Advanced Synthesis of Diazafluorenone and Spirobifluorene Derivatives for Commercial OLED Applications
Advanced Synthesis of Diazafluorenone and Spirobifluorene Derivatives for Commercial OLED Applications
The rapid evolution of the organic electronics sector demands materials that combine exceptional thermal stability with efficient charge transport properties. Patent CN102002003A introduces a groundbreaking class of aryl-substituted 3,4-diazafluorenone, fluorene, and spirobifluorene derivatives designed specifically to meet these rigorous standards. These compounds are characterized by the strategic introduction of diverse aryl substituent groups at the 2-position of the 3,4-diazafluorene and spirobifluorene cores. The innovation lies not only in the molecular architecture but also in the streamlined preparation method, which employs condensation reactions between acetyl-substituted aromatic compounds, ninhydrin, and hydrazine hydrate. This approach facilitates the formation of various 2-aryl-substituted 3,4-diazafluorenone derivatives through a robust ring-closure mechanism. Furthermore, the patent outlines subsequent reduction steps using hydrazine hydrate or reactions with lithium salts and Grignard reagents to generate tertiary alcohol intermediates,最终 leading to the final acidification and ring closure. These materials are pivotal for organic semiconductor devices, offering high glass transition temperatures that ensure device longevity.
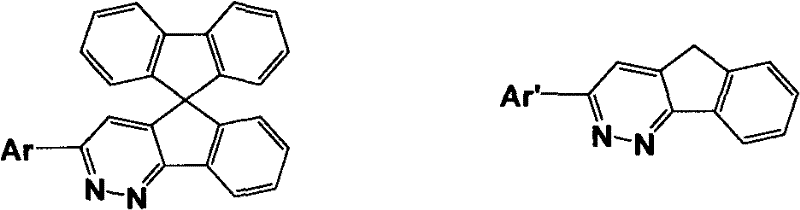
For R&D directors and procurement specialists seeking a reliable OLED material supplier, understanding the structural versatility of these derivatives is crucial. The ability to modulate the electronic properties by varying the aryl groups allows for fine-tuning of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) levels. This tunability is essential for optimizing the efficiency of organic light-emitting diodes and organic electrical memory devices. The synthesis route described provides a scalable pathway to access these high-performance materials, addressing the industry's need for cost reduction in organic semiconductor manufacturing while maintaining stringent purity specifications required for commercial display applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing complex heterocyclic systems like diazafluorenones often suffer from significant drawbacks that hinder their commercial viability. Conventional methods frequently rely on multi-step sequences involving harsh reaction conditions, expensive transition metal catalysts, and difficult-to-remove impurities that compromise the electronic performance of the final material. Many prior art processes require tedious purification protocols, such as repeated column chromatography or sublimation, which drastically increase production costs and reduce overall yield. Additionally, the lack of thermal stability in some conventional fluorene derivatives limits their application in high-temperature processing environments common in vacuum deposition techniques. The inability to easily introduce diverse functional groups at specific positions without protecting group strategies further complicates the synthesis, leading to longer lead times and supply chain bottlenecks for high-purity organic semiconductors.
The Novel Approach
The methodology disclosed in CN102002003A represents a paradigm shift by utilizing a direct condensation strategy that bypasses many of these historical inefficiencies. By reacting acetyl-substituted aromatic hydrocarbons directly with ninhydrin and 85% hydrazine hydrate in glacial acetic acid, the process achieves ring closure in a single pot under reflux conditions for 3 to 4 hours. This eliminates the need for exotic catalysts and reduces the number of isolation steps. The versatility of this approach is demonstrated by the wide range of aryl groups that can be incorporated, as illustrated in the specific examples provided in the patent documentation. The subsequent conversion to spirobifluorene derivatives via Grignard addition or reduction followed by acidification offers a modular platform for generating a library of materials. This streamlined workflow not only enhances the commercial scale-up of complex polymer additives and small molecule semiconductors but also ensures a consistent supply of high-quality intermediates for downstream device fabrication.
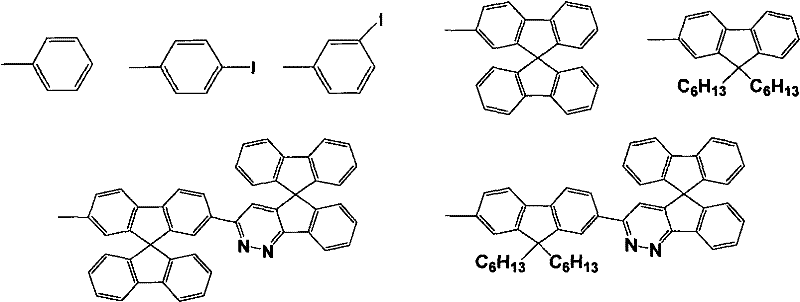
Mechanistic Insights into Condensation and Cyclization Reactions
The core of this synthetic innovation lies in the mechanistic pathway of the condensation reaction between the ketone functionality of the acetyl-substituted aromatic compound and the carbonyl groups of ninhydrin. In the presence of hydrazine hydrate, a nucleophilic attack occurs, leading to the formation of a hydrazone intermediate. This intermediate subsequently undergoes an intramolecular cyclization facilitated by the acidic environment of the glacial acetic acid solvent. The reflux temperature provides the necessary activation energy to drive the dehydration steps required to form the stable pyridazine ring system characteristic of the 3,4-diazafluorenone core. The use of a 1:1:1.5 molar ratio of acetyl compound to ninhydrin to hydrazine ensures complete conversion while minimizing side reactions. This precise stoichiometric control is critical for maintaining high purity, as unreacted starting materials can act as quenchers in electroluminescent devices. The mechanism effectively locks the molecular conformation, contributing to the observed high glass transition temperatures.
