Advanced Synthesis of Non-Conjugated Fluorene-Triphenylamine Derivatives for High-Performance OLED Manufacturing
The rapid evolution of the organic electronics sector demands materials that not only exhibit superior optoelectronic properties but also possess robust thermal stability and processability. Patent CN101747208A introduces a groundbreaking class of non-conjugated derivative materials featuring a fluorene-triphenylamine structure, specifically designed to address the limitations of traditional conjugated systems in OLED applications. This technology leverages a unique molecular architecture where the fluorene and triphenylamine units are linked via an sp3 hybridized carbon at the 9-position of the fluorene ring. This structural modification is pivotal as it independently adjusts energy levels while ensuring excellent matching with the work function of anode materials, ultimately leading to devices with high heat and light stability. For industry leaders seeking a reliable electronic chemical supplier, understanding the nuances of this synthesis is critical for securing a competitive edge in the display and lighting markets.
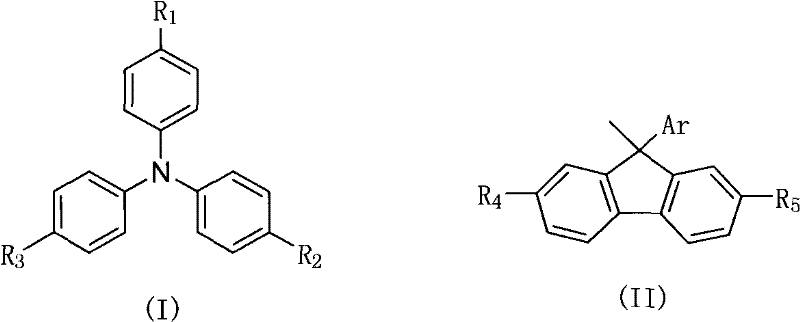
Traditional luminescent materials, particularly those based on rigid planar biphenyl structures like pure fluorene, often suffer from significant drawbacks during device operation. The primary issue lies in the tendency of these conjugated systems to form intermolecular excimers or aggregates in the solid state, which results in undesirable long-wave emission and severely compromises the saturation color purity and efficiency of the device. Furthermore, the energy levels of many conventional conjugated derivatives do not align well with the work functions of the device electrodes, causing imbalances in hole and electron injection. In contrast, the novel approach detailed in this patent utilizes a non-conjugated design that effectively suppresses these aggregation phenomena. By introducing bulky triphenylamine groups at the 9-position, the material maintains high fluorescence performance and photostability while avoiding the crystallization issues that plague simpler fluorene derivatives. This strategic molecular engineering ensures that the material serves not just as a luminescent emitter but also as a superior host or transport material.
Mechanistic Insights into Acid-Catalyzed Friedel-Crafts Alkylation
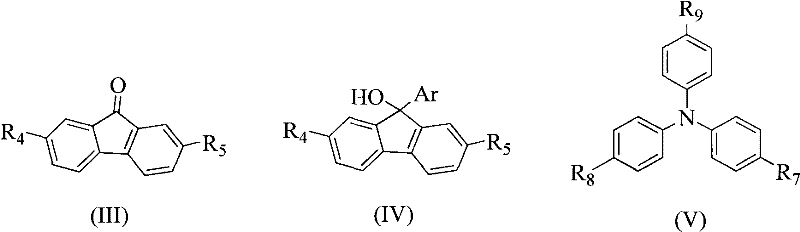
The core of this synthesis lies in a sophisticated acid-catalyzed condensation reaction, effectively a Friedel-Crafts alkylation, which couples the 9-aryl-9-hydroxyfluorene intermediate with a triphenylamine derivative. The mechanism initiates with the protonation of the hydroxyl group on the fluorene intermediate by a strong acid catalyst, such as trifluoromethanesulfonic acid or boron trifluoride etherate. This protonation facilitates the departure of a water molecule, generating a highly reactive carbocation intermediate at the 9-position of the fluorene ring. This carbocation is resonance-stabilized by the adjacent aromatic rings but remains sufficiently electrophilic to attack the electron-rich aromatic rings of the triphenylamine nucleophile. The reaction proceeds through an electrophilic aromatic substitution pathway, where the triphenylamine attacks the carbocation, followed by deprotonation to restore aromaticity and form the final C-C bond. This mechanism is highly advantageous as it avoids the use of expensive palladium or nickel catalysts typically required for C-N or C-C cross-coupling, significantly simplifying the purification process and reducing metal contamination risks in the final electronic material.
Controlling impurities in this synthesis is paramount for achieving the high purity standards required in organic semiconductor manufacturing. The reaction conditions are carefully optimized to minimize side reactions such as polymerization or over-alkylation. The use of specific solvents like 1,4-dioxane or dichloromethane helps in solubilizing both the hydrophobic fluorene intermediates and the polar acid catalysts, ensuring a homogeneous reaction environment that promotes selective mono- or di-substitution as desired. Post-reaction workup involves neutralization with saturated sodium bicarbonate or carbonate solutions to quench the strong acid, followed by extraction and rigorous drying. The final purification is typically achieved through column chromatography using silica gel with petroleum ether and ethyl acetate mixtures, or via recrystallization from solvents like ethanol or hexane. This multi-stage purification strategy ensures that trace amounts of unreacted starting materials, acid residues, or oligomeric byproducts are removed, yielding a product with the stringent purity specifications necessary for high-performance OLED layers.
