precursors like bis(1,5-cyclooctadiene)nickel(0). For R&D directors and process engineers, this represents a critical opportunity to optimize the synthesis of adiponitrile, the vital intermediate for hexamethylenediamine production. The patent outlines a sophisticated ligand architecture involving both monodentate and bidentate phosphites that work in synergy to stabilize the active nickel species, ensuring high selectivity and prolonged catalyst life even under the harsh conditions required for industrial hydrocyanation.
Traditionally, the hydrocyanation of 1,3-butadiene to adiponitrile has relied on catalyst systems that are notoriously difficult to prepare and maintain. Conventional methods often involve the use of Ni(COD)2, which is not only costly to synthesize but also prone to rapid decomposition upon exposure to air or moisture, complicating storage and handling logistics. Furthermore, older techniques that utilize nickel chloride reduced by zinc generate stoichiometric amounts of zinc chloride waste, creating significant downstream purification burdens and environmental disposal costs. These legacy processes often suffer from inconsistent catalyst activity, leading to variable product quality and the frequent need for fresh catalyst charges, which disrupts continuous production flows and inflates operational expenditures.
In stark contrast, the novel approach described in the patent leverages the in situ generation of the active Ni(0) species from high-surface-area metallic nickel powder. By reacting this commodity metal with specific phosphite ligands in a liquid diluent, the system creates a highly active catalytic complex without the need for pre-isolated organometallic intermediates. This method drastically simplifies the supply chain for catalyst precursors, as metallic nickel powder is a standard industrial commodity with stable pricing and availability. The integration of bidentate phosphite ligands further enhances the system's robustness, preventing the formation of inactive nickel cyanide sludge that typically plagues hydrocyanation reactions, thereby ensuring a cleaner reaction profile and higher overall yield of the desired linear dinitrile products.
Mechanistic Insights into Ni(0)-Phosphite Catalyzed Hydrocyanation
The core of this technological advancement lies in the precise coordination chemistry between the nickel center and the phosphite ligands. The catalyst system comprises Ni(0) coordinated with 4 to 10 moles of a monodentate phosphite (Compound I) and 1 to 4 moles of a bidentate phosphite (Compound II) per mole of nickel. The monodentate ligands, typically aryl phosphites such as tri-o-tolyl phosphite, facilitate the initial activation of the metallic surface and help solubilize the nickel species. Meanwhile, the bidentate ligands, which feature a bridging group Y connecting two phosphite units, act as chelating agents that lock the nickel into a stable geometric configuration. This chelation effect is crucial for preventing the dissociation of ligands under reaction conditions, which would otherwise leave the nickel vulnerable to irreversible poisoning by cyanide ions.
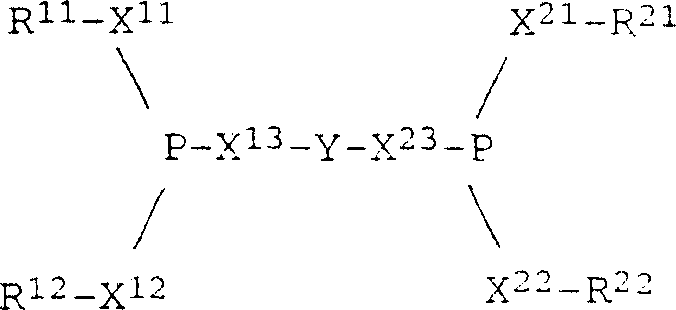
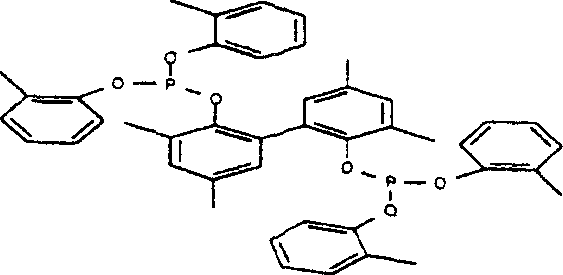
The structural integrity of the bidentate ligand is paramount for maintaining catalytic turnover. As illustrated in the general formula, the bridging group Y can be varied to tune the bite angle and electronic properties of the ligand, allowing for fine-tuning of the catalyst's selectivity towards linear versus branched nitriles. Preferred embodiments utilize bridging groups derived from biphenols or dinaphthols, which provide a rigid backbone that enforces a specific coordination geometry around the nickel atom. This rigidity is essential for discriminating between the various isomers of pentenenitrile formed during the reaction, steering the equilibrium towards the linear 3-pentenenitrile which is the direct precursor to adiponitrile. The synergy between the electron-donating properties of the phosphites and the steric bulk of the aryl substituents creates an electronic environment that favors oxidative addition of HCN and reductive elimination of the nitrile product.
How to Synthesize Adiponitrile Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for implementing this advanced catalyst system in a production setting. The process begins with the preparation of the active catalyst mixture by stirring metallic nickel powder with the monodentate phosphite ligand in a suitable solvent such as toluene or 3-pentenenitrile itself. Once the initial complex is formed, the bidentate ligand is introduced to complete the coordination sphere. This pre-formed catalyst solution is then charged into a reactor along with the 1,3-butadiene feedstock. The reaction is typically conducted at temperatures between 50°C and 150°C and pressures ranging from 1 to 100 bar, conditions that are well within the capabilities of standard high-pressure industrial reactors. Detailed standardized synthesis steps follow below.
