Scalable Two-Step Synthesis of Tetra-Acylated Puerarin for High-Purity Pharmaceutical Applications
Scalable Two-Step Synthesis of Tetra-Acylated Puerarin for High-Purity Pharmaceutical Applications
The pharmaceutical industry continuously seeks advanced intermediates that offer improved bioavailability and solubility profiles for active drug substances. A significant breakthrough in this domain is documented in patent CN101792434A, which details a novel preparation method for tetra-acylated puerarin. This specific derivative addresses the inherent limitations of native puerarin, such as poor solubility and low bioavailability, by chemically modifying its structure to enhance pharmacokinetic properties. The disclosed technology moves away from traditional, inefficient single-step acylation routes that result in complex mixtures, instead proposing a controlled two-step strategy. This approach not only maximizes the yield of the target tetra-acylated compound but also streamlines the purification process, making it a highly attractive candidate for commercial scale-up. For R&D directors and procurement specialists, understanding this technological shift is critical for securing a reliable supply of high-quality cardiovascular and metabolic disease intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetra-acylated puerarin relied on direct, one-step acetylation of puerarin using acetylating agents. This conventional operational path is fraught with significant chemical and economic inefficiencies that hinder industrial adoption. The primary drawback is the lack of regioselectivity, which leads to the simultaneous formation of tetra-, penta-, and hexa-acetyl puerarin byproducts in a chaotic ratio, often approximating 15:20:25. Consequently, the yield of the desired tetra-acylated product is dismally low, typically hovering around 25%, which is economically unsustainable for large-scale manufacturing. Furthermore, separating these structurally similar components necessitates the use of silica gel column chromatography, a technique that is notoriously solvent-intensive, time-consuming, and difficult to scale beyond laboratory quantities. The heavy reliance on chromatography not only inflates production costs but also introduces bottlenecks in supply chain continuity, making it an unreliable method for meeting the rigorous demands of the global pharmaceutical market.
The Novel Approach
In stark contrast to the inefficient traditional methods, the novel approach outlined in the patent data employs a strategic two-step synthesis that prioritizes selectivity and ease of purification. The process begins with the complete acylation of puerarin to form hexaacyl puerarin, ensuring that all reactive hydroxyl groups are protected. This intermediate is then subjected to a controlled selective deacylation reaction, where specific acyl groups are removed under mild conditions to yield the target tetra-acylated puerarin. This methodology fundamentally alters the impurity profile, allowing for the use of simple recrystallization techniques rather than complex chromatography for final purification. The result is a process that is not only operationally simpler but also significantly more economical, as it reduces solvent usage and manpower requirements. By shifting the complexity from separation to reaction control, this method offers a robust pathway for the commercial production of high-purity intermediates.
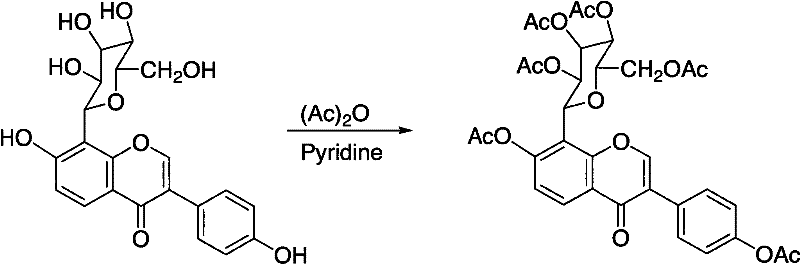
Mechanistic Insights into Selective Acylation and Deacylation
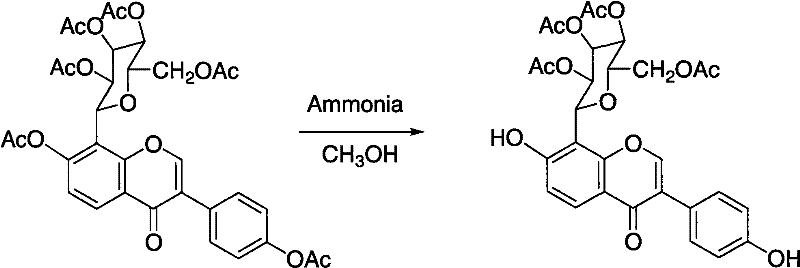
The core of this technological advancement lies in the precise control of acylation and deacylation mechanisms, which dictates the purity and yield of the final product. In the first step, puerarin reacts with an acylating agent, such as acetic anhydride, in the presence of a base like pyridine. This reaction proceeds to completion to form the hexaacyl derivative, effectively masking all hydroxyl groups including those on the sugar moiety and the isoflavone backbone. The second step is the critical differentiator, where a deacylating reagent, such as ammonia or ammonium salts, is introduced in an alcoholic solvent. This reagent selectively cleaves the acyl groups at the 7 and 4' positions on the isoflavone ring while leaving the acyl groups on the sugar moiety intact. This regioselectivity is driven by the electronic and steric environment of the ester bonds, allowing for the targeted generation of the tetra-acylated structure without affecting the rest of the molecule.
