Advanced Synthetic Route for High-Purity Epoxy Ketone Intermediates in Carfilzomib Manufacturing
Introduction to Advanced Carfilzomib Intermediate Synthesis
The global demand for effective oncology treatments continues to drive innovation in the synthesis of complex peptide-mimetic drugs like Carfilzomib, a potent proteasome inhibitor approved for the treatment of relapsed and refractory multiple myeloma. As detailed in patent CN103864889A, a novel preparation method for a critical epoxy ketone compound, designated as Formula (I), has been developed to address the limitations of existing manufacturing processes. This epoxy ketone serves as the pivotal warhead in the Carfilzomib structure, responsible for its irreversible binding to the proteasome. The disclosed technology offers a streamlined, high-yield pathway that enhances both the economic viability and the environmental profile of producing this life-saving medication. By optimizing the construction of the epoxy ketone pharmacophore, this method provides a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates.
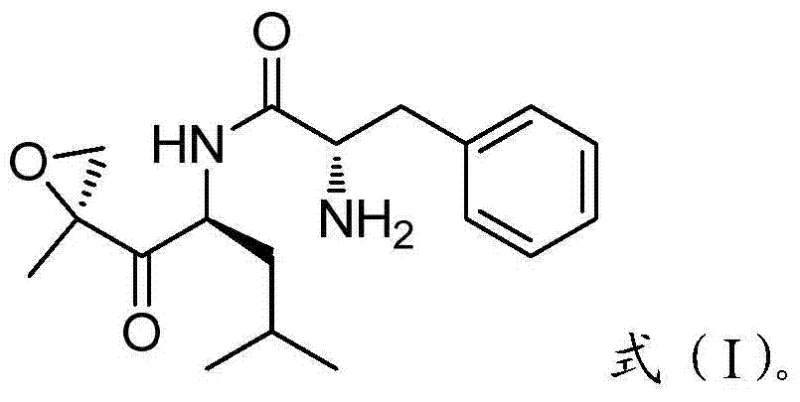
The significance of this intermediate cannot be overstated, as its stereochemical integrity and purity directly influence the efficacy and safety of the final API. Traditional methods often struggle with low linear yields and the generation of difficult-to-remove impurities, which can bottleneck supply chains for high-purity pharmaceutical intermediates. The approach outlined in this patent utilizes a strategic sequence of condensation, amidation, Grignard addition, and selective oxidation to construct the epoxy ketone motif with precision. This technical breakthrough not only simplifies the synthetic route but also aligns with modern green chemistry principles by eliminating toxic reagents, thereby positioning it as a superior choice for cost reduction in pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, such as those described in US20050245435, rely on a linear sequence that introduces significant operational hazards and inefficiencies. A major drawback of these conventional methods is the utilization of chloroacetyl chloride, a highly reactive and corrosive reagent that poses severe safety risks and generates substantial hazardous waste requiring complex disposal protocols. Furthermore, these legacy processes often employ palladium-on-carbon catalyzed hydrogenation steps to remove protecting groups, which introduces the risk of heavy metal contamination in the final product. Removing trace palladium to meet stringent regulatory limits adds costly purification steps and extends production lead times. Additionally, the multi-step nature of the old route results in cumulative yield losses, making the overall process economically inefficient for large-scale production.
The Novel Approach
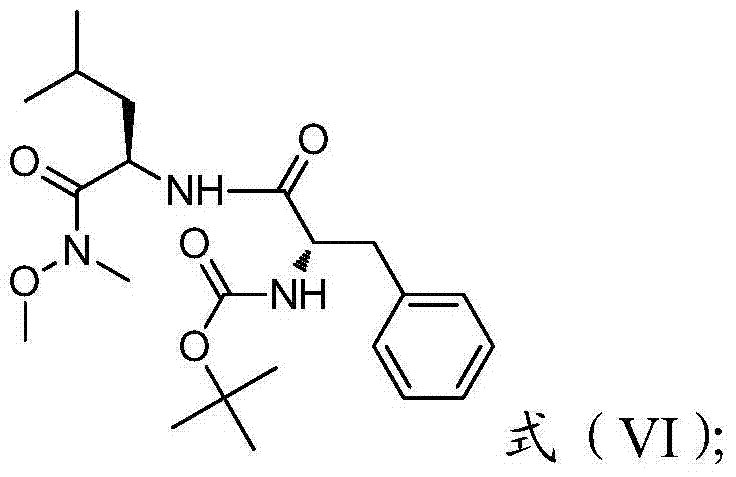
In contrast, the novel methodology presented in CN103864889A circumvents these issues by employing a more direct and chemically elegant strategy. The new route initiates with the condensation of L-leucine ester and N-Boc-L-phenylalanine, followed by conversion to a Weinreb amide, which serves as a stable precursor for the subsequent carbon-carbon bond formation. This approach eliminates the need for chloroacetyl chloride entirely, drastically reducing the environmental footprint and operator exposure risks. The use of a Grignard reagent allows for the precise installation of the unsaturated side chain, which is then selectively oxidized to form the epoxide. This streamlined sequence reduces the total number of unit operations and avoids the use of transition metal catalysts, resulting in a cleaner crude product profile and significantly simplified downstream processing.