Following the formation of the diazafluorenone core, the transformation into spirobifluorene derivatives involves a sophisticated organometallic or reduction pathway. When utilizing Grignard reagents derived from 2-bromobiphenyl, the nucleophilic carbon attacks the carbonyl carbon of the diazafluorenone, generating a tertiary alcohol intermediate. This step is sensitive to moisture and requires an inert atmosphere, typically nitrogen protection, to prevent reagent decomposition. Alternatively, reduction with hydrazine hydrate in polyethylene glycol converts the ketone to a methylene group, yielding the 3,4-diaza-9H-fluorene structure. The final acidification step promotes the dehydration of the tertiary alcohol or the cyclization of the reduced intermediate, locking the spiro-junction or completing the fluorene ring system. Understanding these mechanistic nuances allows process chemists to optimize reaction parameters such as temperature, solvent choice, and addition rates to maximize yield and minimize impurity profiles, ensuring the material meets the rigorous standards of the electronic chemical industry.
How to Synthesize 2-Aryl-Substituted 3,4-Diazafluorenone Efficiently
The synthesis of these high-value organic semiconductors begins with the careful selection of high-purity starting materials, specifically the acetyl-substituted aromatic compounds and ninhydrin. The reaction is typically conducted in glacial acetic acid, which serves as both the solvent and a mild acid catalyst to promote the condensation. The mixture is heated to reflux for a duration of 3 to 4 hours, allowing the system to reach thermodynamic equilibrium and ensuring complete consumption of the limiting reagent. Upon cooling, the addition of hydrazine hydrate induces precipitation of the crude product, which can be isolated via simple filtration. This operational simplicity is a key advantage for scaling operations. For detailed procedural specifics regarding stoichiometry, workup, and purification techniques such as column chromatography or recrystallization, please refer to the standardized synthesis guidelines provided below.
- Condense acetyl-substituted aromatic compounds with ninhydrin and hydrazine hydrate in glacial acetic acid under reflux.
- Reduce the resulting 3,4-diazafluorenone intermediates using hydrazine hydrate or react with Grignard reagents to form tertiary alcohols.
- Perform acidification and ring closure to finalize the 2-aryl substituted 3,4-diazafluorene and spirobifluorene derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits over legacy manufacturing processes. The primary advantage stems from the use of commodity chemicals such as acetophenones, ninhydrin, and hydrazine hydrate, which are readily available in the global market at competitive price points. This reliance on bulk chemicals mitigates the risk of supply disruptions often associated with specialized or proprietary reagents. Furthermore, the reaction conditions are relatively mild, utilizing standard glass-lined reactors capable of handling acetic acid reflux, which reduces the capital expenditure required for specialized high-pressure or cryogenic equipment. The simplified workup procedure, often involving filtration and washing rather than complex distillations, translates to shorter batch cycles and increased throughput. These factors collectively contribute to a more resilient and cost-effective supply chain for organic electronic materials.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive solvents like glacial acetic acid significantly lower the raw material costs per kilogram of product. The high atom economy of the condensation reaction minimizes waste generation, reducing disposal costs and environmental compliance burdens. Additionally, the ability to isolate products via filtration rather than energy-intensive distillation or chromatography on a large scale leads to substantial operational savings. These efficiencies allow for a more competitive pricing structure for high-purity OLED materials without compromising on quality or performance metrics required by end-users.
- Enhanced Supply Chain Reliability: By utilizing widely sourced starting materials, manufacturers can diversify their supplier base, reducing dependency on single-source vendors for critical intermediates. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or pressure, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major display manufacturers. The simplified purification steps also reduce the lead time for high-purity organic semiconductors, enabling faster response to market demand surges.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-ton commercial production. The use of acetic acid, while requiring proper handling, is a well-understood solvent in the chemical industry with established recovery and recycling protocols. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes for new manufacturing facilities. The high thermal stability of the final products also implies safer handling and storage conditions, further enhancing the overall safety profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these diazafluorene derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. These insights are intended to clarify the feasibility of integrating these materials into existing supply chains and device architectures.
Q: What are the key thermal properties of these diazafluorene derivatives?
A: The synthesized fluorene and azaspirobifluorene structural units exhibit high thermal stability and high glass transition temperatures, making them ideal for durable OLED devices.
Q: How does this synthesis method improve upon conventional routes?
A: This method utilizes a direct condensation and cyclization strategy using readily available reagents like ninhydrin and hydrazine hydrate, significantly simplifying the reaction route compared to multi-step traditional syntheses.
Q: Can these materials be scaled for industrial production?
A: Yes, the synthesis involves standard reflux conditions in glacial acetic acid and straightforward purification steps like filtration and recrystallization, which are highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl-Substituted 3,4-Diazafluorenone Supplier
As the demand for advanced organic electronic materials continues to surge, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for diazafluorenone synthesis, including acid-resistant reactors and advanced purification suites. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards of the optoelectronic industry. Our commitment to quality assurance ensures that the materials you receive are free from detrimental impurities that could affect device performance.
We invite you to collaborate with our technical team to explore the full potential of these novel derivatives for your specific applications. Whether you require custom synthesis modifications or large-scale procurement, we are prepared to provide a Customized Cost-Saving Analysis tailored to your project requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your product development cycle and secure a competitive edge in the rapidly evolving landscape of organic semiconductor manufacturing.
