Advanced Synthesis of Obeticholic Acid Intermediates for Commercial Scale Manufacturing
The pharmaceutical industry is constantly seeking robust, scalable, and high-purity synthetic routes for critical API intermediates, particularly for treatments targeting metabolic disorders like Nonalcoholic Steatohepatitis (NASH). Patent CN116178473A introduces a groundbreaking preparation method for Obeticholic Acid, a potent Farnesoid X Receptor (FXR) agonist. This novel methodology addresses long-standing challenges in the synthesis of bile acid derivatives by employing a strategic Weinreb amide protection strategy. By shifting away from traditional direct alkylation or unstable enol condensation methods, this process achieves an exceptional total yield of 87.4% and a final product purity of 99.9%. For R&D directors and procurement managers, this represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, offering a reliable pathway that minimizes waste and maximizes throughput. The technical elegance of this route lies in its ability to stabilize reactive intermediates, thereby simplifying downstream processing and ensuring consistent quality suitable for global regulatory standards.
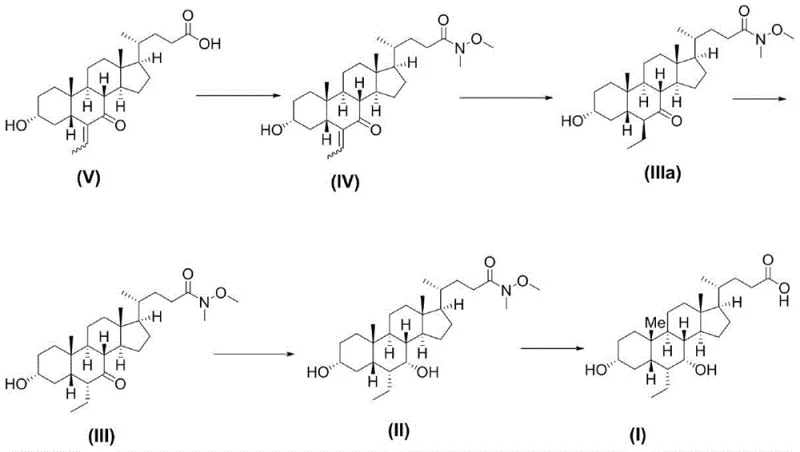
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
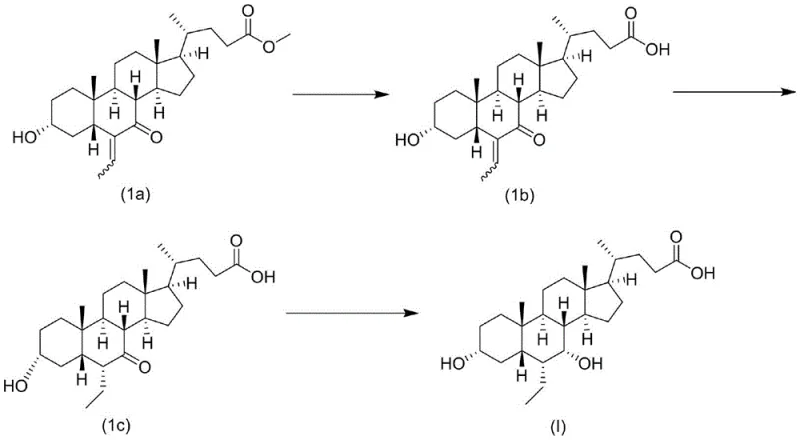
Historically, the synthesis of Obeticholic Acid has been plagued by inefficiencies that hinder large-scale commercial viability. Prior art, such as the method disclosed in WO2006122977, relies on the condensation of silicon-protected enol compounds with acetaldehyde followed by palladium-carbon hydrogenation. While theoretically feasible, this conventional approach suffers from severe practical drawbacks, including poor selectivity during the alkylation step and the formation of numerous by-products. A critical failure point in these legacy processes is the instability of intermediates during purification; specifically, the intermediate corresponding to Compound 1c in the prior art tends to precipitate as an oil rather than a crystal. This oily precipitation makes impurity removal exceptionally difficult, leading to a cumulative total yield of merely 43.9% and a final purity that struggles to reach 98.1%. Such low efficiency translates directly into exorbitant production costs and supply chain volatility, making it an unsustainable choice for a reliable pharmaceutical intermediates supplier aiming for mass production.
