Scalable Synthesis of 2-Amino-4-Bromo-3-Fluoro-5-Iodobenzamide for Commercial API Production
The pharmaceutical industry's relentless pursuit of potent kinase inhibitors has placed a premium on highly functionalized aromatic building blocks, specifically poly-halogenated benzamides. A pivotal advancement in this domain is detailed in patent CN112341351B, which discloses a novel, high-yield preparation method for 2-amino-4-bromo-3-fluoro-5-iodobenzamide. This compound serves as a critical scaffold for next-generation oncology therapeutics, where precise halogen positioning is paramount for binding affinity. The disclosed technology addresses longstanding bottlenecks in traditional synthesis, specifically the reliance on prohibitively expensive starting materials and inefficient purification protocols that have historically hindered commercial viability. By shifting the synthetic strategy to utilize 2,3-difluorobromobenzene as the foundational feedstock, the process achieves a remarkable total yield of approximately 53.7% while completely bypassing the need for column chromatography. For R&D directors and procurement specialists alike, this represents a transformative opportunity to secure a reliable supply chain for high-purity pharmaceutical intermediates at a fraction of the historical cost.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those referenced in WO2018143315 and US20180015087, typically rely on the condensation of 2-fluoro-3-bromonitrobenzene, a starting material characterized by volatile market pricing and limited availability. As illustrated in the reaction scheme below, these legacy routes often involve complex cyclization steps through isatin derivatives or low-efficiency condensation reactions that struggle to drive conversion to completion. 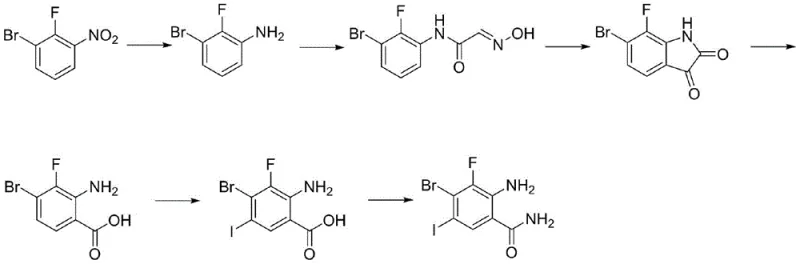 The fundamental flaw in these conventional approaches lies in the final steps, where the yield of the target benzamide drops precipitously, necessitating rigorous purification via column chromatography. This reliance on silica gel purification is not only labor-intensive and solvent-heavy but also creates a significant barrier to industrial amplification, making it nearly impossible to produce multi-kilogram quantities without exorbitant operational expenditures. Furthermore, the impurity profiles generated by these harsh condensation conditions often include difficult-to-remove structural analogs, compromising the purity specifications required for GMP-grade API manufacturing.
The fundamental flaw in these conventional approaches lies in the final steps, where the yield of the target benzamide drops precipitously, necessitating rigorous purification via column chromatography. This reliance on silica gel purification is not only labor-intensive and solvent-heavy but also creates a significant barrier to industrial amplification, making it nearly impossible to produce multi-kilogram quantities without exorbitant operational expenditures. Furthermore, the impurity profiles generated by these harsh condensation conditions often include difficult-to-remove structural analogs, compromising the purity specifications required for GMP-grade API manufacturing.
