Advanced Biocatalytic Reduction for High-Purity Atazanavir Intermediates and Commercial Scalability
Advanced Biocatalytic Reduction for High-Purity Atazanavir Intermediates and Commercial Scalability
The global demand for effective antiretroviral therapies continues to drive innovation in the synthesis of critical pharmaceutical intermediates, particularly for protease inhibitors like Atazanavir. A pivotal advancement in this domain is documented in patent CN103468757A, which discloses a highly efficient biological preparation method for the key chiral intermediate of Atazanavir. This technology represents a significant paradigm shift from hazardous chemical reduction processes to environmentally benign biocatalysis, addressing long-standing challenges in safety, purity, and scalability. For R&D directors and procurement strategists, understanding the nuances of this enzymatic route is essential for optimizing supply chains and ensuring the consistent availability of high-quality antiviral agents. The method utilizes a sophisticated dual-enzyme system involving Ketoreductase (KRED) and Glucose Dehydrogenase (GDH) to achieve asymmetric reduction with exceptional stereoselectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the core chiral alcohol moiety found in Atazanavir has relied heavily on traditional chemical reduction techniques that pose substantial operational risks and environmental burdens. Conventional routes often necessitate the use of large quantities of phenylboronic acid compounds or other aggressive reducing agents that are classified as flammable and explosive chemicals. These reagents introduce severe safety hazards during storage and handling, requiring specialized infrastructure and rigorous safety protocols that inflate operational expenditures. Furthermore, the control of reaction parameters in these chemical processes is notoriously difficult, often leading to inconsistent batch quality and the formation of difficult-to-remove impurities. The generation of hazardous waste streams from these traditional methods also complicates compliance with increasingly stringent environmental regulations, forcing manufacturers to invest heavily in waste treatment facilities. Consequently, the reliance on such volatile chemistry creates bottlenecks in production capacity and threatens the continuity of supply for critical HIV medications.
The Novel Approach
In stark contrast to these legacy methods, the biological preparation method outlined in the patent offers a transformative solution by leveraging the specificity and mildness of enzymatic catalysis. This novel approach employs a Ketoreductase (KRED) to selectively reduce the ketone precursor to the desired chiral alcohol under strictly controlled, mild conditions. 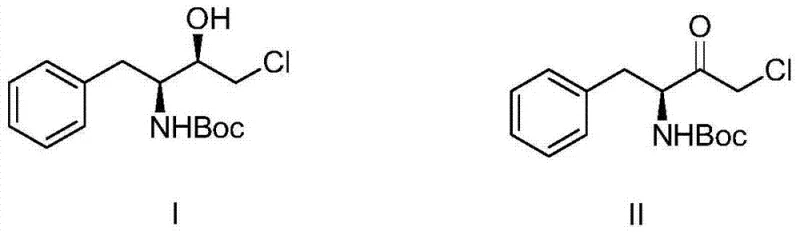 As illustrated in the structural transformation, the process converts the chloro-ketone substrate directly into the hydroxy-intermediate with high fidelity. The elimination of flammable boronic reagents and the transition to an aqueous-based system drastically reduces the risk of fire and explosion, creating a inherently safer manufacturing environment. Moreover, the process operates at moderate temperatures and neutral to slightly alkaline pH levels, which preserves the integrity of sensitive functional groups and minimizes side reactions. This shift not only enhances the safety profile of the facility but also simplifies the downstream purification process, leading to higher overall yields and reduced solvent consumption.
As illustrated in the structural transformation, the process converts the chloro-ketone substrate directly into the hydroxy-intermediate with high fidelity. The elimination of flammable boronic reagents and the transition to an aqueous-based system drastically reduces the risk of fire and explosion, creating a inherently safer manufacturing environment. Moreover, the process operates at moderate temperatures and neutral to slightly alkaline pH levels, which preserves the integrity of sensitive functional groups and minimizes side reactions. This shift not only enhances the safety profile of the facility but also simplifies the downstream purification process, leading to higher overall yields and reduced solvent consumption.
