Advanced D-Arabinose Production: Technical Breakthroughs and Commercial Scalability for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for synthesizing critical carbohydrate derivatives, and patent CN101289439B presents a significant advancement in the preparation of D-arabinose. This specific intellectual property outlines a novel two-step synthetic route that addresses the longstanding challenges of high cost and operational complexity associated with traditional glycal synthesis. By leveraging a zinc-mediated reductive elimination followed by a mild deacetylation process, the technology offers a streamlined pathway that is particularly attractive for the production of high-purity pharmaceutical intermediate. The strategic shift away from expensive transition metal catalysts towards more abundant zinc reagents represents a fundamental improvement in process chemistry that aligns with modern green chemistry principles. For R&D Directors and Procurement Managers evaluating supply chain resilience, this patent provides a compelling framework for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality. The implications of this technology extend beyond mere academic interest, offering tangible benefits for commercial scale-up of complex carbohydrate derivatives in a regulated manufacturing environment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arabinose and related glycal structures has been plagued by reliance on cumbersome and environmentally hazardous reagents that complicate the manufacturing workflow. Prior art, such as methods utilizing chromium-based catalysts or complex substitution reactions on xylose isomers, often suffers from low overall yields and requires rigorous purification steps to remove toxic metal residues. These conventional approaches frequently involve harsh reaction conditions that can degrade sensitive sugar moieties, leading to a broad and difficult-to-manage impurity profile that jeopardizes downstream application suitability. Furthermore, the high cost of specialized reagents and the necessity for extensive waste treatment protocols create significant economic barriers for producers aiming for cost reduction in pharmaceutical intermediate manufacturing. The operational risks associated with handling toxic heavy metals also impose strict regulatory burdens on facilities, potentially disrupting supply continuity and increasing the lead time for high-purity sugar intermediates. Consequently, the industry has long needed a more efficient alternative that balances chemical efficacy with economic and environmental sustainability.
The Novel Approach
The methodology disclosed in the patent introduces a transformative strategy by utilizing 1-bromoacetylated arabinose as a starting material in a zinc powder and sodium dihydrogen phosphate system. This innovative route operates under mild conditions, typically at room temperature, which significantly reduces energy consumption and minimizes the thermal degradation of the product. The use of acetone and water as solvent systems not only lowers material costs but also simplifies the workup procedure, allowing for easier separation of the organic phase from inorganic byproducts. By avoiding the use of expensive and toxic chromium reagents found in older literature, this process drastically simplifies the purification workflow and enhances the safety profile of the manufacturing plant. The result is a high-yield transformation that produces D-acetylated arabinose with exceptional efficiency, setting the stage for a seamless subsequent deacetylation step. This approach exemplifies how modern process chemistry can overcome the limitations of legacy methods to deliver a superior value proposition for global supply chains.
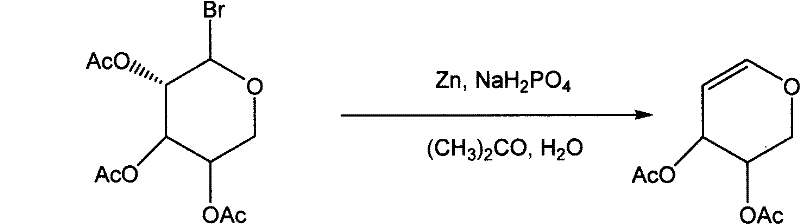
Mechanistic Insights into Zinc-Mediated Reductive Elimination
The core of this technological breakthrough lies in the precise mechanistic interaction between the zinc powder and the bromo-substituted sugar derivative within the phosphate-buffered acetone medium. The zinc acts as a reducing agent that facilitates the elimination of the bromine atom, triggering the formation of the double bond characteristic of the glycal structure without compromising the stereochemical integrity of the adjacent chiral centers. The presence of sodium dihydrogen phosphate serves a critical role in buffering the reaction environment, preventing unwanted side reactions that could arise from acidic or basic conditions during the reductive phase. This careful control of the reaction milieu ensures that the formation of D-acetylated arabinose proceeds with high selectivity, minimizing the generation of regioisomers or degradation products that often plague carbohydrate chemistry. For technical teams, understanding this mechanism is vital for troubleshooting and optimizing the process during technology transfer, as it highlights the importance of reagent quality and mixing efficiency. The robustness of this mechanistic pathway suggests that it can be reliably reproduced across different scales, from laboratory benchtop to multi-ton commercial reactors.
