Advanced Amine-Mediated Demethylation for Commercial Scale Production of Gastric Drug Intermediates
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical active pharmaceutical ingredient (API) intermediates, particularly those serving high-volume therapeutic areas like gastrointestinal disorders. A pivotal advancement in this domain is detailed in Chinese Patent CN103387494A, which discloses a novel and highly efficient method for the preparation of 2-hydroxy-4,5-dimethoxybenzoic acid. This compound serves as a vital building block for the synthesis of Acotiamide hydrochloride (Z-338), a potent gastric motility agent. The patent introduces a paradigm shift away from traditional, hazard-prone Lewis acid chemistry toward a safer, amine-mediated demethylation strategy. By utilizing linear or cyclic amines in polar aprotic solvents, this technology achieves exceptional regioselectivity and yield while drastically simplifying the operational complexity. For global supply chain leaders and R&D directors, this innovation represents a significant opportunity to optimize the manufacturing of complex aromatic intermediates, ensuring both economic viability and adherence to stringent environmental and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-hydroxy-4,5-dimethoxybenzoic acid from its trimethoxy precursor has relied heavily on harsh demethylation protocols involving strong Lewis acids. As illustrated in the reaction scheme below, conventional processes typically employ reagents such as boron trifluoride etherate (BF3·Et2O) or aluminum chloride (AlCl3) in the presence of alkali metal bromides or iodides. These traditional methodologies suffer from severe inherent drawbacks that hinder their suitability for modern large-scale industrial production. Boron trifluoride etherate is not only prohibitively expensive but also exhibits high volatility and instability, decomposing readily and posing significant risks to labor protection and operator safety. Furthermore, the use of metal-based Lewis acids generates substantial quantities of heavy metal waste, creating a burdensome environmental liability and necessitating complex, costly waste treatment infrastructure. The corrosive nature of these reagents also demands specialized, corrosion-resistant reactor materials, further inflating capital expenditure and maintenance costs for manufacturing facilities.
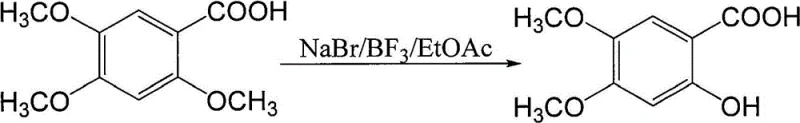
The Novel Approach
In stark contrast to the hazardous legacy methods, the technology disclosed in CN103387494A offers a streamlined and economically superior alternative by leveraging organic amines as the demethylating agents. This innovative approach involves reacting 2,4,5-trimethoxybenzoic acid with linear or cyclic amines, such as piperazine, morpholine, or piperidine, within a polar aprotic solvent system. The elimination of volatile Lewis acids and metal salts fundamentally alters the safety profile of the process, rendering it far more conducive to industrial scale-up. The use of stable, solid amines like piperazine simplifies handling and dosing operations, while the reaction proceeds with remarkable efficiency at elevated temperatures. This method not only circumvents the environmental and safety pitfalls associated with metal catalysts but also delivers high yields and excellent purity profiles. The transition to this amine-based chemistry signifies a move towards greener, more sustainable manufacturing practices that align with the evolving regulatory landscape of the global fine chemical sector.
Mechanistic Insights into Amine-Mediated Selective Demethylation
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the interaction between the cyclic amine and the methoxy substituents on the aromatic ring. In the presence of a high-boiling polar aprotic solvent such as N,N-dimethylacetamide (DMAc) or dimethyl sulfoxide (DMSO), the amine acts as a potent nucleophile. At reaction temperatures ranging from 130°C to 150°C, the amine attacks the methyl group of the methoxy functionality. While the precise electronic nuances are complex, the reaction demonstrates a profound preference for demethylation at the 2-position of the benzoic acid scaffold. This regioselectivity is critical, as it ensures the formation of the desired 2-hydroxy-4,5-dimethoxybenzoic acid without significant contamination from other isomeric byproducts. The polar aprotic solvent plays a dual role: it solubilizes the reactants effectively at high temperatures and likely stabilizes the transition state or the resulting ammonium salt intermediates, driving the equilibrium toward product formation. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as amine stoichiometry and thermal profiles, to maximize conversion rates.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed routes. Traditional Lewis acid methods often lead to side reactions, including ring halogenation or over-demethylation, due to the aggressive nature of the catalysts. In the amine-mediated process, the byproduct profile is significantly cleaner, primarily consisting of the corresponding methylated amine salts which are easily removed during the aqueous workup. The absence of metal ions eliminates the risk of metal-catalyzed degradation pathways that can compromise the stability of the final API intermediate. Consequently, the crude product obtained from this reaction requires less rigorous purification steps, such as extensive chromatography or recrystallization, to meet pharmaceutical grade specifications. This inherent purity advantage translates directly into reduced processing time and lower solvent consumption during the isolation phase, reinforcing the overall economic efficiency of the synthetic route.
