Scalable Synthesis of 2-Acetonyl-3-Methoxypiperidine Intermediates for Halofuginone Production
Introduction to Advanced Intermediate Synthesis
The pharmaceutical landscape for veterinary antiparasitics and potential human therapeutics is heavily reliant on the efficient production of Halofuginone, a potent derivative of Febrifugine. As detailed in Chinese Patent CN102603615B, a significant technological breakthrough has been achieved in the synthesis of the critical piperidine ring intermediate, 2-acetonyl-3-methoxypiperidine. This patent addresses long-standing bottlenecks in the manufacturing of Halofuginone by introducing a novel synthetic pathway that bypasses traditional, cost-prohibitive methods. The structural complexity of Halofuginone, which combines a quinazolinone moiety with a substituted piperidine ring, has historically made its commercial scale-up challenging. 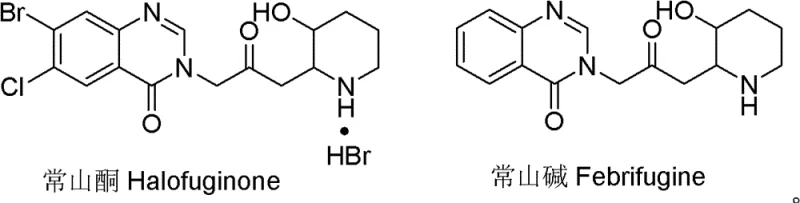 . By focusing on the optimization of the piperidine segment, this technology offers a viable route for reliable pharmaceutical intermediate supplier networks to secure stable supplies of this high-value compound.
. By focusing on the optimization of the piperidine segment, this technology offers a viable route for reliable pharmaceutical intermediate supplier networks to secure stable supplies of this high-value compound.
The significance of this innovation extends beyond mere academic interest; it represents a tangible shift towards more sustainable and economically feasible manufacturing processes. Halofuginone is widely recognized for its efficacy against coccidiosis in poultry and livestock, and emerging research suggests potential applications in treating fibrosis and certain malignancies. Consequently, the demand for high-purity intermediates is projected to grow. The methodology outlined in CN102603615B utilizes methoxyacetone as a foundational building block, a strategic choice that fundamentally alters the cost structure of the synthesis. This report analyzes the technical merits of this approach, contrasting it with legacy methods to highlight its value proposition for R&D directors and procurement strategists alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the piperidine ring for Halofuginone synthesis has been fraught with economic and technical inefficiencies. Prior art, such as the route described in Chinese Patent CN1651428A, relied heavily on 3-hydroxypyridine as the starting material. This approach necessitates a series of complex transformations, including aminomethylation, acetylation, and bromination, before the ring can be functionalized. A critical bottleneck in this conventional pathway is the hydrogenation step, which traditionally employs expensive rhodium-carbon catalysts to reduce the pyridine ring to the corresponding piperidine derivative. 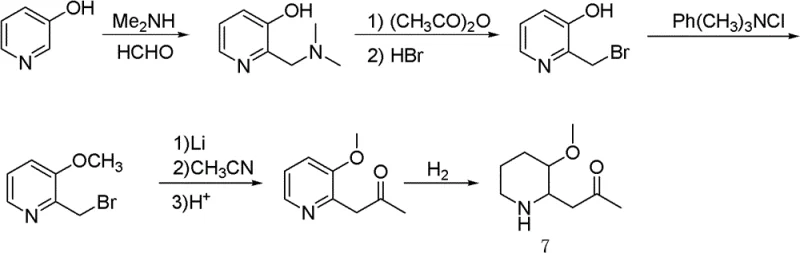 . The reliance on rhodium not only inflates the direct material costs but also introduces supply chain vulnerabilities associated with precious metal availability. Furthermore, the yields in these multi-step sequences are often suboptimal, leading to significant material loss and increased waste generation, which complicates environmental compliance and escalates the overall cost of goods sold.
