Advanced Two-Step Synthesis of Trisphaeridine for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking more efficient pathways to access complex bioactive scaffolds, and the recent disclosure in patent CN114773354B represents a significant leap forward in the synthesis of Trisphaeridine. This benzophenanthridine alkaloid, known for its potent inhibitory activity against human lung, liver, and colon cancer cell lines, has traditionally been difficult to source due to reliance on plant extraction or convoluted synthetic routes. The new methodology outlined in this intellectual property introduces a streamlined, two-step protocol that merges palladium-catalyzed cross-coupling with modern visible-light photocatalysis. For R&D directors and procurement specialists alike, this innovation signals a shift towards more sustainable and economically viable manufacturing processes for high-value anticancer intermediates. By leveraging mild reaction conditions and readily available starting materials, this technology addresses critical bottlenecks in supply chain continuity and cost management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of Trisphaeridine has been plagued by significant logistical and chemical challenges that hinder its widespread application in drug discovery programs. Traditional methods often rely on the isolation of this alkaloid from natural sources such as Narcissus asturiensis, a process that is inherently limited by seasonal availability, geographical constraints, and low natural abundance. Furthermore, existing synthetic routes described in prior literature frequently involve multi-step sequences requiring harsh reagents, extreme temperatures, and complex purification protocols that generate substantial toxic waste. These factors collectively drive up the cost of goods sold (COGS) and extend lead times, making it difficult for pharmaceutical companies to secure a reliable supply of high-purity material for preclinical and clinical studies. The environmental footprint of these legacy methods is also increasingly untenable in an era of strict regulatory compliance regarding solvent use and waste disposal.
The Novel Approach
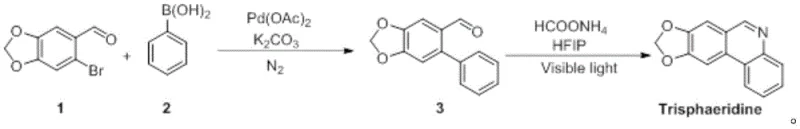
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent utilizes a concise two-step strategy that dramatically simplifies the molecular construction of the target scaffold. The first stage employs a robust Suzuki-Miyaura coupling to construct the biaryl backbone, followed immediately by a sophisticated 6π electrocyclization driven by visible light. This tandem strategy not only reduces the number of unit operations but also operates under remarkably mild conditions, typically between 20°C and 35°C, thereby preserving the integrity of sensitive functional groups. The use of aqueous DMF mixtures and benign solvents like HFIP in the second step underscores a commitment to green chemistry principles. For a reliable pharmaceutical intermediate supplier, adopting this route means transitioning from a resource-intensive extraction model to a predictable, chemically controlled manufacturing environment that ensures consistent quality and batch-to-batch reproducibility.
Mechanistic Insights into Pd-Catalyzed Coupling and Photocyclization
The success of this synthetic route hinges on the precise orchestration of two distinct catalytic cycles, beginning with the palladium-mediated formation of the carbon-carbon bond. In the first step, 6-bromo-3,4-methylenedioxybenzaldehyde undergoes oxidative addition with the Pd(0) species generated in situ from Pd(OAc)2. The presence of K2CO3 serves a dual purpose: it activates the phenylboronic acid to facilitate transmetallation and neutralizes the acidic byproducts formed during the catalytic cycle. The choice of a DMF/water solvent system is particularly ingenious, as it enhances the solubility of the inorganic base while maintaining a homogeneous reaction medium for the organic substrates. This careful balance of reagents and conditions minimizes the formation of homocoupling byproducts and ensures high conversion rates, typically achieving isolated yields of the intermediate aldehyde between 70% and 84%.
Following the construction of the biaryl framework, the second step leverages the power of photochemistry to close the final ring system through a 6π electrocyclization mechanism. Upon irradiation with purple LED light (400-420nm), the conjugated system of the intermediate absorbs photons to reach an excited state, lowering the activation energy required for ring closure. Ammonium formate acts as a crucial hydrogen source and potentially facilitates proton transfer events necessary for aromatization. The use of 1,1,1,3,3,3-hexafluoro-2-propanol (HFIP) as the solvent is critical here; its unique hydrogen-bond donating ability stabilizes polar transition states and radical intermediates, effectively suppressing side reactions. This mechanistic elegance allows the reaction to proceed at ambient temperatures, avoiding the thermal degradation often seen in traditional thermal cyclization methods, thus resulting in a cleaner impurity profile and simplified downstream processing.
