Advanced Catalytic Hydrocyanation Technology for High-Purity C5 Mononitriles Manufacturing
Advanced Catalytic Hydrocyanation Technology for High-Purity C5 Mononitriles Manufacturing
The global demand for high-performance polyamides and specialty chemical intermediates continues to drive innovation in C4 chemistry processing. Patent CN1144781C introduces a transformative methodology for the preparation of monoolefinic C5 mononitriles, specifically targeting the efficient conversion of industrial 1,3-butadiene-containing hydrocarbon mixtures. This technology addresses the longstanding economic and technical challenges associated with utilizing crude C4 fractions by implementing a critical pre-treatment step involving selective hydrogenation. By rigorously reducing the proportion of interfering components such as alkynes and 1,2-dienes prior to the hydrocyanation reaction, the process achieves unprecedented levels of catalyst stability and product selectivity. This breakthrough allows manufacturers to bypass the costly extraction distillation typically required to isolate pure 1,3-butadiene, thereby unlocking substantial value from readily available petroleum cracking streams. The implications for large-scale production of adiponitrile precursors are profound, offering a pathway to more sustainable and cost-effective nylon supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing C5 mononitriles often rely heavily on the use of purified 1,3-butadiene, which necessitates complex and energy-intensive separation processes from industrial C4 cuts. When crude mixtures are used without adequate pre-treatment, the presence of trace impurities like vinylacetylene, propyne, and allene leads to severe catalyst deactivation. These impurities participate in side reactions that generate conjugated nitriles, such as 2-pentenenitrile, which act as potent catalytic poisons in subsequent hydrocyanation steps. The accumulation of these conjugated by-products not only inhibits the reaction kinetics but also complicates the downstream purification of the desired non-conjugated isomers. Furthermore, conventional methods often suffer from lower hydrogen cyanide conversion rates, leading to increased handling risks and higher costs associated with the disposal of unreacted toxic reagents. The inability to effectively recycle the expensive nickel catalyst due to contamination further exacerbates the operational expenditure, making the process less competitive in a margin-sensitive market.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally shifts the paradigm by introducing a selective hydrogenation stage immediately preceding the hydrocyanation reaction. This pre-treatment effectively scrubs the feedstock of catalyst-poisoning alkynes and 1,2-dienes, reducing their total content to negligible levels, often below 1000 ppm or even 100 ppm in optimized embodiments. By ensuring the feed stream is substantially free of these interfering components, the subsequent nickel-catalyzed hydrocyanation proceeds with remarkable efficiency and selectivity. This approach enables the direct utilization of industrial C4 fractions containing as little as 10% to 50% 1,3-butadiene by volume, eliminating the need for expensive purification infrastructure. The result is a robust process that maintains high catalytic activity over extended periods, significantly improving the nickel balance sheet and allowing for higher throughput rates. This methodological refinement transforms a previously problematic feedstock into a reliable resource for high-value nitrile production.
Mechanistic Insights into Nickel-Catalyzed Hydrocyanation
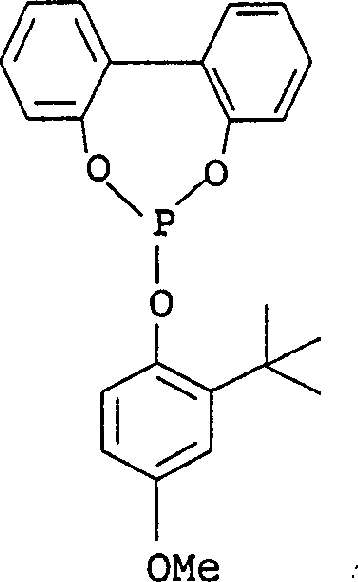
The core of this technological advancement lies in the sophisticated interplay between the nickel catalyst system and the purified hydrocarbon feedstock. The reaction employs homogeneous Nickel(0) complexes coordinated with phosphite ligands, which are essential for directing the regioselectivity towards the desired linear 3-pentenenitrile and 2-methyl-3-butenenitrile isomers. The removal of alkynes prevents the formation of stable, conjugated pi-systems that would otherwise coordinate irreversibly to the nickel center, blocking active sites. In the absence of these poisons, the catalytic cycle proceeds through the oxidative addition of hydrogen cyanide, followed by the insertion of the diene and reductive elimination to form the mononitrile. The specific electronic and steric properties of the phosphite ligands, such as those depicted in the structural analysis below, play a pivotal role in stabilizing the low-valent nickel species and facilitating the anti-Markovnikov addition required for adiponitrile precursors.
Furthermore, the mechanistic pathway ensures that the thermodynamically stable but catalytically toxic 2-pentenenitrile is minimized, typically keeping its concentration below 1% by weight. The process allows for the isomerization of less desirable branched isomers into the linear 3-pentenenitrile, which is the critical intermediate for the second hydrocyanation step to form adiponitrile. By maintaining a clean reaction environment through feedstock pre-treatment, the catalyst system avoids the formation of nickel cyanide precipitates that are difficult to regenerate. This preservation of catalyst integrity is crucial for continuous operation modes, where the catalyst is recycled multiple times. The ability to operate at moderate temperatures ranging from 70°C to 120°C and pressures between 5 bar and 20 bar further underscores the kinetic efficiency of this poisoned-free system, enabling safer and more controlled reaction conditions compared to traditional high-severity processes.