How to Synthesize Fluorene-Triphenylamine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a robust and scalable pathway for producing these advanced materials, making it highly attractive for commercial adoption. The process begins with the preparation of a Grignard reagent from an aryl halide and magnesium powder in anhydrous ether or tetrahydrofuran, which is then reacted with 9-fluorenone to generate the crucial 9-aryl-9-hydroxyfluorene intermediate. This intermediate is subsequently coupled with a triphenylamine derivative under acidic conditions to form the final target molecule. The detailed standardized synthesis steps below provide a comprehensive guide for replicating this high-yield process in a laboratory or pilot plant setting, ensuring consistency and quality control throughout the production batch.
- Prepare Grignard reagent from aryl halide and magnesium powder in anhydrous ether or THF, then react with 9-fluorenone to form 9-aryl-9-hydroxyfluorene.
- Mix the 9-aryl-9-hydroxyfluorene intermediate with a triphenylamine derivative in an organic solvent such as dichloromethane or dioxane.
- Add an acid catalyst like trifluoromethanesulfonic acid and stir at 10-80°C to effect dehydration and coupling, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers substantial strategic benefits beyond mere technical performance. The process relies on commodity chemicals such as fluorenone, aryl halides, and triphenylamine, which are widely available in the global chemical market, thereby mitigating supply chain risks associated with scarce or specialized reagents. The elimination of precious metal catalysts not only reduces the direct material cost but also simplifies the downstream processing requirements, as there is no need for complex metal scavenging steps to meet ppm-level impurity limits. This streamlined workflow translates directly into faster turnaround times and more predictable production schedules, allowing manufacturers to respond agilely to market demands for new display technologies without being bottlenecked by complex synthesis logistics.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive starting materials and the avoidance of costly transition metal catalysts. Traditional cross-coupling methods often require palladium complexes and specialized ligands, which represent a significant portion of the raw material cost. By utilizing a Brønsted or Lewis acid-catalyzed condensation, the process drastically lowers the cost of goods sold. Furthermore, the high yields reported in the examples, often exceeding 70-80% after purification, mean less waste and higher throughput per batch. The simplicity of the workup procedure, involving basic aqueous washes and standard chromatography, further reduces operational expenditures related to solvent consumption and labor hours, delivering significant cost savings in electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like 9-fluorenone and common aryl halides ensures a resilient supply chain. Unlike air-sensitive organometallic reagents that require strict storage conditions and immediate use, the precursors for this synthesis can be stocked in bulk, buffering against market volatility. The reaction conditions are also relatively mild, operating between 10°C and 80°C, which reduces the energy burden on manufacturing facilities and allows for the use of standard glass-lined or stainless steel reactors without the need for extreme cryogenic or high-pressure equipment. This operational flexibility enhances the reliability of supply, ensuring that production timelines are met consistently even during periods of high demand or logistical constraints.
- Scalability and Environmental Compliance: Scaling this synthesis from gram to kilogram or ton scale is straightforward due to the absence of exothermic hazards associated with some metal-catalyzed reactions. The acid catalysts used, such as trifluoromethanesulfonic acid, can be managed with standard safety protocols, and the aqueous waste streams generated during neutralization are easier to treat compared to heavy metal-containing waste. This aligns well with increasingly stringent environmental regulations in the chemical industry. The ability to produce high-purity materials with a lower environmental footprint makes this process not only commercially attractive but also sustainable, supporting the long-term goals of green chemistry in the specialty chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorene-triphenylamine derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for stakeholders evaluating this technology for integration into their supply chains. Understanding these details is essential for making informed decisions about material sourcing and process implementation.
Q: Why is the sp3 hybridization at the 9-position of fluorene beneficial for OLED materials?
A: The sp3 hybridized carbon at the 9-position breaks the conjugation between the fluorene and triphenylamine units. This non-conjugated structure effectively inhibits the formation of intermolecular excimers and aggregates, which are common causes of long-wave emission and reduced color purity in traditional conjugated systems. Furthermore, this structural feature significantly enhances the thermal stability and amorphous film-forming properties of the material.
Q: What are the key advantages of the acid-catalyzed condensation method described in the patent?
A: The acid-catalyzed condensation method utilizes readily available raw materials such as 9-fluorenone and triphenylamine derivatives, avoiding the need for expensive transition metal catalysts often required in cross-coupling reactions. The reaction conditions are relatively mild, typically ranging from 10°C to 80°C, and the process yields high-purity products after simple workup procedures like washing and column chromatography, making it highly suitable for cost-effective industrial scale-up.
Q: How does this material improve charge injection in organic electronic devices?
A: The incorporation of triphenylamine units provides excellent hole-transporting capabilities due to their low ionization potential and high hole mobility. When combined with the fluorene backbone, the resulting derivative exhibits energy levels that are well-matched with the work function of common anode materials. This alignment facilitates balanced injection of holes and electrons, thereby improving the overall luminous efficiency and operational stability of the organic electroluminescent device.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorene-Triphenylamine Derivatives Supplier
As the demand for high-efficiency OLED materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM is essential for navigating the complexities of commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to mass market deployment is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and thermal stability testing, guaranteeing that every batch meets the exacting standards required for next-generation display and lighting applications. We understand that consistency is key in the electronics industry, and our robust quality management systems are designed to deliver that reliability.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these advanced materials. Whether you require a Customized Cost-Saving Analysis for your current sourcing strategy or need specific COA data and route feasibility assessments for new derivatives, our experts are ready to assist. By leveraging our synthesis expertise and manufacturing capacity, you can secure a stable supply of high-performance fluorene-triphenylamine derivatives, enabling you to focus on innovation and product development while we handle the complexities of chemical production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