The Novel Approach
In stark contrast, the methodology described in CN116178473A revolutionizes the synthesis by introducing a Weinreb amide protection group at the C24 carboxyl position early in the sequence. This strategic modification transforms the chemical nature of the side chain, converting a potentially problematic carboxylic acid into a stable amide that facilitates superior crystallization properties. As illustrated in the comparative reaction pathways, this new route bypasses the formation of oily impurities entirely. The process involves the conversion of Compound V to the Weinreb amide Compound IV, followed by stereoselective hydrogenation and epimerization to install the crucial 6-alpha ethyl group. The result is a dramatic improvement in process metrics: the total yield more than doubles to 87.4%, and the final purity reaches an impressive 99.9%. This approach not only simplifies the operational workflow but also significantly enhances the commercial scale-up of complex pharmaceutical intermediates by ensuring that each step proceeds with high conversion and minimal by-product formation.
Mechanistic Insights into Weinreb Amide Protection and Stereoselective Reduction
The core innovation of this synthesis lies in the mechanistic advantages provided by the Weinreb amide functionality. In the initial step, Compound V reacts with dimethylhydroxylamine hydrochloride in the presence of coupling agents like EDCI and HOBt to form Compound IV. Mechanistically, the Weinreb amide acts as a robust protecting group that prevents unwanted side reactions at the carboxyl terminus while remaining inert to the subsequent reduction conditions. Unlike free acids which can complicate extraction and crystallization due to salt formation, the neutral amide allows for clean phase separation and high-efficiency crystallization from solvents like n-heptane. This stability is crucial for maintaining high purity throughout the multi-step sequence, as it prevents the carryover of acidic impurities that could catalyze degradation in later stages. Furthermore, the amide group serves as a latent carboxylic acid, easily revealed in the final step under mild alkaline hydrolysis, ensuring the integrity of the sensitive steroid backbone is preserved until the very end of the synthesis.
Stereochemical control is another pillar of this mechanism, particularly regarding the installation of the 6-alpha ethyl group. The process first generates the 6-beta isomer (Compound IIIa) via catalytic hydrogenation using 10% Pd/C. This kinetic product is then subjected to base-mediated epimerization using sodium methoxide in tetrahydrofuran. Under these thermodynamic conditions, the bulky ethyl group equilibrates to the more stable alpha-configuration, yielding Compound III with high diastereoselectivity. Subsequent reduction of the 7-keto group using sodium borohydride at low temperatures (-10 to 0°C) ensures the selective formation of the 7-alpha hydroxyl moiety without affecting the newly installed ethyl group. This precise control over stereochemistry is vital for the biological activity of Obeticholic Acid, as the wrong isomer would be inactive or potentially toxic. The combination of kinetic hydrogenation followed by thermodynamic equilibration provides a robust method for achieving the exact spatial arrangement required for FXR receptor activation.
How to Synthesize Obeticholic Acid Efficiently
The synthesis of Obeticholic Acid via this novel route is designed for operational simplicity and high reproducibility, making it ideal for both pilot and commercial scales. The process begins with the activation of the carboxyl group in Compound V, followed by a sequence of reduction, epimerization, and deprotection steps that utilize common, commercially available reagents. Detailed standard operating procedures for temperature control, reagent addition rates, and work-up protocols are essential to replicate the high yields reported in the patent. For technical teams looking to implement this chemistry, understanding the critical crystallization points—specifically the use of n-heptane for Compound IV and ethyl acetate for Compound II—is key to maximizing recovery. The detailed standardized synthesis steps are outlined below to guide your process development team.
- Protect the carboxyl group of Compound V using dimethylhydroxylamine hydrochloride and EDCI/HOBt to form the stable Weinreb amide Compound IV.
- Perform catalytic hydrogenation on Compound IV using 10% Pd/C to obtain Compound IIIa, followed by base-mediated epimerization to secure the 6-alpha ethyl configuration in Compound III.
