Advanced Synthesis of Pyridine Ethylthio Compounds: Enhancing Yield and Catalyst Performance for Industrial Applications
Advanced Synthesis of Pyridine Ethylthio Compounds: Enhancing Yield and Catalyst Performance for Industrial Applications
The chemical industry constantly seeks methods to optimize yield and purity, particularly for complex heterocyclic intermediates used in pharmaceutical and agrochemical synthesis. Patent CN100418949C introduces a groundbreaking approach to the manufacture of pyridine ethylthio compounds, addressing long-standing issues regarding low recovery rates caused by feedstock impurities. This technology not only refines the synthesis of valuable sulfur-containing intermediates like 2-(4-pyridyl)ethanethiol but also extends its utility to the modification of acidic ion exchangers used in bisphenol A production. By strictly controlling the concentration of specific positional isomers and by-products in the vinylpyridine starting material, manufacturers can achieve substantially improved process efficiency. This report analyzes the technical merits of this invention, offering strategic insights for R&D directors and procurement leaders seeking reliable pyridine ethylthio compound supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of pyridylalkylthiols involves reacting vinylpyridine with sulfur-containing compounds such as thiourea or thioacetic acid. However, historical data indicates that yields were often unexpectedly low, creating bottlenecks in cost reduction in pharmaceutical intermediates manufacturing. The root cause was identified not in the reaction mechanism itself, but in the quality of the raw material. Commercially available 4-vinylpyridine typically contains various impurities resulting from its own synthesis via the hydroxymethylation of gamma-picoline. These impurities include unreacted gamma-picoline, ethylpyridine, and critically, compounds represented by general formula (1) such as isopropenylpyridine and propenylpyridine. In conventional processes, these impurities were often ignored or only partially removed via simple distillation aimed at eliminating coloring matter or polymers. Consequently, these reactive impurities would compete with the vinylpyridine during the thiolation step, forming unwanted thioether by-products that complicated purification and reduced the overall recovery of the target molecule.
The Novel Approach
The novel approach detailed in the patent fundamentally shifts the focus to feedstock purification as a prerequisite for high-yield synthesis. The inventors discovered that by ensuring the content of compounds represented by general formula (1) is maintained at 4% by weight or less, preferably through rigorous vacuum distillation using packed columns, the yield of the pyridine ethylthio compound improves drastically. This method eliminates the competitive side reactions that plague traditional routes. Furthermore, the patent introduces an innovative method for modifying acidic ion exchangers using these high-purity compounds. Instead of dissolving the hydrophobic modifier in an acidic aqueous solution, the new method disperses the modifier as an emulsion in water. This emulsification technique enhances the contact efficiency between the modifier and the resin surface, resulting in a catalyst with superior performance for bisphenol condensation reactions. This dual innovation offers a robust pathway for commercial scale-up of complex sulfur-containing intermediates.
Mechanistic Insights into Thiourea-Mediated Thiolation
The core chemical transformation relies on the addition of a sulfur nucleophile across the vinyl double bond of the pyridine ring. When thiourea is employed as the sulfur source, the reaction proceeds through the formation of an isothiouronium salt intermediate. As illustrated in the reaction scheme below, the vinyl group undergoes electrophilic addition in the presence of an acid catalyst, such as p-toluenesulfonic acid or sulfuric acid. The nitrogen of the thiourea attacks the beta-carbon of the vinyl group, generating a stable cationic species. This step is highly sensitive to the electronic environment of the double bond; therefore, the presence of conjugated impurities like isopropenylpyridine can disrupt the stoichiometry and generate stable by-products that do not convert to the desired thiol. Maintaining high purity ensures that the acid catalyst is utilized efficiently for the intended transformation rather than being consumed by side reactions with impurities.
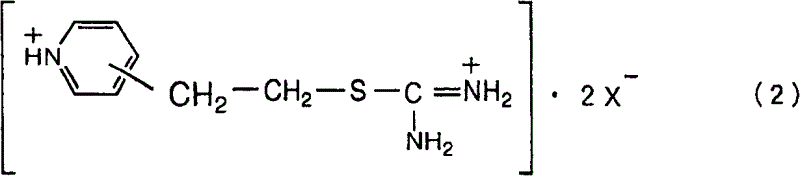
Following the formation of the isothiouronium salt, the process requires a decomposition step to liberate the free thiol. This is achieved by treating the salt with a base, preferably aqueous ammonia, under controlled thermal conditions. The mechanism involves the nucleophilic attack of ammonia or hydroxide ions on the thiocarbonyl carbon, leading to the release of the mercapto group and the formation of urea or guanidine by-products. The patent specifies that this decomposition should occur at temperatures between 30°C and 70°C to balance reaction rate and selectivity. Higher temperatures risk promoting side reactions of the newly formed thiol, while lower temperatures result in impractically slow kinetics. The subsequent workup involves cooling to precipitate guanidinium salts (if aromatic sulfonic acids are used) and extracting the product into an organic solvent like toluene. This precise control over reaction parameters ensures the production of high-purity pyridine derivatives suitable for sensitive downstream applications.
