Revolutionizing Peptide Synthesis: High-Purity Fmoc-beta-Ala-Gly-OH via Rare Earth Catalysis
The landscape of polypeptide synthesis intermediates is undergoing a significant transformation driven by the need for higher purity and reduced manufacturing costs. Patent CN110240551B introduces a groundbreaking preparation method for Fmoc-beta-Ala-Gly-OH, a critical building block in the assembly of complex therapeutic peptides. Unlike conventional strategies that suffer from cumbersome protection-deprotection cycles, this invention leverages a rare earth catalytic system to streamline the coupling of Fmoc-beta-alanine derivatives with glycine. By utilizing ytterbium boride as a specialized catalyst, the process achieves rapid reaction kinetics and exceptional selectivity, effectively addressing the long-standing challenges of low yield and difficult purification that have plagued the industry. This technological leap not only enhances the economic viability of producing high-quality peptide intermediates but also establishes a robust foundation for scalable industrial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
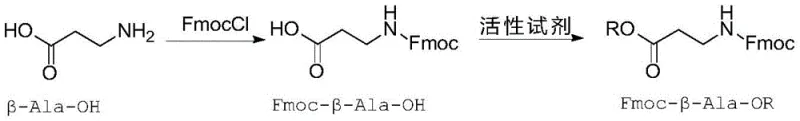
Historically, the synthesis of Fmoc-beta-Ala-Gly-OH has been hindered by inefficient liquid-phase and solid-phase strategies that impose severe bottlenecks on production efficiency. Traditional liquid-phase methods typically employ a full protection route, necessitating the generation of an activated ester, followed by coupling, purification, saponification, and finally, the re-introduction of the Fmoc protecting group. This multi-step sequence is inherently flawed; the removal and subsequent re-addition of the Fmoc group drastically increase reagent consumption and operational complexity, often resulting in overall yields below 50 percent. Furthermore, the cumulative effect of multiple reaction steps invariably leads to the accumulation of impurities, making it exceptionally difficult to achieve final product purity levels above 95 percent without extensive and costly chromatographic purification.
The Novel Approach
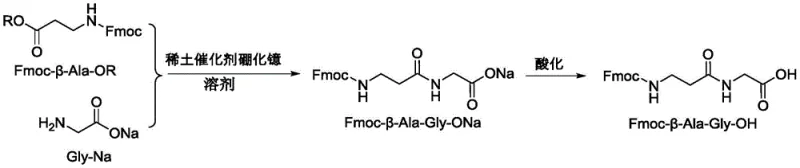
In stark contrast, the methodology disclosed in CN110240551B offers a streamlined, direct coupling pathway that bypasses the unnecessary deprotection steps of legacy processes. By reacting an activated Fmoc-beta-Ala-OR ester directly with glycine sodium salt in the presence of a ytterbium boride catalyst, the synthesis is condensed into a highly efficient single-pot coupling followed by a simple acidification workup. This approach eliminates the need for expensive resin supports used in solid-phase synthesis and avoids the yield losses associated with repetitive protection group manipulation. The result is a robust process capable of delivering yields exceeding 80 percent with purity levels consistently surpassing 99 percent, representing a paradigm shift in how peptide intermediates are manufactured commercially.
Mechanistic Insights into Ytterbium Boride-Catalyzed Amidation
The core innovation of this patented process lies in the strategic application of ytterbium boride as a Lewis acid catalyst to facilitate the nucleophilic attack of the glycine amine on the activated ester. In standard amidation reactions without catalysis, the reaction rate can be sluggish, and competing hydrolysis of the activated ester often leads to significant byproduct formation. The ytterbium ion coordinates with the carbonyl oxygen of the activated ester, increasing its electrophilicity and thereby lowering the activation energy required for the amine attack. This coordination effect ensures that the coupling proceeds rapidly even at mild temperatures, typically between 0°C and 50°C, which is crucial for preserving the integrity of the sensitive Fmoc protecting group and preventing racemization.
Furthermore, the catalytic system plays a pivotal role in impurity control, which is a primary concern for R&D directors focused on downstream processing. The presence of the rare earth catalyst suppresses side reactions such as the self-condensation of the activated ester or the formation of diketopiperazines, which are common pitfalls in peptide synthesis. By maintaining a precise molar ratio of reactants—optimally around 100:200:10 for the ester, glycine salt, and catalyst respectively—the reaction environment is tuned to favor the formation of the desired dipeptide sodium salt. Subsequent acidification allows for the clean precipitation of the free acid form, while the use of solvents like tetrahydrofuran or dioxane ensures excellent solubility profiles that facilitate easy separation of the product from inorganic salts and catalyst residues.