- Preparation of the active catalyst system by reacting high-surface-area metallic nickel powder with monodentate phosphite ligands (Compound I) in a liquid diluent.
- Addition of bidentate phosphite ligands (Compound II) to the mixture to stabilize the Ni(0) center and enhance selectivity towards linear nitriles.
- Introduction of 1,3-butadiene and hydrogen cyanide under controlled temperature (50-150°C) and pressure conditions to effect hydrocyanation and isomerization to adiponitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Ni(0) catalyst system offers profound strategic benefits centered around cost stability and operational reliability. The most immediate impact is the elimination of dependency on specialized organometallic suppliers for Ni(COD)2, replacing it with widely available metallic nickel powder. This shift not only reduces the raw material cost of the catalyst significantly but also mitigates the risk of supply disruptions associated with niche chemical intermediates. Furthermore, the enhanced stability of the catalyst means that fewer catalyst replenishment cycles are required, reducing the frequency of reactor shutdowns for maintenance and catalyst exchange. This leads to higher plant utilization rates and a more consistent output of high-purity adiponitrile, which is critical for meeting the stringent quality specifications of downstream polymer manufacturers.
- Cost Reduction in Manufacturing: The transition to metallic nickel powder as the nickel source eliminates the premium pricing associated with pre-complexed organometallic nickel compounds. Additionally, the improved selectivity of the catalyst system minimizes the formation of by-products such as 2-methylglutaronitrile, reducing the load on downstream separation columns and lowering energy consumption for purification. The absence of zinc chloride waste generation also removes the costs associated with waste treatment and disposal, contributing to a leaner and more environmentally compliant manufacturing process that aligns with modern sustainability goals.
- Enhanced Supply Chain Reliability: By utilizing commodity-grade metallic nickel and commercially synthesized phosphite ligands, the supply chain becomes far more resilient to market volatility. The robust nature of the catalyst system allows for longer campaign lengths without significant loss of activity, ensuring a steady flow of product to customers. This reliability is particularly valuable for reliable polymer intermediate supplier operations where consistent delivery schedules are paramount for maintaining just-in-time inventory levels for nylon producers. The ability to tolerate slight variations in feedstock quality further buffers the process against upstream supply fluctuations.
- Scalability and Environmental Compliance: The homogeneous nature of the catalyst system facilitates easy scale-up from pilot plants to full commercial production without the mass transfer limitations often encountered with heterogeneous catalysts. The process operates efficiently in continuous or semi-continuous modes, maximizing throughput. Moreover, the reduction in hazardous waste generation, specifically the avoidance of heavy metal salts like zinc chloride, simplifies regulatory compliance and reduces the environmental footprint of the facility. This aligns with global trends towards greener chemistry and can potentially qualify the production site for environmental incentives or preferred vendor status with eco-conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this hydrocyanation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing production infrastructure. Understanding these nuances is essential for making informed decisions about process adoption and capital investment.
Q: What is the primary advantage of using metallic nickel powder over Ni(COD)2 in this catalyst system?
A: Metallic nickel powder is significantly cheaper and more commercially available than complex organometallic precursors like Ni(COD)2. Furthermore, the patent demonstrates that generating the active Ni(0) species in situ from powder avoids the decomposition issues associated with pre-formed complexes, leading to a more robust and longer-lasting catalyst lifecycle.
Q: How does the bidentate ligand (Compound II) improve catalyst performance?
A: The bidentate phosphite ligand acts as a chelating agent that stabilizes the low-valent nickel center against oxidation and poisoning by cyanide ions. This structural reinforcement prevents the formation of inactive nickel cyanide precipitates, thereby maintaining high catalytic activity and selectivity for adiponitrile over extended reaction periods.
Q: Can this catalyst system handle crude 1,3-butadiene streams?
A: Yes, the system is designed to tolerate hydrocarbon mixtures containing 1,3-butadiene. However, the patent suggests that removing catalyst poisons such as alkynes and vinylacetylene via partial hydrogenation prior to the reaction is beneficial to maximize conversion efficiency and prevent premature catalyst deactivation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adiponitrile Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Ni(0)-phosphite catalyst system described in CN1293942C for the global nylon industry. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for both the catalyst ligands and the final adiponitrile product. We understand that consistency is key in polymer intermediate manufacturing, and our quality management systems are designed to deliver batch-after-batch reliability that meets the exacting standards of international pharmaceutical and chemical corporations.
We invite you to collaborate with us to explore how this advanced catalytic technology can be integrated into your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and current process constraints. Please contact our technical procurement team today to request specific COA data for our high-purity phosphite ligands and comprehensive route feasibility assessments. Let us partner with you to drive efficiency, reduce costs, and secure your position as a leader in the high-performance polymer market through the adoption of next-generation hydrocyanation chemistry.
