Revolutionizing D-Ribulose Manufacturing: A Deep Dive into Immobilized Multi-Enzyme Cascade Technology
The pharmaceutical and biotechnology industries have long faced significant challenges in sourcing high-purity pentose sugars like D-ribulose due to the prohibitive costs associated with traditional extraction and chemical synthesis methods. Patent CN110452942B introduces a groundbreaking approach to this problem by detailing a highly efficient preparation method for D-ribulose utilizing an immobilized enzyme catalysis system. This technology leverages a sophisticated multi-enzyme cascade that converts inexpensive D-gluconic acid directly into D-ribulose through a series of continuous biological transformations. By integrating enzyme immobilization techniques with a novel coenzyme regeneration cycle, this process not only achieves high conversion rates but also addresses the critical economic barriers that have historically restricted the widespread application of ribulose derivatives in drug development and metabolic engineering.
Furthermore, the strategic design of this metabolic pathway allows for the complete utilization of the starting material, minimizing waste and maximizing the yield of the target ketose. The patent explicitly outlines a workflow where multiple enzymes, including D-Gluconate Dehydrogenase (GDH), Ketoacid Isomerase (GIS), Ketoacid Kinase (GK), and Ketoacid Decarboxylase (GDC), work in concert to drive the reaction forward. This represents a paradigm shift from step-wise chemical synthesis, which often suffers from low overall yields due to the accumulation of losses at each protection and deprotection stage. For R&D directors and process chemists, this biological route offers a compelling alternative that aligns with green chemistry principles while delivering the structural complexity required for advanced pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of ribulose has been hindered by the inherent difficulties in manipulating carbohydrate chemistry through purely synthetic means. Traditional chemical synthesis of sugar molecules requires rigorous control over multiple chiral centers, necessitating a lengthy sequence of selective hydroxyl protection and deprotection reactions. Each of these steps introduces additional reagents, solvents, and purification requirements, which cumulatively drive up the production cost and extend the manufacturing timeline significantly. Moreover, separating the desired ketose from other similar carbohydrate byproducts generated during these reactions is notoriously difficult due to their high water solubility and structural similarity, often resulting in poor final purity and substantial material loss. These factors make conventional chemical routes economically unviable for large-scale applications, particularly when the target molecule is needed as a bulk intermediate for further biosynthesis.
The Novel Approach
In stark contrast, the method disclosed in patent CN110452942B circumvents these stereochemical challenges by harnessing the innate specificity of biocatalysts. The novel approach employs a one-pot cascade reaction where D-gluconic acid is continuously converted into 2-oxo-D-gluconic acid, then to 3-oxo-D-gluconic acid-6-phosphate, and finally to D-ribulose-5-phosphate before dephosphorylation. This seamless integration of four distinct enzymatic activities ensures that intermediates are consumed as soon as they are formed, effectively pushing the equilibrium towards the final product. By immobilizing these enzymes on a solid support, the process gains the added advantage of catalyst reusability, which is a critical factor for industrial feasibility. This biological strategy transforms a complex, multi-step synthetic nightmare into a streamlined, single-vessel operation that is both robust and scalable.
Mechanistic Insights into Multi-Enzyme Cascade and Coenzyme Regeneration
The core of this technological breakthrough lies in the intricate coordination of the enzymatic machinery and the intelligent management of energy cofactors. The reaction begins with the oxidation of D-gluconic acid by GDH, a step that consumes NAD+ and produces NADH. To prevent the depletion of this expensive cofactor, the system incorporates Lactate Dehydrogenase (LDH) and pyruvate, which oxidize the accumulated NADH back to NAD+, thereby creating a closed loop for nicotinamide regeneration. Simultaneously, the phosphorylation step catalyzed by GK requires ATP, which is regenerated in situ by Polyphosphate Kinase (PPK) using inexpensive polyphosphate as a phosphate donor. This dual regeneration system is pivotal, as it eliminates the need for stoichiometric amounts of ATP and NAD+, reducing the raw material cost to a fraction of what would be required in a non-regenerative system.
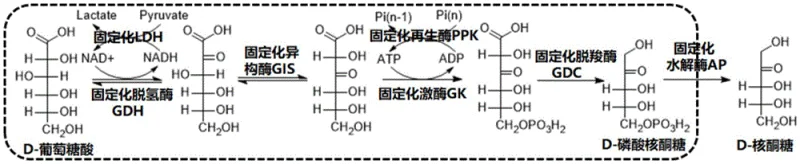
Following the initial oxidation and isomerization, the pathway proceeds through a kinase-mediated phosphorylation and a decarboxylation event catalyzed by GDC, which releases carbon dioxide and yields D-ribulose-5-phosphate. The final step involves the hydrolysis of the phosphate group by a phosphohydrolase (AP) to release free D-ribulose. The patent emphasizes the importance of optimizing the ratio of these enzymes, noting that the GDH reaction is equilibrium-limited and thus requires a higher loading compared to the more facile decarboxylation step. Understanding these kinetic nuances is essential for R&D teams aiming to replicate or optimize this process, as the balance between enzyme activities directly influences the accumulation of intermediates and the overall space-time yield of the reactor. The immobilization on epoxy resin further stabilizes these proteins, allowing them to withstand operational stresses better than their free counterparts.