How to Synthesize Fmoc-beta-Ala-Gly-OH Efficiently
The execution of this synthesis requires careful attention to stoichiometry and solvent selection to maximize the benefits of the catalytic cycle. The process begins with the preparation or procurement of the activated ester, such as Fmoc-beta-Ala-OBt or Fmoc-beta-Ala-OSu, which serves as the electrophilic partner. Detailed standardized operating procedures regarding the specific addition rates, temperature controls during the exothermic mixing phase, and the precise pH adjustment during the acidification step are critical for reproducibility. For a comprehensive guide on the exact experimental parameters and safety protocols required to implement this route in a GMP environment, please refer to the structured synthesis guide provided below.
- Mix Fmoc-beta-Ala-OR activated ester, Gly-Na, and ytterbium boride catalyst in a solvent like THF or dioxane.
- Stir the reaction mixture at room temperature until TLC confirms the disappearance of the starting ester.
- Concentrate the solution, extract with ethyl acetate, acidify to pH ≤ 2, and purify the resulting solid via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic synthesis route translates directly into tangible operational efficiencies and risk mitigation. The elimination of multiple synthetic steps significantly reduces the consumption of raw materials, particularly the expensive Fmoc-chloride reagent and organic solvents, leading to a substantial reduction in the overall cost of goods sold. Moreover, the simplified workflow decreases the total production cycle time, allowing manufacturers to respond more agilely to market demand fluctuations without the burden of maintaining large inventories of intermediate compounds. This efficiency gain is compounded by the high purity of the crude product, which minimizes the need for resource-intensive purification processes like preparative HPLC, further driving down manufacturing expenses.
- Cost Reduction in Manufacturing: The streamlined nature of this process removes the economic burden of the traditional "protect-couple-deprotect-recouple" cycle. By avoiding the double usage of Fmoc reagents and eliminating the need for costly solid-phase resins, the material cost profile is drastically improved. Additionally, the high conversion rates mean less waste generation and lower disposal costs, contributing to a leaner and more profitable manufacturing model that enhances competitiveness in the global peptide market.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like glycine sodium salt and common organic solvents ensures a stable supply chain that is less susceptible to disruptions. The robustness of the reaction conditions, which tolerate ambient temperatures and do not require extreme pressures or cryogenic cooling, simplifies equipment requirements and allows for production in a wider range of facilities. This flexibility ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, securing the continuity of supply for downstream drug manufacturers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method offers distinct advantages over legacy technologies. The reduction in solvent usage and the avoidance of hazardous deprotection reagents align with green chemistry principles, simplifying regulatory compliance and waste treatment. The process is inherently scalable from kilogram to multi-ton scales without significant re-engineering, as the catalytic mechanism remains effective regardless of batch size. This scalability ensures that suppliers can meet growing commercial demands for peptide therapeutics while maintaining strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method outperforms traditional approaches in terms of yield, purity, and operational simplicity. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: What is the primary advantage of using ytterbium boride in this synthesis?
A: Ytterbium boride acts as a highly efficient Lewis acid catalyst that significantly accelerates the amide coupling reaction while suppressing side reactions, leading to much higher yields compared to uncatalyzed methods.
Q: How does this method improve upon traditional liquid-phase synthesis?
A: Traditional methods often require removing and re-adding the Fmoc protecting group, which increases steps and cost. This novel route avoids those extra steps, directly coupling the protected amino acid to glycine, thereby reducing production time and reagent consumption.
Q: What purity levels can be achieved with this patented process?
A: The process is designed to minimize impurities from the start. Through optimized reaction conditions and a simple ethyl acetate pulping purification step, HPLC purity exceeding 99% is consistently achievable, surpassing the typical <95% purity of older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fmoc-beta-Ala-Gly-OH Supplier
As the demand for high-quality peptide intermediates continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies like the one described in CN110240551B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Fmoc-beta-Ala-Gly-OH meets the highest international standards for peptide synthesis applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative catalytic route can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this high-efficiency process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your peptide development projects proceed with the most reliable and cost-effective intermediates available in the market.