Mechanistic Insights into KRED-Catalyzed Asymmetric Reduction
The core of this technological breakthrough lies in the intricate design of the biocatalytic cycle, which ensures both high conversion efficiency and economic viability through cofactor regeneration. The reaction mechanism relies on the synergistic action of Ketoreductase (KRED) and Glucose Dehydrogenase (GDH) within a carefully buffered aqueous environment. 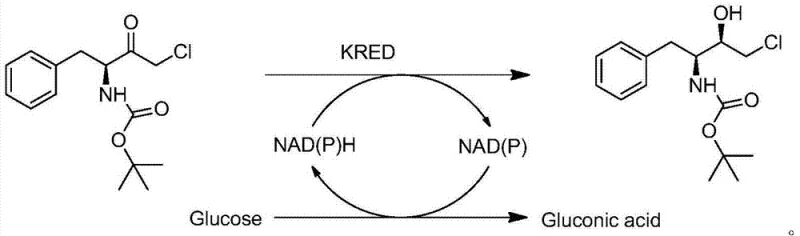 As depicted in the catalytic cycle, the KRED enzyme facilitates the hydride transfer from the reduced cofactor NAD(P)H to the carbonyl group of the substrate, generating the chiral alcohol and oxidizing the cofactor to NAD(P)+. To prevent the accumulation of the expensive oxidized cofactor and drive the equilibrium forward, GDH simultaneously oxidizes glucose to gluconic acid, regenerating NAD(P)H in situ. This closed-loop system means that only catalytic amounts of the cofactor are required, rather than stoichiometric quantities, which is a critical factor in reducing the cost of goods sold. The patent specifies maintaining the reaction temperature between 32°C and 34°C, a range that optimizes enzyme activity while preventing thermal denaturation, ensuring stable performance over extended reaction times. This precise control over the biocatalytic environment allows for the consistent production of the (2S, 3R) stereoisomer, which is crucial for the biological activity of the final drug product.
As depicted in the catalytic cycle, the KRED enzyme facilitates the hydride transfer from the reduced cofactor NAD(P)H to the carbonyl group of the substrate, generating the chiral alcohol and oxidizing the cofactor to NAD(P)+. To prevent the accumulation of the expensive oxidized cofactor and drive the equilibrium forward, GDH simultaneously oxidizes glucose to gluconic acid, regenerating NAD(P)H in situ. This closed-loop system means that only catalytic amounts of the cofactor are required, rather than stoichiometric quantities, which is a critical factor in reducing the cost of goods sold. The patent specifies maintaining the reaction temperature between 32°C and 34°C, a range that optimizes enzyme activity while preventing thermal denaturation, ensuring stable performance over extended reaction times. This precise control over the biocatalytic environment allows for the consistent production of the (2S, 3R) stereoisomer, which is crucial for the biological activity of the final drug product.
Furthermore, the mechanism includes robust impurity control features inherent to the enzyme's active site specificity. Unlike chemical catalysts which may reduce multiple functional groups non-selectively, the KRED enzyme is engineered or selected to recognize the specific steric and electronic environment of the target ketone. This high degree of chemoselectivity ensures that the chloro-group and the carbamate protecting group remain intact during the reduction, preventing the formation of dehalogenated or deprotected byproducts that are common in harsh chemical reductions. The use of a triethanolamine buffered solution at pH 8-9 further stabilizes the enzyme structure and maintains the ionization state of the substrate for optimal binding. By minimizing the formation of structural analogs and diastereomers, the process significantly reduces the burden on downstream chromatography or crystallization steps. This results in a final product with chemical purity exceeding 97% and optical purity greater than 99.5%, meeting the rigorous specifications required for pharmaceutical intermediates intended for human use.
How to Synthesize Atazanavir Intermediate Efficiently
Implementing this biocatalytic route requires precise adherence to the reaction parameters defined in the patent to ensure reproducibility and maximum yield. The process begins with the preparation of a heterogeneous reaction mixture where the hydrophobic substrate is dispersed in a system containing a water-miscible cosolvent such as acetonitrile or toluene. The detailed standardized synthetic steps involve specific mass ratios of enzymes to substrate, ensuring that the catalytic turnover is sufficient to drive the reaction to completion within a commercially viable timeframe. Operators must carefully monitor the conversion rate using HPLC analysis, aiming for a transformation efficiency of at least 96% before quenching the reaction. The following guide outlines the critical operational phases for executing this synthesis effectively.