Impurity control is inherently built into this two-step mechanism, providing a significant advantage over single-step processes. By fully acylating the starting material first, the reaction eliminates the possibility of forming partially acylated byproducts that are difficult to separate later. The subsequent deacylation step is tuned to stop at the tetra-acyl stage, preventing over-hydrolysis back to the starting puerarin. The use of mild bases like ammonia or ammonium bicarbonate ensures that the reaction conditions remain gentle, preserving the integrity of the sensitive isoflavone core. This mechanistic precision means that the crude product obtained after the reaction is already of high purity, requiring only a final recrystallization to meet stringent pharmaceutical specifications. For quality assurance teams, this translates to a more consistent impurity profile and reduced risk of batch-to-batch variability.
How to Synthesize Tetra-Acylated Puerarin Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the benefits of the two-step design. The process begins with the acylation of puerarin in pyridine at ambient or slightly elevated temperatures, followed by the isolation of the hexaacyl intermediate. The critical second step involves treating this intermediate with a deacylating agent in a solvent system such as methanol or ethanol. The reaction can be conducted at room temperature or with mild heating, offering flexibility depending on the available infrastructure. Detailed standard operating procedures regarding stoichiometry, temperature profiles, and work-up protocols are essential for reproducibility. For a comprehensive breakdown of the standardized synthesis steps, please refer to the technical guide below.
- Perform acylation of puerarin with acetic anhydride in pyridine to generate hexaacyl puerarin intermediate.
- Conduct selective deacylation using ammonia or ammonium salts in alcohol solvents to remove specific acyl groups.
- Purify the final tetra-acylated product through recrystallization to achieve high purity without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this two-step synthesis method offers profound advantages for procurement managers and supply chain heads looking to optimize costs and reliability. The elimination of silica gel column chromatography is the most significant value driver, as it removes a major bottleneck associated with solvent consumption, waste disposal, and processing time. This simplification of the downstream processing directly translates to substantial cost savings in manufacturing, making the final intermediate more price-competitive without sacrificing quality. Additionally, the use of common, inexpensive reagents like ammonia and acetic anhydride ensures that raw material costs remain stable and predictable. The robustness of the recrystallization purification method further enhances supply chain reliability by reducing the risk of production delays associated with complex separation techniques.
- Cost Reduction in Manufacturing: The primary economic benefit stems from the complete removal of column chromatography from the production workflow. Traditional methods require vast quantities of silica gel and organic solvents, which are expensive to purchase and dispose of in compliance with environmental regulations. By switching to a recrystallization-based purification, manufacturers can drastically reduce solvent usage and eliminate the need for costly chromatographic media. This process intensification leads to a leaner manufacturing operation with lower overheads, allowing for more competitive pricing structures in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often compromised by complex purification steps that are prone to variability and failure. The new method utilizes robust chemical transformations that are easier to control and scale, reducing the likelihood of batch failures. Furthermore, the reagents required, such as ammonia and alcohols, are commodity chemicals with stable global supply chains, minimizing the risk of raw material shortages. This reliability ensures that production schedules can be met consistently, providing downstream drug manufacturers with the confidence of a steady supply of critical intermediates for their own formulation processes.
- Scalability and Environmental Compliance: Scaling chemical processes often exacerbates environmental challenges, particularly regarding waste generation. This novel route significantly reduces the environmental footprint by minimizing solvent waste and avoiding the disposal of spent silica gel. The mild reaction conditions also contribute to lower energy consumption compared to processes requiring extreme temperatures or pressures. These factors make the technology highly scalable and compliant with increasingly stringent environmental regulations, positioning it as a sustainable choice for long-term manufacturing partnerships in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of tetra-acylated puerarin. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical properties of this intermediate. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this material into their development pipelines. The answers provided reflect the consensus on the superior performance of the two-step method over legacy technologies.
Q: Why is the two-step method superior to direct acetylation for puerarin derivatives?
A: Direct acetylation yields a complex mixture of tetra-, penta-, and hexa-acetyl products with low selectivity, requiring difficult column chromatography. The two-step method ensures high selectivity for the tetra-acylated form through controlled deacylation, simplifying purification.
Q: What are the key reaction conditions for the deacylation step?
A: The deacylation can be performed under mild conditions using ammonia or ammonium salts in methanol or ethanol. Temperatures range from ambient to 80°C, allowing flexibility for scale-up while maintaining selectivity.
Q: How does this process impact commercial scalability?
A: By eliminating silica gel column chromatography and utilizing recrystallization for purification, the process significantly reduces solvent consumption and processing time, making it highly suitable for large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetra-Acylated Puerarin Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of complex acylation and deacylation chemistries, ensuring that the transition from laboratory patent data to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tetra-acylated puerarin meets the exacting standards required for pharmaceutical applications. Our commitment to quality and scalability makes us the ideal partner for companies seeking to secure a long-term supply of this high-value intermediate.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing processes, we can help you reduce lead time for high-purity pharmaceutical intermediates while maintaining cost efficiency. Please contact our technical procurement team to request specific COA data and route feasibility assessments. We are ready to support your supply chain with reliable, high-quality chemical solutions that drive your product development forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