How to Synthesize Trisphaeridine Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The process begins with the preparation of the biaryl aldehyde intermediate via Suzuki coupling, where controlling the molar ratios of the boronic acid and base is essential for driving the reaction to completion. Once the intermediate is isolated and purified, it is subjected to the photocyclization step in a dedicated photoreactor equipped with specific wavelength LEDs. The concentration of the substrate in HFIP must be carefully maintained between 0.05 mol/L and 0.3 mol/L to ensure efficient light penetration and reaction kinetics. Detailed standard operating procedures regarding quenching, extraction, and column chromatography are vital for recovering the final yellow solid product with the reported high purity.
- Perform Suzuki coupling of 6-bromo-3,4-methylenedioxybenzaldehyde with phenylboronic acid using Pd(OAc)2 and K2CO3 in DMF/H2O at 20-30°C to obtain the biaryl aldehyde intermediate.
- Subject the intermediate to 6π electrocyclization using ammonium formate in HFIP solvent under 400-420nm visible light irradiation at 20-35°C.
- Purify the final crude product via column chromatography using petroleum ether and ethyl acetate to achieve high-purity Trisphaeridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain; by replacing scarce natural sources with commodity chemicals like phenylboronic acid and bromo-veratraldehyde, manufacturers can mitigate the risks associated with agricultural variability and geopolitical instability. This shift ensures a continuous flow of raw materials, significantly reducing lead time for high-purity pharmaceutical intermediates. Moreover, the elimination of toxic heavy metal oxidants and the reduction of high-energy thermal steps translate directly into lower operational expenditures. The ability to run reactions at near-room temperature reduces the load on HVAC and cooling systems, contributing to substantial cost savings in utility consumption over the lifecycle of the product.
- Cost Reduction in Manufacturing: The economic impact of this route is profound, primarily driven by the reduction in step count and the use of inexpensive, commercially available reagents. Unlike traditional methods that may require expensive chiral auxiliaries or precious metal oxidants in stoichiometric amounts, this catalytic approach uses palladium in minute quantities (0.01 to 0.06 equivalents). Furthermore, the simplified workup procedure, which avoids complex distillation or recrystallization from hazardous solvents, lowers the cost of waste treatment and disposal. By streamlining the process from multiple days to a matter of hours per step, labor costs are also significantly optimized, allowing for cost reduction in API manufacturing that can be passed down to the end client.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical transformations involved. The Suzuki coupling is a well-understood industrial reaction with a vast global supply base for catalysts and ligands, ensuring that production is never halted due to single-source dependency. Additionally, the photocyclization step utilizes standard LED technology which is durable and energy-efficient, removing the reliance on specialized high-pressure lamps that often have short lifespans. This reliability ensures that delivery schedules can be met consistently, a critical factor for pharmaceutical partners who operate on tight clinical trial timelines. The scalability demonstrated in the patent, moving seamlessly from milligram to gram scales without yield loss, confirms that the process is ready for immediate commercial scale-up of complex alkaloids.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry mandates. The absence of toxic waste streams simplifies the permitting process for manufacturing facilities and reduces the liability associated with hazardous material handling. The use of water as a co-solvent in the first step and the recyclable nature of the HFIP solvent in the second step minimize the overall solvent intensity of the process. This eco-friendly profile not only enhances the corporate sustainability metrics of the manufacturer but also future-proofs the supply chain against tightening environmental regulations. The ability to scale this process to 100 kgs or even 100 MT annual production volumes without compromising safety or yield makes it an ideal candidate for long-term strategic sourcing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production pipelines. The answers provided are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the overall yield advantage of this new Trisphaeridine synthesis method?
A: The patented process achieves a total yield ranging from 27.3% to 46%, which is significant for a complex alkaloid structure, especially considering the brevity of the two-step route compared to traditional multi-step extractions.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates successful gram-scale preparation (Example 4) with yields comparable to milligram scales, indicating robust scalability for commercial manufacturing without negative efficiency impacts.
Q: How does the visible-light step improve environmental compliance?
A: By utilizing visible light (400-420nm) and ammonium formate instead of harsh oxidants or high-temperature thermal cyclization, the process eliminates toxic waste generation and reduces energy consumption, aligning with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trisphaeridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of Trisphaeridine as a next-generation anticancer agent and are fully prepared to support its development from bench to bedside. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with advanced photoreactors and stringent purity specifications to handle sensitive photochemical transformations like the one described in CN114773354B. With our rigorous QC labs and commitment to quality, we guarantee that every batch of Trisphaeridine meets the highest international standards for pharmaceutical intermediates, providing you with the confidence to advance your drug discovery programs.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, high-quality supply of this critical oncology intermediate.