How to Synthesize D-Ribulose Efficiently
The synthesis of D-ribulose via this immobilized enzyme method represents a significant advancement in biocatalytic process design, offering a clear roadmap for transitioning from bench-scale discovery to pilot production. The protocol involves the initial expression and purification of seven distinct enzymes, followed by their immobilization onto an epoxy resin matrix to create a robust heterogeneous catalyst system. This setup allows for the continuous conversion of cheap D-gluconic acid into the valuable sugar product under mild aqueous conditions. For detailed procedural instructions regarding gene amplification, enzyme immobilization ratios, and specific reaction parameters such as pH and temperature control, please refer to the standardized synthesis guide provided below.
- Amplify and express genes for GDH, GIS, GK, GDC, LDH, PPK, and AP in E. coli, followed by purification and immobilization on epoxy resin.
- Conduct a one-pot cascade reaction converting D-gluconic acid to D-ribulose-5-phosphate using the immobilized mixed enzyme system with ATP and NAD+ regeneration.
- Dephosphorylate D-ribulose-5-phosphate using immobilized phosphohydrolase (AP) and purify the final D-ribulose product via ion exchange and desalting.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this immobilized enzyme technology presents a compelling value proposition centered on cost efficiency and supply security. The primary driver of cost reduction is the substitution of expensive, petrochemical-derived starting materials with readily available and low-cost D-gluconic acid. Furthermore, the implementation of the coenzyme regeneration system effectively removes the financial burden of purchasing stoichiometric quantities of high-value cofactors like ATP and NAD+, which are typically major cost drivers in enzymatic processes. By enabling the reuse of immobilized enzymes through simple filtration, the process significantly lowers the recurring cost of biocatalysts, transforming what was once a disposable expense into a durable capital asset that can be utilized over multiple production batches.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior to chemical synthesis because it eliminates the need for hazardous organic solvents and complex purification trains associated with protecting group chemistry. The ability to run the reaction in a single pot with high conversion efficiency means that downstream processing is simplified, requiring fewer unit operations to isolate the final product. This reduction in process complexity translates directly into lower utility consumption, reduced waste disposal costs, and a smaller physical footprint for the manufacturing facility, all of which contribute to a substantially improved margin profile for the final D-ribulose product.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on fermentation-derived enzymes and abundant carbohydrate feedstocks mitigates the risk of raw material shortages that often plague synthetic organic chemistry. The robustness of the immobilized enzyme system ensures consistent product quality and batch-to-batch reproducibility, which is critical for maintaining uninterrupted supply to downstream pharmaceutical customers. Additionally, the mild reaction conditions reduce the safety risks associated with high-pressure or high-temperature operations, ensuring that production schedules are not disrupted by safety incidents or regulatory compliance issues related to hazardous chemical handling.
- Scalability and Environmental Compliance: The scalability of this biocatalytic route is inherently high, as enzyme reactors can be easily scaled from liters to cubic meters without significant changes to the reaction kinetics. The process generates minimal hazardous waste, primarily consisting of biodegradable protein residues and benign salts, which simplifies environmental compliance and wastewater treatment. This alignment with green chemistry principles not only reduces the environmental impact but also future-proofs the supply chain against increasingly stringent global regulations on chemical manufacturing emissions and solvent usage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this immobilized enzyme technology for D-ribulose production. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations. Stakeholders are encouraged to review these points to assess the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does the immobilized enzyme method reduce the cost of D-ribulose production compared to chemical synthesis?
A: The method utilizes cheap D-gluconic acid as a starting material and employs a coenzyme regeneration system (LDH/PPK) that drastically reduces the consumption of expensive cofactors like ATP and NAD+, eliminating the need for complex chemical protection and deprotection steps.
Q: What represents the key innovation in the coenzyme regeneration system described in patent CN110452942B?
A: The innovation lies in the simultaneous introduction of Lactate Dehydrogenase (LDH) with pyruvate for NAD+ regeneration and Polyphosphate Kinase (PPK) with polyphosphate for ATP regeneration, allowing the costly cofactors to cycle continuously within the reaction system.
Q: Is this enzymatic process scalable for industrial manufacturing of pharmaceutical intermediates?
A: Yes, the use of immobilized enzymes allows for easy recovery and reuse of the biocatalysts (maintaining 50-75% activity after recovery), and the mild reaction conditions (25-40°C) facilitate safe and efficient scale-up from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Ribulose Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the immobilized enzyme catalysis method described in patent CN110452942B for the production of high-value sugar intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to meet stringent purity specifications required by the global pharmaceutical industry while maintaining the highest standards of quality assurance and regulatory compliance.
We invite you to collaborate with our technical team to explore how this cost-effective biocatalytic route can enhance your supply chain for D-ribulose and its derivatives. By leveraging our expertise in enzyme engineering and process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and economical supply of this critical pharmaceutical intermediate.
