Advanced Clean Production of Tetrabenzylthiuram Disulfide for High-Performance Rubber Applications
The global rubber industry is undergoing a significant transformation driven by stringent environmental regulations and the demand for safer processing aids. Patent CN110256318B introduces a groundbreaking clean production method for tetrabenzylthiuram disulfide (TBzTD), a high-performance vulcanization accelerator that eliminates nitrosamine hazards. This technology represents a paradigm shift from traditional oxidation methods, utilizing a two-step reaction sequence that allows for the complete recycling of mother liquor. By integrating condensation and oxidation in separate vessels with intermediate isolation, the process achieves near-zero wastewater discharge while maintaining exceptional product quality. For R&D directors and procurement leaders, this patent offers a viable pathway to sustainable manufacturing without compromising on the technical specifications required for premium rubber compounds. The ability to recycle solvents and reactants not only aligns with green chemistry principles but also fundamentally alters the cost structure of producing complex thiuram accelerators.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of thiuram accelerators has relied on oxidation methods involving sodium nitrite, chlorine, or electrolytic processes, all of which carry substantial environmental and operational burdens. Traditional sodium nitrite-air oxidation and chlorine-air oxidation methods generate significant amounts of inorganic salts and acidic wastewater, necessitating expensive treatment facilities to meet discharge standards. Furthermore, these processes often suffer from yields not exceeding 90%, as valuable intermediates remain dissolved in the waste mother liquor and are lost forever. The use of chlorine gas poses severe safety risks, requiring specialized corrosion-resistant equipment and rigorous safety protocols to prevent toxic leaks. Electrolytic oxidation, while cleaner, demands high energy consumption and complex technical operations, making it economically unviable for many production sites. These legacy methods create a bottleneck for manufacturers seeking to scale production sustainably, as the cost of waste treatment and raw material loss erodes profit margins and supply chain reliability.
The Novel Approach
The innovative method disclosed in the patent overcomes these deficiencies by decoupling the condensation and oxidation reactions, allowing for precise control over each stage and the recovery of byproducts. Instead of a one-pot reaction where waste accumulates, this approach isolates the dibenzyl dithiocarbamate intermediate through crystallization at 0°C, enabling the mother liquor to be reused as the solvent for the next batch. The subsequent oxidation step utilizes hydrogen peroxide in an aqueous medium, avoiding hazardous organic solvents and producing only water and sodium sulfate as byproducts. This separation strategy ensures that unreacted dibenzylamine and intermediate salts are not discarded but are cycled back into the process, driving raw material utilization to theoretical maximums. The result is a robust, scalable process that eliminates the need for complex wastewater treatment infrastructure while delivering a product with superior purity and consistent physical properties. This structural change in the synthesis route provides a competitive edge for suppliers aiming to offer cost-effective and environmentally compliant rubber additives.
Mechanistic Insights into Two-Step Oxidation and Recycling
The core of this technology lies in the precise manipulation of reaction equilibria during the condensation phase, where dibenzylamine reacts with carbon disulfide in the presence of an alkali. The reaction is conducted in water, with carbon disulfide added dropwise at a controlled rate of 9-15 ml/min to manage exothermic heat and ensure complete conversion. Following the reaction, the solution is cooled to approximately 0°C, inducing the crystallization of the sodium dibenzyl dithiocarbamate intermediate while leaving impurities and unreacted materials in the solution. This crystallization step is critical for purity control, as it physically separates the desired intermediate from the reaction matrix before oxidation occurs. The mother liquor, rich in unreacted alkali and amine, is not treated as waste but is directly recycled into the next condensation batch, acting as both solvent and reactant carrier. This closed-loop mechanism minimizes the introduction of fresh water and chemicals, significantly reducing the volumetric load on the production system.
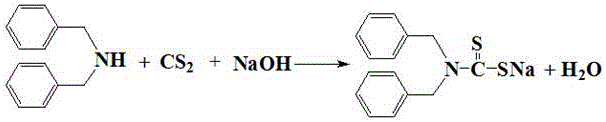
Following the isolation of the intermediate, the process moves to the oxidation phase, where the dibenzyl dithiocarbamate is dissolved and subjected to acid neutralization and oxidative coupling. The neutralization is carefully controlled using sulfuric acid to maintain a pH of 2 to 3 at temperatures between 8-13°C, preventing premature decomposition or side reactions. Hydrogen peroxide is then introduced dropwise as the oxidant, facilitating the formation of the disulfide bond without generating toxic nitrogen oxides or chlorinated byproducts. The reaction mixture is subsequently heated to 30-35°C to ensure complete conversion, after which the final tetrabenzylthiuram disulfide product is separated via filtration. Crucially, the mother liquor from this oxidation step, containing dissolved sodium sulfate and trace organics, is also recycled to dissolve the intermediate in the next cycle. This dual-recycling strategy ensures that the process operates with minimal fresh water intake and zero liquid discharge, setting a new standard for clean chemical manufacturing.