- Reduce the 7-keto group of Compound III using sodium borohydride at low temperature (-10 to 0°C) to generate the 7-alpha hydroxy intermediate Compound II.
- Hydrolyze the Weinreb amide protecting group of Compound II under alkaline conditions (NaOH/MeOH) at elevated temperature to yield high-purity Obeticholic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative economic and logistical benefits. The most immediate impact is the substantial increase in overall yield, which jumps from approximately 44% in conventional methods to over 87% in this new process. This near-doubling of efficiency means that for the same amount of starting material, manufacturers can produce nearly twice the volume of finished API, effectively halving the raw material cost per kilogram of product. Additionally, the elimination of difficult-to-remove oily impurities reduces the need for extensive chromatographic purification or multiple recrystallizations, which are both time-consuming and solvent-intensive. This streamlining of the purification process leads to significant cost reduction in manufacturing operations and shortens the overall production cycle time, allowing for faster response to market demand.
- Cost Reduction in Manufacturing: The implementation of the Weinreb amide strategy eliminates the need for expensive silicon-protecting groups and reduces the consumption of solvents and reagents associated with low-yield steps. By avoiding the formation of intractable oily by-products, the process minimizes material loss during purification, directly translating to lower cost of goods sold (COGS). Furthermore, the use of standard catalysts like 10% Pd/C and common reducing agents like sodium borohydride ensures that reagent costs remain low and predictable, avoiding the volatility associated with specialized or scarce reagents.
- Enhanced Supply Chain Reliability: The robustness of the crystallization steps in this new method ensures a consistent supply of high-quality intermediates. In previous methods, the tendency of intermediates to oil out created bottlenecks and batch failures, leading to unpredictable lead times. With the improved solid-state properties of Compounds IV, III, and II, manufacturers can guarantee steady production schedules. This reliability is critical for maintaining continuous API supply chains, reducing the risk of stockouts, and ensuring that downstream drug formulation timelines are met without delay.
- Scalability and Environmental Compliance: The simplified workflow and reduced solvent usage make this process highly scalable from kilogram to multi-ton production. The ability to use greener solvents like ethyl acetate and n-heptane, combined with higher atom economy due to improved yields, aligns with modern environmental, social, and governance (ESG) goals. Reduced waste generation lowers the burden on wastewater treatment facilities and decreases the environmental footprint of the manufacturing site, facilitating easier regulatory approval and community acceptance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Obeticholic Acid synthesis route. These answers are derived directly from the experimental data and mechanistic insights provided in patent CN116178473A, offering clarity on yield expectations, purity profiles, and process scalability. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for commercial production.
Q: What is the primary advantage of using Weinreb amide protection in Obeticholic Acid synthesis?
A: The use of Weinreb amide protection significantly improves the purification efficiency of intermediates. Unlike free carboxylic acids which can form difficult-to-remove salts or oils, the Weinreb amide allows for stable crystallization, leading to higher purity intermediates (up to 99.2%) and a much higher overall yield compared to conventional methods.
Q: How does this new method improve the stereochemical control at the C6 position?
A: The process utilizes a two-step sequence involving catalytic hydrogenation to form the 6-beta ethyl isomer (Compound IIIa), followed by a controlled base-mediated epimerization using sodium methoxide. This ensures the thermodynamically stable 6-alpha ethyl configuration required for biological activity is achieved with high selectivity.
Q: What is the expected purity of the final Obeticholic Acid product using this route?
A: According to the patent data, the final Obeticholic Acid produced via this method achieves a purity of 99.9%. This is a significant improvement over prior art methods which typically struggle to exceed 98.1% purity due to persistent oily impurities during crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Obeticholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value pharmaceutical intermediates like Obeticholic Acid. Our team of expert chemists has extensively analyzed the methodology described in CN116178473A and possesses the technical capability to execute this complex multi-step synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate or API we deliver meets the highest global standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By leveraging our expertise in process optimization and cost management, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how NINGBO INNO PHARMCHEM can become your trusted partner in delivering high-quality Obeticholic Acid intermediates for the global market.