- Prepare the reaction mixture by adding the substrate, cosolvent (acetonitrile or toluene), and glucose into the reactor with uniform stirring.
- Dissolve Ketoreductase (KRED), Glucose Dehydrogenase (GDH), and cofactor (NAD/NADP) in a buffered aqueous solution at pH 8-9.
- Add the enzyme solution to the reactor, maintain temperature at 32-34°C, and monitor conversion until it exceeds 96%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this biocatalytic technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The transition from hazardous chemical reagents to benign biological catalysts fundamentally alters the risk profile of the manufacturing process, leading to significant reductions in insurance premiums and safety compliance costs. By eliminating the need for specialized handling of explosive phenylboronic compounds, facilities can operate with greater flexibility and reduced downtime associated with safety audits and hazard mitigation protocols. This operational streamlining allows for more predictable production schedules and enhances the reliability of supply for downstream API manufacturers. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cooling, contributing to a lower carbon footprint and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The implementation of the cofactor regeneration system described in the patent offers a profound impact on raw material costs by minimizing the consumption of expensive nicotinamide cofactors. Instead of purchasing stoichiometric amounts of NAD(P)H, the process utilizes a catalytic amount regenerated continuously by glucose, which is a low-cost and readily available commodity chemical. This drastic reduction in reagent intensity lowers the variable cost per kilogram of the intermediate, providing a competitive pricing advantage in the global market. Furthermore, the high selectivity of the enzyme reduces the loss of valuable starting materials to side products, improving the overall mass balance and atom economy of the synthesis. These factors combined result in a more economically robust process that can withstand fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The stability of the enzymatic process under mild conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with pharmaceutical clients. Unlike chemical processes that may suffer from runaway reactions or sensitivity to trace impurities, this biological system is robust and forgiving, reducing the incidence of failed batches that disrupt supply continuity. The ability to operate at higher substrate concentrations compared to some prior art biological methods means that reactor throughput is maximized, allowing suppliers to meet large volume demands without proportional increases in capital equipment. This scalability ensures that the supply chain can respond agilely to surges in demand for antiretroviral therapies without compromising on delivery timelines.
- Scalability and Environmental Compliance: From an environmental perspective, the replacement of organic solvents and hazardous reagents with aqueous buffers and glucose significantly simplifies waste management and treatment. The effluent generated from this process is far less toxic and easier to treat than waste streams from traditional boronic acid reductions, reducing the liability and cost associated with environmental compliance. The process generates gluconic acid as a byproduct, which is biodegradable and non-toxic, further enhancing the green credentials of the manufacturing route. This alignment with green chemistry principles not only satisfies regulatory requirements but also appeals to end-users who are increasingly prioritizing sustainable sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic process for Atazanavir intermediate production. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on performance metrics and operational requirements. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing frameworks.
Q: What are the primary safety advantages of this biological method over traditional chemical reduction?
A: Unlike traditional methods requiring flammable and explosive phenylboronic acid compounds, this biocatalytic process operates under mild aqueous conditions, significantly reducing fire hazards and toxic waste generation.
Q: How does the cofactor regeneration system improve process economics?
A: By utilizing Glucose Dehydrogenase (GDH) and glucose to regenerate NAD(P)H in situ, the process minimizes the requirement for expensive stoichiometric amounts of cofactors, thereby lowering overall production costs.
Q: What optical purity levels can be achieved with this KRED-catalyzed route?
A: The patented method demonstrates the capability to achieve optical purity greater than 99.5% and chemical purity exceeding 97%, meeting stringent requirements for antiviral API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atazanavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced biocatalytic technologies to meet the evolving needs of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in enzymatic processes allows us to optimize reaction conditions further, maximizing yield and minimizing impurities to deliver a product that exceeds industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a reliable and cost-effective supply of this vital antiviral intermediate.
