Scalable Synthesis of S-1-(4-ethoxybenzyl)-3-azapentane-1,5-diamine for High-Purity MRI Contrast Agents
The pharmaceutical industry's demand for high-quality Magnetic Resonance Imaging (MRI) contrast agents has driven significant innovation in the synthesis of their key precursors. Patent CN103896788A introduces a groundbreaking preparation method for S-1-(4-ethoxybenzyl)-3-azapentane-1,5-diamine trihydrochloride, a critical intermediate for Gadoxetic acid disodium. This novel approach addresses long-standing safety and efficiency bottlenecks found in legacy manufacturing processes by replacing hazardous reagents with safer, more cost-effective alternatives. By utilizing amino-protected L-tyrosine alkyl esters as the starting material, the process achieves a total yield of up to 89% while maintaining exceptional product purity above 99%. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more reliable supply chains for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
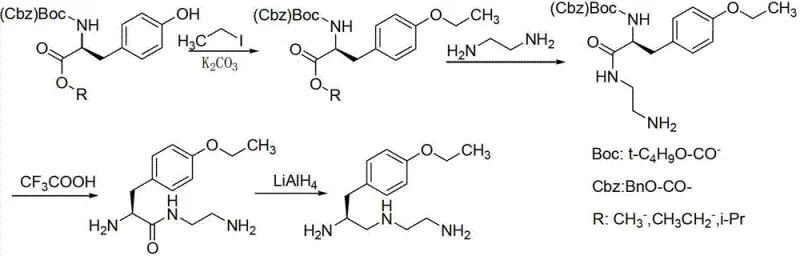
Historically, the synthesis of this vital diamine intermediate relied on perilous chemical pathways that posed severe risks to both personnel and the environment. As illustrated in prior art routes, traditional methods frequently employed iodoethane, a highly carcinogenic and virulent alkylating agent, which necessitates rigorous containment protocols and increases operational liability. Furthermore, conventional reduction steps often utilized lithium aluminum hydride (LiAlH4) or diborane, reagents that require strictly anhydrous conditions and present significant explosion hazards during large-scale handling. The reliance on high-pressure catalytic hydrogenation with palladium on carbon (Pd/C) further complicated the process, introducing risks of heavy metal contamination and requiring expensive, specialized pressure vessels. These factors collectively resulted in lower total yields, typically around 53% to 55%, and difficult post-treatment procedures involving toxic solvents like trifluoroacetic acid.
The Novel Approach
The innovative methodology described in the patent fundamentally re-engineers the synthetic pathway to eliminate these critical vulnerabilities. By substituting iodoethane with bromoethane, the process drastically reduces toxicity profiles while maintaining high alkylation efficiency through the use of potassium iodide as a catalyst. The replacement of dangerous hydride reducers with sodium borohydride allows the reduction step to proceed safely at room temperature in common solvent systems like tetrahydrofuran and methanol. Additionally, the deprotection strategy shifts from harsh acidic conditions or high-pressure hydrogenation to a mild hydrogen chloride gas treatment in methanol. This strategic redesign not only enhances operator safety but also streamlines the purification workflow, removing the need for complex heavy metal scavenging steps and enabling a more robust, scalable manufacturing process suitable for modern GMP facilities.
Mechanistic Insights into Sodium Borohydride Reduction and Mesylation Strategy
The core chemical innovation lies in the seamless transition from the ester functionality to the diamine side chain via a stabilized alcohol intermediate. In the second step of the novel route, the ethyl ester moiety of the tyrosine derivative is selectively reduced to a primary alcohol using sodium borohydride. Unlike aggressive reducing agents, sodium borohydride offers chemoselectivity that preserves the carbamate protecting group and the ether linkage, preventing unwanted side reactions that could generate difficult-to-remove impurities. Following reduction, the resulting hydroxyl group is activated through mesylation using alkyl sulfonyl chloride, converting a poor leaving group into an excellent sulfonate ester. This activation is crucial for the subsequent nucleophilic substitution, where the mesylate is displaced by ethylenediamine. This two-step sequence ensures precise control over the stereochemistry and regioselectivity of the amine installation, which is vital for the biological activity of the final MRI contrast agent.
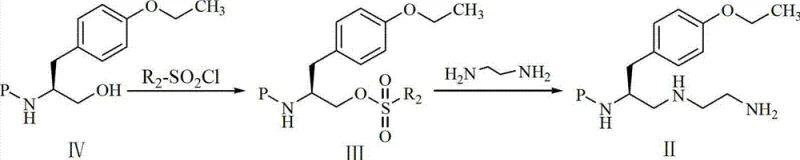
Impurity control is inherently built into this mechanistic design through the choice of mild reaction conditions and specific reagent stoichiometry. The use of excess ethylenediamine drives the substitution reaction to completion, minimizing the presence of unreacted mesylate intermediates which could otherwise persist as genotoxic impurities. Furthermore, the final deprotection step utilizing hydrogen chloride gas in methanol ensures the quantitative removal of the Boc or Cbz protecting groups without inducing racemization or degradation of the sensitive diamine backbone. The formation of the trihydrochloride salt at the end of the process not only stabilizes the molecule for storage but also facilitates crystallization, allowing for the effective rejection of organic byproducts and ensuring the final API intermediate meets the stringent purity standards required for injectable diagnostic agents.