How to Synthesize 2-Hydroxy-4,5-Dimethoxybenzoic Acid Efficiently
The practical implementation of this synthesis involves a straightforward protocol that balances reaction kinetics with operational simplicity. The process begins by suspending the starting material, 2,4,5-trimethoxybenzoic acid, and the selected amine reagent in the chosen polar aprotic solvent. Maintaining an inert atmosphere, typically using nitrogen flow, is recommended to prevent oxidative degradation of the reagents at high temperatures. The mixture is then heated to the optimal range of 130°C to 150°C, with 150°C often yielding the best results in terms of reaction rate and completion. Following the reaction period, the solvent is removed, and the residue is subjected to pH adjustment and filtration to isolate the target acid. For a comprehensive, step-by-step technical guide including specific molar ratios and workup details, please refer to the standardized procedure outlined below.
- Suspend 2,4,5-trimethoxybenzoic acid and a linear or cyclic amine (preferably piperazine) in a polar aprotic solvent such as N,N-dimethylacetamide.
- Heat the reaction mixture to a temperature between 130°C and 150°C under an inert nitrogen atmosphere and stir for 1 to 4 hours to effect selective demethylation.
- Remove the solvent via rotary evaporation, adjust the pH of the residue, stir overnight at room temperature, and filter to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this amine-mediated synthesis route presents a compelling value proposition centered on cost optimization and supply security. The shift away from specialized, hazardous reagents to commodity chemicals fundamentally reshapes the cost structure of manufacturing this key intermediate. By eliminating the dependency on expensive Lewis acids like boron trifluoride etherate, manufacturers can achieve substantial raw material cost savings. Additionally, the simplified waste stream, devoid of heavy metals, reduces the financial burden associated with environmental compliance and hazardous waste disposal. These factors combine to create a more resilient and cost-effective supply chain, capable of withstanding market fluctuations in specialty chemical pricing.
- Cost Reduction in Manufacturing: The replacement of high-cost Lewis acid catalysts with inexpensive cyclic amines like piperazine drives a significant decrease in direct material costs. Furthermore, the operational simplicity of the process reduces energy consumption and labor hours required for complex safety monitoring and waste neutralization. The ability to use standard stainless steel reactors instead of specialized corrosion-resistant equipment also lowers capital depreciation costs, contributing to a more favorable overall manufacturing margin.
- Enhanced Supply Chain Reliability: Sourcing stability is markedly improved as the key reagents—piperazine, DMAc, and the trimethoxy precursor—are widely available commodity chemicals with robust global supply networks. Unlike specialized Lewis acid complexes which may face supply bottlenecks or long lead times, these materials can be procured from multiple qualified vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The absence of volatile, toxic gases and heavy metal effluents simplifies the permitting process for new manufacturing lines and ensures compliance with increasingly strict environmental regulations. The high yield and selectivity reported in the patent data suggest that the process can be scaled from pilot plant to multi-ton production with minimal loss of efficiency, supporting the growing demand for gastric motility therapeutics globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the practical benefits of switching to this amine-based methodology. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value of this supply route.
Q: Why is the amine-mediated method superior to traditional Lewis acid demethylation?
A: Traditional methods utilize hazardous and expensive reagents like boron trifluoride etherate or aluminum chloride, which pose significant safety risks due to volatility and generate heavy metal waste. The novel amine-mediated process replaces these with stable, low-cost organic amines like piperazine, eliminating corrosive hazards and simplifying waste treatment.
Q: What represents the key cost-saving factor in this new synthesis route?
A: The primary cost reduction stems from the substitution of expensive Lewis acid catalysts and alkali metal halides with commodity chemicals like piperazine. Furthermore, the use of standard polar aprotic solvents allows for simpler reactor requirements compared to the corrosion-resistant equipment needed for strong Lewis acids.
Q: How does this process ensure high regioselectivity for the 2-hydroxy product?
A: The reaction conditions, specifically the use of cyclic amines in high-boiling polar aprotic solvents at elevated temperatures (130-150°C), facilitate a highly selective nucleophilic attack on the methoxy group at the 2-position, minimizing the formation of unwanted isomers and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-4,5-Dimethoxybenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the production of high-value pharmaceutical intermediates. Our technical team has thoroughly analyzed the advancements presented in CN103387494A and possesses the expertise to implement this amine-mediated demethylation technology at a commercial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of this essential building block. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-hydroxy-4,5-dimethoxybenzoic acid meets the exacting standards required for API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized manufacturing process can benefit your specific supply chain requirements. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering superior quality and value as your trusted partner in fine chemical manufacturing.