. The reliance on rhodium not only inflates the direct material costs but also introduces supply chain vulnerabilities associated with precious metal availability. Furthermore, the yields in these multi-step sequences are often suboptimal, leading to significant material loss and increased waste generation, which complicates environmental compliance and escalates the overall cost of goods sold.
The Novel Approach
In stark contrast, the novel approach disclosed in CN102603615B circumvents these issues by adopting a linear synthesis strategy starting from methoxyacetone and acrylonitrile. This route avoids the formation of pyridine intermediates entirely, thereby eliminating the need for difficult aromatic reductions. Instead, it constructs the piperidine ring through a sequence of Michael addition, Claisen condensation, and intramolecular cyclization. The key innovation lies in the final hydrogenation step, where the exocyclic double bond of the intermediate 3-methoxy-2-(2-oxopropylidene)piperidine is reduced. Crucially, this reduction can be effectively catalyzed by palladium-carbon, a significantly cheaper and more accessible catalyst compared to rhodium. 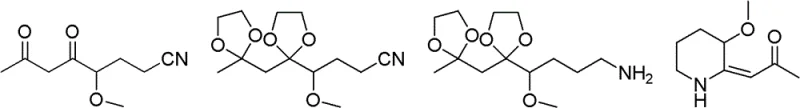 . This substitution drastically reduces reagent costs while maintaining high reaction efficiency. The process is designed with industrial scalability in mind, utilizing standard unit operations such as distillation and filtration, making it an attractive option for cost reduction in API manufacturing.
. This substitution drastically reduces reagent costs while maintaining high reaction efficiency. The process is designed with industrial scalability in mind, utilizing standard unit operations such as distillation and filtration, making it an attractive option for cost reduction in API manufacturing.
Mechanistic Insights into the Catalytic Cyclization and Reduction
The core of this synthetic advancement lies in the precise control of reaction mechanisms to ensure high purity and yield. The process begins with a Michael addition between methoxyacetone and acrylonitrile, facilitated by a base catalyst like cyclohexylamine, to form 4-methoxy-5-oxocapronitrile. This is followed by a Claisen condensation with ethyl acetate to extend the carbon chain, creating a 1,3-dicarbonyl system. To prevent unwanted side reactions during subsequent steps, the ketone carbonyls are protected using ethylene glycol under acidic conditions, forming a stable ketal. The nitrile group is then selectively reduced to a primary amine using Raney nickel or lithium aluminum hydride, setting the stage for ring closure. Under acidic hydrolysis, the protecting groups are removed, and the molecule undergoes spontaneous intramolecular cyclization to form the enamine intermediate, 3-methoxy-2-(2-oxopropylidene)piperidine.
The final and most critical mechanistic step is the selective hydrogenation of the exocyclic double bond. Unlike the aromatic reduction of pyridine which requires harsh conditions and noble metals, the reduction of this enamine system is kinetically favorable. The use of palladium-carbon catalysts at moderate hydrogen pressures (1.5-3 MPa) allows for the clean conversion of the alkene to the saturated alkane without affecting the methoxy ether or the ketone functionality. This chemoselectivity is paramount for maintaining the integrity of the final product, 2-acetonyl-3-methoxypiperidine. The mechanism ensures that impurities related to over-reduction or deprotection are minimized, resulting in a cleaner crude product that requires less intensive downstream purification. This level of control is essential for meeting the stringent purity specifications required for veterinary and potential human pharmaceutical applications.
How to Synthesize 2-Acetonyl-3-Methoxypiperidine Efficiently
The practical implementation of this synthesis involves a carefully orchestrated sequence of reactions that balance yield, safety, and operational simplicity. The patent outlines three distinct variations of the route, offering flexibility depending on available equipment and reagent preferences. The primary route involves the initial Michael addition at elevated temperatures (140°C-220°C) to drive the reaction to completion, followed by base-catalyzed condensation. The protection and reduction steps are conducted under controlled atmospheres to prevent oxidation of sensitive intermediates. The final cyclization is triggered by acid hydrolysis, which simultaneously removes the ethylene glycol protecting groups and promotes ring closure. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Perform Michael addition of methoxyacetone and acrylonitrile to obtain 4-methoxy-5-oxocapronitrile.