Mechanistic Insights into Peptide Coupling and Epoxidation
The core of this synthetic innovation lies in the controlled formation of the peptide backbone and the subsequent functionalization of the side chain. The process begins with a peptide coupling reaction between L-leucine ester and N-Boc-L-phenylalanine using activating agents such as DIC or EDCI in the presence of additives like HOBT. This activation ensures high coupling efficiency while minimizing racemization, a critical factor for maintaining the biological activity of the final drug. Following the initial coupling, the ester group is hydrolyzed under mild basic conditions to reveal the free carboxylic acid, which is then transformed into a Weinreb amide using N,O-dimethylhydroxylamine. This specific amide functionality is crucial because it stabilizes the tetrahedral intermediate during the subsequent nucleophilic attack, preventing over-addition of the Grignard reagent and ensuring the formation of the ketone rather than an alcohol.
The final stages involve the stereoselective construction of the epoxy ketone warhead. The Weinreb amide reacts with pseudoallyl magnesium bromide to generate an alpha,beta-unsaturated ketone. This enone intermediate is then subjected to oxidation using potassium hydrogen persulfate composite salt (Oxone), a safe and effective oxidant that converts the double bond into the strained epoxide ring with high regioselectivity. The reaction conditions are carefully controlled, typically between -15°C and 0°C, to prevent ring opening or degradation of the sensitive epoxide. Finally, acidic deprotection removes the Boc group to yield the target epoxy ketone compound. This mechanistic pathway ensures that chiral centers remain intact throughout the synthesis, delivering a product with the specific stereochemistry required for potent proteasome inhibition.
How to Synthesize Epoxy Ketone Compound Efficiently
The synthesis of this critical intermediate requires precise control over reaction parameters to maximize yield and purity. The process is designed to be robust, utilizing readily available starting materials and standard organic solvents like acetonitrile, dichloromethane, and THF. Operators must pay close attention to temperature control during the Grignard addition and oxidation steps to avoid exothermic runaways and ensure product stability. The workup procedures involve standard aqueous extractions and crystallization techniques, making the process compatible with existing multipurpose reactor infrastructure. For a detailed breakdown of the specific molar ratios, solvent volumes, and isolation techniques validated in the patent examples, please refer to the standardized protocol below.
- Condense L-leucine ester with N-Boc-L-phenylalanine using a coupling agent, followed by deprotection to obtain the dipeptide acid.
- React the dipeptide acid with N,O-dimethylhydroxylamine hydrochloride to form the Weinreb amide intermediate.
- Treat the Weinreb amide with pseudoallyl magnesium bromide to generate the enone species.
- Oxidize the enone with potassium hydrogen persulfate composite salt and perform final deprotection to yield the epoxy ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere technical feasibility. By shifting away from hazardous reagents and precious metal catalysts, manufacturers can achieve substantial cost savings in raw material procurement and waste management. The elimination of palladium catalysts removes the need for expensive scavenging resins and extensive metal testing, directly lowering the cost of goods sold. Furthermore, the simplified workflow reduces the overall cycle time per batch, allowing for increased throughput without the need for additional capital investment in reactor capacity. This efficiency translates into a more resilient supply chain capable of meeting fluctuating market demands for oncology therapeutics.
- Cost Reduction in Manufacturing: The removal of chloroacetyl chloride and palladium catalysts significantly lowers the direct material costs associated with the synthesis. Chloroacetyl chloride requires specialized handling equipment and neutralization protocols, the elimination of which reduces both CAPEX and OPEX. Additionally, the higher overall yield of the new route means less starting material is required to produce the same amount of final API, further driving down the unit cost. The use of Oxone as an oxidant is also economically favorable compared to traditional heavy metal oxidants, offering a cheaper and safer alternative that does not require complex wastewater treatment for heavy metal removal.
- Enhanced Supply Chain Reliability: The starting materials for this process, such as L-leucine esters and N-Boc-phenylalanine, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source bottlenecks. Unlike processes dependent on specialized catalysts or custom-synthesized reagents that may have long lead times, this route relies on a robust supply base. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites without significant re-validation, ensuring continuity of supply even in the event of regional disruptions or facility maintenance.
- Scalability and Environmental Compliance: This synthetic method is inherently scalable, having been demonstrated to work effectively with standard industrial solvents and equipment. The absence of toxic chloroacetyl chloride and heavy metals simplifies the environmental compliance burden, making it easier to obtain necessary operating permits in regions with strict environmental regulations. The reduced generation of hazardous waste lowers disposal costs and aligns with corporate sustainability goals. The process safety profile is improved by avoiding pyrophoric reagents and high-pressure hydrogenation, creating a safer working environment and reducing insurance and liability costs associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding the quality, scalability, and regulatory implications of this new manufacturing route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the advantages of this new epoxy ketone synthesis route over prior art?
A: This method avoids the use of hazardous chloroacetyl chloride and expensive palladium catalysts found in older routes, significantly reducing environmental pollution and raw material costs while improving overall yield.
Q: What is the purity profile of the epoxy ketone intermediate produced?
A: The patented process consistently achieves high purity levels, often exceeding 98% after standard workup and purification, which is critical for the subsequent synthesis of high-quality Carfilzomib API.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (ranging from -15°C to 30°C) and utilize commercially available reagents, making the process highly scalable and robust for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epoxy Ketone Compound Supplier
As the pharmaceutical industry continues to evolve, the need for efficient and compliant manufacturing partners has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN103864889A to deliver high-value intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of epoxy ketone intermediate meets the highest standards required for API synthesis, providing you with peace of mind and regulatory confidence.
We invite you to collaborate with us to optimize your supply chain for Carfilzomib and related analogues. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to a dedicated technical procurement team capable of delivering specific COA data and comprehensive route feasibility assessments. Contact us today to discuss how our innovative manufacturing capabilities can support your long-term strategic goals in the oncology sector.