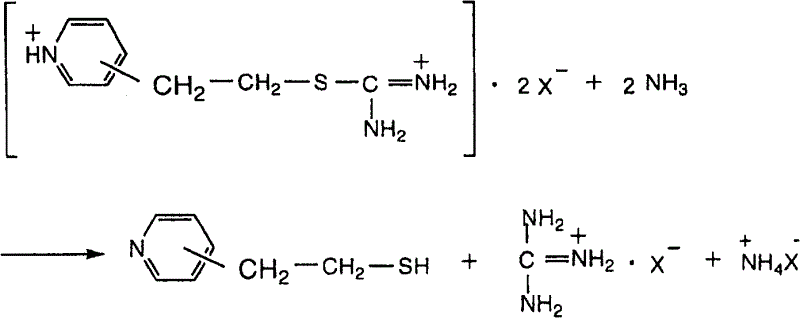
How to Synthesize 2-(4-Pyridyl)ethanethiol Efficiently
Implementing this synthesis route requires strict adherence to the purification protocols outlined in the patent to maximize yield. The process begins with the fractional distillation of the vinylpyridine feedstock to remove specific olefinic impurities, followed by the acid-catalyzed reaction with thiourea. The reaction mixture is then basified to release the thiol, which is subsequently extracted and purified. This workflow minimizes waste and ensures consistent product quality, which is essential for regulatory compliance in pharmaceutical supply chains. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Purify 4-vinylpyridine via vacuum distillation to ensure specific impurity levels (Formula 1 compounds) remain below 4% by weight.
- React the purified vinylpyridine with thiourea in the presence of an acid catalyst (e.g., p-toluenesulfonic acid) to form the isothiouronium salt intermediate.
- Decompose the salt using aqueous ammonia under controlled temperature conditions to yield the final pyridine ethanethiol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible operational efficiencies and risk mitigation. The primary advantage lies in the significant yield improvement derived from feedstock purification. By eliminating the formation of difficult-to-remove thioether by-products, the process reduces the load on downstream purification units, such as distillation columns and crystallization tanks. This simplification of the workflow leads to substantial cost savings in terms of energy consumption and solvent usage. Furthermore, the consistency of the product quality reduces the likelihood of batch failures, thereby enhancing supply chain reliability and ensuring on-time delivery to downstream customers who rely on these intermediates for API synthesis.
- Cost Reduction in Manufacturing: The elimination of competitive side reactions means that raw materials are converted more efficiently into the desired product. This higher atom economy reduces the cost per kilogram of the final intermediate. Additionally, the emulsion-based modification method for ion exchangers removes the need for large volumes of strong acid to dissolve the modifier, lowering chemical procurement costs and reducing the corrosive burden on reactor equipment. These factors collectively contribute to a more lean and cost-effective manufacturing operation without compromising on product specifications.
- Enhanced Supply Chain Reliability: High-purity feedstocks and optimized reaction conditions result in a more predictable production schedule. The reduced formation of polymeric and tarry by-products minimizes reactor fouling, which in turn decreases downtime required for cleaning and maintenance. This operational stability allows for longer campaign runs and more consistent output, which is critical for meeting the demanding delivery schedules of global pharmaceutical clients. Reducing lead time for high-purity pyridine derivatives becomes achievable when the process is robust against feedstock variability.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard unit operations like vacuum distillation and liquid-liquid extraction. The use of ammonia for salt decomposition generates urea or ammonium salts as by-products, which are generally easier to handle and dispose of compared to heavy metal waste from alternative catalytic routes. The emulsion modification technique further aligns with green chemistry principles by using water as the dispersion medium, reducing the reliance on volatile organic solvents during the catalyst preparation phase. This facilitates easier compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. They are derived from the specific problem-solution dynamics presented in the patent documentation, focusing on impurity control and catalyst modification techniques. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: Why is controlling impurities in vinylpyridine critical for yield?
A: Impurities such as isopropenylpyridine react with sulfur compounds to form difficult-to-separate thioethers, significantly lowering the recovery rate of the target pyridine ethylthio compound.
Q: What are the advantages of the emulsion modification method for ion exchangers?
A: Using an emulsion of the modifier in water enhances surface reactivity on the ion exchange resin, leading to higher catalytic activity in bisphenol production compared to traditional acid-dissolution methods.
Q: Can this process be scaled for industrial bisphenol A production?
A: Yes, the patent specifically addresses industrial scalability, noting that even minor improvements in catalyst performance translate to significant economic benefits at the 20,000-ton/year production scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Pyridyl)ethanethiol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipelines. Our technical team has extensively analyzed the methodologies described in patent CN100418949C and possesses the expertise to replicate and optimize these pathways for commercial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of pyridine ethylthio compound meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage these advanced synthesis techniques for your specific projects. Our team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to our optimized manufacturing route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your unique chemical requirements. Let us be your partner in driving efficiency and innovation in your supply chain.