- Conduct ketoester condensation with ethyl acetate followed by ethylene glycol protection to form the protected nitrile intermediate.
- Reduce the nitrile group to an amine, remove the protecting group, and cyclize under basic conditions to yield the final piperidine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for Halofuginone intermediates. The shift away from specialized pyridine derivatives to commodity chemicals like methoxyacetone and acrylonitrile fundamentally de-risks the supply chain. These starting materials are produced on a massive global scale, ensuring consistent availability and shielding manufacturers from the price volatility often seen with fine chemical precursors. Furthermore, the elimination of the rhodium-carbon catalyst removes a significant cost center. Palladium-carbon is not only cheaper but also easier to recover and recycle, contributing to a more sustainable and cost-effective operation. This transition supports substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality.
- Cost Reduction in Manufacturing: The replacement of expensive rhodium catalysts with palladium-carbon directly lowers the bill of materials. Additionally, the use of methoxyacetone as a starting material is significantly more economical than purchasing protected pyridine derivatives. The streamlined process reduces the number of isolation steps, which in turn lowers solvent consumption and energy usage. By simplifying the synthetic pathway, the overall production cost is drastically reduced, allowing for more competitive pricing in the final API market. This efficiency gain is critical for maintaining margins in the highly competitive veterinary pharmaceutical sector.
- Enhanced Supply Chain Reliability: Relying on bulk chemicals rather than custom-synthesized intermediates enhances supply security. Methoxyacetone and acrylonitrile are readily available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate standard industrial equipment, means that production can be easily scaled or shifted between manufacturing sites if necessary. This flexibility ensures continuity of supply, a key metric for supply chain heads managing just-in-time inventory for large-scale animal health production lines.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as distillation and filtration that are standard in chemical plants. The avoidance of heavy metal contaminants associated with rhodium simplifies waste treatment and regulatory compliance. The higher yields reported in the patent examples mean less waste generation per kilogram of product, aligning with green chemistry principles. This makes the technology not only economically viable but also environmentally responsible, facilitating smoother regulatory approvals and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis route is essential for stakeholders evaluating its adoption. The following questions address common concerns regarding the feasibility, quality, and economic impact of the technology described in CN102603615B. These answers are derived directly from the patent specifications and provide clarity on how this method compares to existing industry standards. For further technical discussions or specific data requests, our team is available to provide deeper insights.
Q: What is the primary cost advantage of this new synthesis route?
A: The process replaces expensive 3-hydroxypyridine starting materials and costly rhodium-carbon catalysts with cheap methoxyacetone and standard palladium-carbon catalysts, significantly lowering raw material and reagent costs.
Q: How does this method improve supply chain reliability?
A: By utilizing commodity chemicals like methoxyacetone and acrylonitrile instead of specialized pyridine derivatives, the supply chain becomes more robust against raw material shortages and price volatility.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly designs the route for industrialization by avoiding complex purification steps and using standard hydrogenation equipment compatible with palladium-carbon catalysts at moderate pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acetonyl-3-Methoxypiperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis technology described in CN102603615B for the global Halofuginone market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this route, including high-pressure hydrogenation and precise distillation processes. We are committed to delivering high-purity 2-acetonyl-3-methoxypiperidine that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise ensures that the theoretical benefits of this patent are realized in practical, commercial-scale manufacturing.
We invite potential partners to engage with us to explore how this cost-effective synthesis route can enhance your supply chain. By leveraging our technical proficiency, you can secure a reliable source of this critical intermediate while achieving significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your long-term production goals and help you navigate the complexities of modern pharmaceutical manufacturing.