How to Synthesize S-1-(4-ethoxybenzyl)-3-azapentane-1,5-diamine Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing this complex intermediate with high reproducibility. The process begins with the alkylation of the tyrosine precursor, followed by reduction, activation, and amination, culminating in salt formation. Each step has been optimized for ambient or near-ambient temperatures, reducing energy consumption and thermal stress on the equipment. The detailed standardized synthesis steps below outline the specific molar ratios, solvent choices, and reaction times necessary to achieve the reported 89% overall yield and >99% purity.
- Alkylation of amino-protected L-tyrosine alkyl ester with bromoethane using potassium carbonate and potassium iodide catalyst to form the ethoxy derivative.
- Reduction of the ester group to a primary alcohol using sodium borohydride in a tetrahydrofuran and methanol solvent system.
- Activation of the alcohol via mesylation followed by nucleophilic substitution with excess ethylenediamine to install the diamine side chain.
- Final deprotection of the amino group using hydrogen chloride gas in methanol to yield the target trihydrochloride salt with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere technical feasibility. The elimination of high-risk reagents such as iodoethane and lithium aluminum hydride significantly lowers the regulatory burden and insurance costs associated with chemical storage and transport. By avoiding the use of palladium catalysts, manufacturers can bypass the expensive and time-consuming heavy metal clearance testing and purification stages that often bottleneck production schedules. The shift to milder reaction conditions also extends the lifespan of standard stainless steel reactors, reducing capital expenditure on specialized high-pressure equipment. These operational efficiencies translate directly into a more resilient supply chain capable of meeting the growing global demand for liver-specific MRI contrast agents without compromising on safety or quality standards.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents with commodity chemicals like bromoethane and sodium borohydride drives down raw material costs significantly. Furthermore, the simplified workup procedures, which avoid complex extractions and heavy metal scavenging, reduce solvent consumption and waste disposal fees. The higher overall yield of the process means less starting material is required per kilogram of final product, maximizing resource utilization and improving the gross margin profile for the intermediate. This economic efficiency makes the final MRI contrast agent more accessible to healthcare systems while maintaining profitability for manufacturers.
- Enhanced Supply Chain Reliability: By removing dependencies on specialized high-pressure hydrogenation services and toxic alkylating agents, the supply chain becomes less vulnerable to logistical disruptions and regulatory shutdowns. The use of stable, shelf-stable reagents allows for better inventory management and reduces the risk of production delays caused by the short shelf-life of sensitive catalysts. The robustness of the process ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with major pharmaceutical companies developing diagnostic imaging portfolios. This reliability fosters stronger partnerships between intermediate suppliers and downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing safer solvents and generating less hazardous waste streams compared to traditional methods. The absence of heavy metal catalysts simplifies wastewater treatment and aligns with increasingly strict environmental regulations in major manufacturing hubs. The mild reaction conditions facilitate easy scale-up from pilot plant to multi-ton commercial production without the need for extensive re-engineering of safety protocols. This scalability ensures that suppliers can rapidly respond to market surges in demand for Gadoxetic acid disodium, securing their position as a reliable pharmaceutical intermediate supplier in a competitive landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of S-1-(4-ethoxybenzyl)-3-azapentane-1,5-diamine trihydrochloride. These insights are derived directly from the patented methodology and are intended to clarify the advantages of this specific synthetic route for stakeholders evaluating potential sourcing partners. Understanding these technical nuances is essential for making informed decisions about long-term supply agreements and quality assurance protocols.
Q: Why is bromoethane preferred over iodoethane in this synthesis?
A: Bromoethane is significantly less toxic and carcinogenic compared to iodoethane, reducing safety risks for operators and environmental hazards during industrial scale-up, while maintaining high reaction efficiency.
Q: How does this method improve upon traditional lithium aluminum hydride reductions?
A: This method utilizes sodium borohydride, which operates under much milder conditions without the strict anhydrous requirements or explosion risks associated with lithium aluminum hydride, simplifying post-treatment and enhancing safety.
Q: What is the expected purity of the final trihydrochloride salt?
A: The patented process consistently achieves an HPLC purity of greater than 99%, meeting the stringent quality specifications required for pharmaceutical intermediates used in MRI contrast agent production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-1-(4-ethoxybenzyl)-3-azapentane-1,5-diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of life-saving diagnostic tools. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of S-1-(4-ethoxybenzyl)-3-azapentane-1,5-diamine trihydrochloride meets the highest international standards. Our commitment to safety and quality makes us the ideal partner for pharmaceutical companies seeking to optimize their MRI contrast agent supply chains.
We invite you to contact our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis route can reduce your overall production costs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify our capability to deliver consistent, high-quality results for your critical projects.