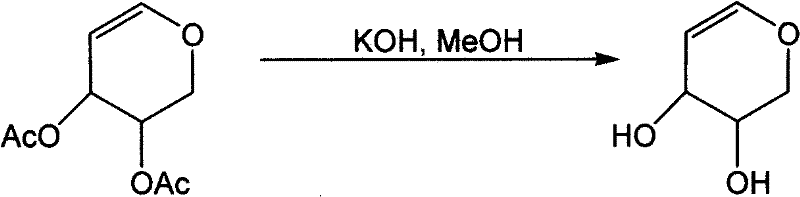
Following the initial reduction, the subsequent deacetylation step employs mild basic conditions to remove the acetyl protecting groups and reveal the free hydroxyl functionalities of the final D-arabinose product. The patent specifies the use of catalysts such as potassium carbonate or potassium hydroxide in alcoholic solvents like methanol or ethanol, which provides a gentle yet effective means of cleaving the ester bonds. This step is crucial for achieving the desired chemical structure while maintaining the stability of the enol ether double bond, which can be sensitive to harsh hydrolysis conditions. The rapid reaction time of merely 10 to 30 minutes at room temperature indicates a highly favorable kinetic profile, allowing for high throughput processing in a manufacturing setting. By controlling the stoichiometry of the base and the solvent ratio, producers can fine-tune the reaction to ensure complete deprotection without inducing decomposition, thereby securing a high-purity D-arabinose output. This level of control over the chemical transformation is essential for meeting the stringent purity specifications required by downstream pharmaceutical applications.
How to Synthesize D-Arabinose Efficiently
Implementing this synthesis route requires a disciplined approach to reagent preparation and process monitoring to fully realize the efficiency gains promised by the patent data. The procedure begins with the careful suspension of zinc powder and sodium dihydrogen phosphate in acetone, followed by the controlled addition of the bromo-sugar substrate and water to initiate the reductive elimination. Operators must maintain strict adherence to the specified mass ratios and stirring times to ensure complete conversion, as verified by thin-layer chromatography (TLC) analysis before proceeding to the workup phase. Once the intermediate is isolated and purified, it is dissolved in an alcoholic solvent and treated with a base catalyst to effect deacetylation, followed by standard extraction and chromatographic purification techniques. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety precautions.
- Prepare a reaction system containing zinc powder and sodium dihydrogen phosphate in acetone, then add 1-bromoacetylated arabinose and water to generate D-acetylated arabinose.
- Dissolve the resulting D-acetylated arabinose in methanol or ethanol and add a base catalyst such as potassium carbonate to facilitate deacetylation.
- Monitor the reaction via TLC, filter the mixture, and purify the organic phase through silica gel column chromatography to isolate the final D-arabinose product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, the adoption of this patented process offers substantial strategic benefits that extend well beyond the laboratory bench. The elimination of expensive transition metal catalysts and the use of commodity solvents like acetone and methanol directly translate to significant cost savings in pharmaceutical intermediate manufacturing without compromising product quality. The simplified workflow reduces the number of unit operations required, which in turn lowers labor costs and minimizes the potential for human error during production runs. Furthermore, the reduced environmental footprint associated with avoiding toxic heavy metals simplifies waste disposal compliance and lowers the overall cost of ownership for the manufacturing facility. These factors combine to create a more resilient and cost-effective supply chain capable of withstanding market fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The substitution of costly chromium reagents with abundant zinc powder fundamentally alters the cost structure of the synthesis, removing a major expense driver from the bill of materials. Additionally, the ability to operate at room temperature eliminates the need for energy-intensive heating or cooling systems, further driving down utility costs associated with production. The high yield reported in the patent examples implies less raw material waste per kilogram of product, maximizing the return on investment for every batch processed. These cumulative efficiencies allow for a more competitive pricing structure while maintaining healthy margins for the producer.
- Enhanced Supply Chain Reliability: By relying on readily available and stable reagents, the risk of supply disruptions due to raw material scarcity is significantly mitigated compared to methods requiring specialized or regulated chemicals. The robustness of the reaction conditions means that the process is less sensitive to minor variations in environmental factors, ensuring consistent output quality across different production batches and facilities. This reliability is critical for maintaining continuous supply to downstream customers who depend on just-in-time delivery models for their own manufacturing schedules. Consequently, partners can expect a more stable and predictable supply of high-purity D-arabinose to support their long-term production planning.
- Scalability and Environmental Compliance: The use of common solvents and the absence of toxic heavy metals make this process inherently easier to scale from pilot plant to full commercial production without major engineering redesigns. The reduced generation of hazardous waste simplifies the environmental permitting process and lowers the liability associated with chemical handling and disposal. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile of the manufacturing organization. Such environmental stewardship is increasingly becoming a key criterion for selection by global pharmaceutical companies seeking responsible partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific D-arabinose synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and technical evaluators. Understanding these details is crucial for making informed decisions about integrating this route into existing production portfolios or sourcing strategies.
Q: What are the primary advantages of this zinc-mediated method over chromium-based prior art?
A: The zinc-mediated method eliminates the use of toxic and expensive chromium reagents, significantly reducing environmental waste and raw material costs while simplifying the purification process.
Q: Is this process suitable for large-scale commercial production of carbohydrate intermediates?
A: Yes, the process utilizes common solvents like acetone and methanol and operates at room temperature, making it highly scalable and safe for industrial manufacturing environments.
Q: How does this method impact the purity profile of the final D-arabinose intermediate?
A: By avoiding complex transition metal catalysts, the method reduces the risk of heavy metal contamination, resulting in a cleaner impurity profile that meets stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Arabinose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex carbohydrate derivatives like D-arabinose in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical industrial setting. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the exacting standards required for drug substance manufacturing. Our infrastructure is designed to support the rapid technology transfer and optimization necessary to bring this innovative process to market speed.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain and reduce overall project costs. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability as your trusted partner in fine chemical manufacturing.