The Novel Approach
In stark contrast, the innovative route presented in CN112341351B leverages a linear, seven-step sequence that prioritizes atom economy and operational simplicity. Starting from the commercially abundant 2,3-difluorobromobenzene (Compound II), the process employs a regioselective lithiation-carboxylation strategy to install the carboxylic acid moiety with high precision. 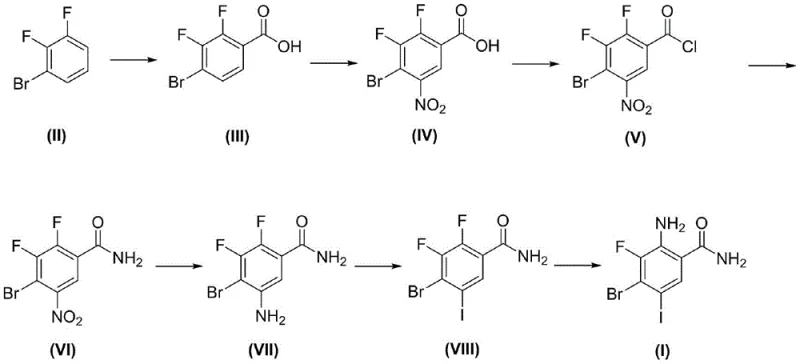 This is followed by a carefully controlled nitration using mixed acid, conversion to the acid chloride, and subsequent amidation. The brilliance of this approach is evident in the downstream processing; the reduction of the nitro group using iron powder and the subsequent Sandmeyer iodination proceed with yields exceeding 82% at each stage. Crucially, the final high-pressure ammonolysis step efficiently displaces the remaining fluorine atom to generate the target diamine structure. By eliminating column chromatography entirely and relying on crystallization and filtration, this method offers a direct pathway for cost reduction in pharmaceutical intermediate manufacturing, ensuring that the process is not just chemically elegant but economically robust for large-scale production.
This is followed by a carefully controlled nitration using mixed acid, conversion to the acid chloride, and subsequent amidation. The brilliance of this approach is evident in the downstream processing; the reduction of the nitro group using iron powder and the subsequent Sandmeyer iodination proceed with yields exceeding 82% at each stage. Crucially, the final high-pressure ammonolysis step efficiently displaces the remaining fluorine atom to generate the target diamine structure. By eliminating column chromatography entirely and relying on crystallization and filtration, this method offers a direct pathway for cost reduction in pharmaceutical intermediate manufacturing, ensuring that the process is not just chemically elegant but economically robust for large-scale production.
Mechanistic Insights into Regioselective Lithiation and Sandmeyer Iodination
The cornerstone of this synthesis is the initial lithiation step, which dictates the entire substitution pattern of the final molecule. By treating 2,3-difluorobromobenzene with n-butyllithium at cryogenic temperatures of -78°C, the protocol exploits the directing effects of the adjacent fluorine and bromine atoms to achieve exclusive deprotonation at the C-2 position. This generates a highly reactive aryl lithium species that, upon quenching with carbon dioxide, forms the benzoic acid intermediate (Compound III) with a molar yield of up to 93.6%. This low-temperature control is vital; any deviation could lead to competing metal-halogen exchange or non-selective deprotonation, which would introduce isomeric impurities that are notoriously difficult to separate in later stages. The subsequent nitration step utilizes concentrated sulfuric acid and nitric acid at 0-30°C, leveraging the meta-directing nature of the newly formed carboxyl group to place the nitro moiety precisely at the C-5 position, setting the stage for the eventual amino group installation.
Further down the synthetic chain, the transformation of the aniline intermediate (Compound VII) into the iodo-analog (Compound VIII) via the Sandmeyer reaction is a masterclass in impurity control. Traditional electrophilic aromatic iodination often suffers from poly-iodination or poor regioselectivity on activated rings. However, by converting the amine to a diazonium salt using sodium nitrite at -10 to 0°C and immediately displacing it with potassium iodide or cuprous iodide at 60-80°C, the process ensures a clean substitution. The use of iron powder for the preceding nitro reduction is also strategically chosen over catalytic hydrogenation to avoid potential dehalogenation of the sensitive bromine and fluorine substituents. This meticulous attention to chemoselectivity ensures that the final product maintains a purity greater than 98% as confirmed by GC analysis, meeting the stringent requirements for a reliable pharmaceutical intermediate supplier without the need for extensive downstream polishing.
How to Synthesize 2-Amino-4-Bromo-3-Fluoro-5-Iodobenzamide Efficiently
The execution of this seven-step synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry, to maintain the high yields reported in the patent data. The process begins with the careful addition of 2.5M n-butyllithium to the substrate in tetrahydrofuran at -78°C, followed by the introduction of carbon dioxide gas to trap the lithiated species. Subsequent steps involve standard unit operations such as acid-base extraction, solvent swapping, and thermal reflux, making the route highly compatible with existing multipurpose reactor infrastructure. The detailed standardized synthesis steps, including specific molar ratios and workup procedures for each of the seven transformations, are outlined in the guide below to assist process chemists in replicating this high-efficiency protocol.