How to Synthesize C5 Mononitriles Efficiently
Implementing this synthesis route requires precise control over the feedstock composition and reaction parameters to maximize yield and catalyst life. The process begins with the selective hydrogenation of the C4 fraction using copper or palladium-based catalysts to target specific unsaturated impurities without saturating the valuable 1,3-butadiene. Following this purification, the treated stream is introduced into a hydrocyanation reactor along with hydrogen cyanide and the nickel-phosphite catalyst system. Detailed standard operating procedures regarding stoichiometry, residence time, and separation techniques are critical for replicating the high conversion rates observed in the patent examples. For a comprehensive breakdown of the specific operational steps and safety protocols required for this synthesis, please refer to the technical guide below.
- Pre-treat the industrial C4 fraction containing 1,3-butadiene via selective hydrogenation to reduce alkynes and 1,2-dienes to below 1000 ppm.
- Feed the partially hydrogenated C4 fraction, hydrogen cyanide, and a Nickel(0)-phosphite catalyst system into a reactor at 70-120°C and 5-20 bar.
- Separate unconverted reactants via distillation and recover the active nickel catalyst for recycling to ensure high conversion rates exceeding 99%.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this technology offers compelling advantages by decoupling production costs from the volatile pricing of purified 1,3-butadiene. By enabling the use of mixed C4 streams directly from steam crackers, manufacturers can secure raw materials at a significantly lower cost basis while avoiding the capital expenditure associated with extractive distillation units. The enhanced stability of the catalyst system translates directly into reduced consumption of expensive nickel complexes and ligands, lowering the overall variable cost per ton of product. Additionally, the drastic improvement in hydrogen cyanide conversion minimizes the inventory and handling requirements for this hazardous reagent, thereby reducing safety compliance costs and insurance premiums. Supply chain reliability is further bolstered by the process's tolerance to feedstock variations, ensuring consistent output even when the quality of incoming C4 cuts fluctuates within specified limits.
- Cost Reduction in Manufacturing: The elimination of complex feedstock purification steps and the extension of catalyst life cycle contribute to a leaner manufacturing cost structure. By avoiding the formation of catalyst poisons, the process reduces the frequency of catalyst replenishment and the loss of valuable nickel during recovery operations. This efficiency gain allows for a more competitive pricing model for downstream derivatives like adiponitrile and hexamethylenediamine. The qualitative reduction in waste generation also lowers disposal fees, adding another layer of financial benefit to the operation without compromising on product quality or throughput capacity.
- Enhanced Supply Chain Reliability: Utilizing widely available industrial C4 fractions mitigates the risk of supply disruptions associated with specialized purified monomers. The robustness of the pre-treatment and reaction sequence ensures that production schedules can be maintained consistently, supporting just-in-time delivery models for key customers. This reliability is critical for maintaining long-term contracts with major polymer producers who require uninterrupted flows of intermediates. The ability to source feedstock from multiple petrochemical suppliers without stringent purity specifications provides a strategic buffer against regional market shortages or logistics bottlenecks.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in both continuous stirred-tank reactors and semi-continuous autoclave configurations suitable for commercial expansion. The high conversion rates significantly reduce the environmental footprint by minimizing the release of unreacted hydrogen cyanide and reducing the volume of toxic waste streams requiring treatment. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals. The simplified downstream processing also reduces energy consumption, making the facility more resilient to rising utility costs and carbon taxation schemes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrocyanation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this process into existing production lines or new greenfield projects. The answers highlight the specific operational parameters and expected performance metrics that differentiate this method from legacy technologies.
Q: Why is selective hydrogenation critical before hydrocyanation?
A: Selective hydrogenation removes alkynes and 1,2-dienes which act as catalyst poisons, preventing the formation of conjugated nitriles that inhibit the reaction and reduce nickel recovery.
Q: What catalyst system is preferred for this process?
A: The process utilizes homogeneous Nickel(0) complexes with phosphite ligands, optionally promoted by Lewis acids, to achieve high selectivity for linear 3-pentenenitrile.
Q: How does this method improve commercial viability?
A: By utilizing crude C4 cuts instead of pure butadiene and achieving higher catalyst turnover, the method significantly reduces raw material costs and waste disposal expenses associated with toxic nickel cyanide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C5 Mononitriles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced catalytic technologies to maintain competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity C5 mononitriles that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in managing complex nickel-catalyzed systems allows us to optimize the balance between yield, selectivity, and cost, providing our partners with a distinct advantage in their respective supply chains.
We invite you to engage with our technical procurement team to discuss how this patented hydrocyanation process can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits applicable to your operation. We encourage you to contact us to obtain specific COA data and route feasibility assessments, ensuring that your project is built on a foundation of verified technical performance and commercial viability. Let us collaborate to drive efficiency and innovation in your chemical manufacturing endeavors.