- Perform regioselective lithiation of 2,3-difluorobromobenzene at -78°C followed by carboxylation with CO2 to form the benzoic acid intermediate.
- Execute nitration using concentrated sulfuric and nitric acid at controlled temperatures (0-30°C) to introduce the nitro group ortho to the carboxyl.
- Convert the carboxylic acid to acid chloride using thionyl chloride, then react with ammonia to form the primary amide.
- Reduce the nitro group to an amine using iron powder in an acetic acid/ethanol mixture, followed by Sandmeyer iodination.
- Complete the synthesis via high-pressure ammonolysis to replace the remaining fluorine atom with an amino group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers immediate and tangible benefits that extend far beyond simple chemistry. The primary driver of value is the substitution of the starting material; replacing the expensive and supply-constrained 2-fluoro-3-bromonitrobenzene with 2,3-difluorobromobenzene results in substantial cost savings on raw material acquisition. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates supply risk, as the new starting material is a commodity chemical produced by multiple global vendors, ensuring continuity of supply even during market fluctuations. Furthermore, the elimination of column chromatography removes a major bottleneck in production throughput, drastically reducing solvent consumption and waste disposal costs associated with silica gel treatment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive purification steps and the optimization of reaction yields. By achieving individual step yields consistently above 82% and avoiding the low-efficiency condensation of prior art, the overall material throughput is significantly improved. The avoidance of transition metal catalysts in the reduction step (using iron powder instead) further reduces the cost burden associated with catalyst recovery and heavy metal testing, leading to a leaner, more cost-effective manufacturing profile that enhances margin potential for the final API.
- Enhanced Supply Chain Reliability: Dependence on niche starting materials often leads to extended lead times and vulnerability to supplier disruptions. This new route utilizes 2,3-difluorobromobenzene, a widely available bulk chemical, which simplifies sourcing strategies and reduces lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without significant yield loss, ensures that production schedules can be met reliably, providing a stable foundation for long-term supply agreements with downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is designed for seamless commercial scale-up of complex pharmaceutical intermediates. The use of aqueous workups and simple filtration for isolation minimizes the generation of hazardous organic waste streams compared to chromatographic methods. Additionally, the high-pressure ammonolysis step, while requiring specialized equipment, is a well-understood unit operation in the fine chemical industry, posing no novel safety risks. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-amino-4-bromo-3-fluoro-5-iodobenzamide, based on the specific embodiments and data provided in patent CN112341351B. These insights are intended to clarify the feasibility of the process for potential partners and highlight the distinct advantages over legacy methods. Understanding these technical nuances is essential for evaluating the suitability of this route for your specific project requirements.
Q: What is the key advantage of using 2,3-difluorobromobenzene over 2-fluoro-3-bromonitrobenzene?
A: Using 2,3-difluorobromobenzene significantly reduces raw material costs as it is cheaper and more readily available than the expensive nitro-starting material used in prior art. Additionally, it allows for a more controlled introduction of functional groups, avoiding low-yield condensation steps.
Q: Does this process require column chromatography for purification?
A: No, one of the major benefits of this patented method is that it eliminates the need for column chromatography. The intermediates and final product can be purified through simple filtration, pulping, and recrystallization, which is crucial for industrial scale-up.
Q: What is the overall yield of the new synthetic route?
A: The process demonstrates high efficiency with individual step yields ranging from 82% to 94.5%, resulting in a robust total yield of approximately 53.7%, which is superior to conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-Bromo-3-Fluoro-5-Iodobenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel kinase inhibitors depends on the availability of high-quality, cost-effective building blocks. Our team of expert process chemists has thoroughly analyzed the route disclosed in CN112341351B and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. Our state-of-the-art facilities are equipped to handle the specific conditions required for this synthesis, including cryogenic lithiation and high-pressure ammonolysis, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets your exacting standards.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this new route for your specific volume needs. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how we can support your supply chain with a reliable source of this critical intermediate.