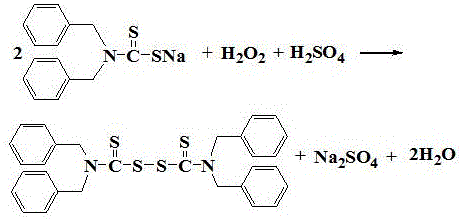
How to Synthesize Tetrabenzylthiuram Disulfide Efficiently
Implementing this synthesis route requires strict adherence to temperature profiles and addition rates to maximize the efficiency of the mother liquor recycling loop. The process begins with the preparation of the reaction vessel where dibenzylamine and alkali are mixed, followed by the controlled addition of carbon disulfide to form the intermediate salt. Operators must monitor the crystallization temperature closely to ensure optimal recovery of the solid intermediate, as this dictates the purity of the final accelerator. The subsequent oxidation step demands precise pH control and slow addition of hydrogen peroxide to manage the exotherm and prevent localized overheating which could degrade the product. Detailed standard operating procedures regarding the ratio of recycled mother liquor to fresh water are essential to maintain the balance of the system over multiple batches. For a comprehensive guide on the specific operational parameters and safety protocols required for this synthesis, please refer to the technical breakdown below.
- React dibenzylamine with alkali and carbon disulfide in water, cool to 0°C to crystallize the intermediate, and separate the mother liquor for recycling.
- Dissolve the intermediate in water, neutralize with acid at 8-13°C, and oxidize using hydrogen peroxide while maintaining strict temperature control.
- Separate the final solid product and recycle the oxidation mother liquor back into the dissolution step for the next batch to ensure zero wastewater discharge.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this clean production method offers profound advantages for procurement managers and supply chain heads looking to optimize costs and mitigate risks. The ability to recycle mother liquor in both reaction steps drastically reduces the consumption of raw materials such as dibenzylamine and carbon disulfide, leading to substantial cost savings in variable production expenses. By eliminating the generation of hazardous wastewater, manufacturers can avoid the high capital and operational expenditures associated with environmental compliance and waste treatment facilities. The use of water as the primary solvent and hydrogen peroxide as the oxidant enhances process safety, reducing the insurance premiums and safety infrastructure costs linked to handling toxic gases or flammable organic solvents. Furthermore, the high yield and purity achieved through this method minimize the need for reprocessing or refining, streamlining the production timeline and ensuring consistent supply availability. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-performance rubber additives.
- Cost Reduction in Manufacturing: The implementation of a closed-loop mother liquor recycling system fundamentally changes the cost dynamics of thiuram accelerator production. By recovering unreacted raw materials and intermediates from the waste stream, the process significantly lowers the effective cost per kilogram of the final product. The elimination of expensive waste treatment processes and the reduction in fresh water consumption further contribute to a leaner cost structure. Additionally, the high selectivity of the hydrogen peroxide oxidation reduces the formation of byproducts that would otherwise require costly purification steps. This efficiency allows suppliers to offer competitive pricing while maintaining healthy margins, providing a distinct economic advantage in the global market for rubber chemicals.
- Enhanced Supply Chain Reliability: The simplified process flow and the use of readily available, non-hazardous raw materials enhance the overall reliability of the supply chain. Unlike methods relying on chlorine gas or specialized electrolytic equipment, this technology can be implemented in standard chemical reactors with minimal risk of shutdown due to safety incidents. The robustness of the recycling system ensures that production can continue smoothly even if there are minor fluctuations in raw material quality, as the process is designed to tolerate and recover variations. This stability is crucial for long-term supply contracts, as it guarantees consistent delivery schedules and reduces the likelihood of force majeure events disrupting the flow of critical rubber additives to downstream manufacturers.
- Scalability and Environmental Compliance: Scaling this technology from pilot to commercial production is straightforward due to the absence of complex unit operations or hazardous intermediates. The process is inherently green, meeting stringent international environmental standards without the need for end-of-pipe treatment solutions. This compliance future-proofs the manufacturing asset against tightening regulations on industrial emissions and wastewater discharge. The ability to operate with zero liquid discharge makes the facility attractive for investment in regions with strict environmental zoning. Consequently, the technology supports rapid capacity expansion to meet growing demand for nitrosamine-free accelerators, ensuring that supply can scale in tandem with the automotive and tire industries' recovery and growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this clean synthesis technology. These answers are derived directly from the patent data and practical considerations for industrial application, providing clarity for stakeholders evaluating this method. Understanding the nuances of the recycling mechanism and the quality parameters of the final product is essential for making informed procurement and R&D decisions. The insights provided here aim to bridge the gap between theoretical patent claims and practical manufacturing realities.
Q: How does the mother liquor recycling process impact the overall yield of tetrabenzylthiuram disulfide?
A: By recycling the mother liquor from both the condensation and oxidation steps, unreacted raw materials and dissolved intermediates are recovered. This mechanism pushes the yield close to 100%, significantly higher than traditional methods where mother liquor is discarded as waste.
Q: What are the safety advantages of using hydrogen peroxide over chlorine or nitrite oxidation?
A: Hydrogen peroxide oxidation eliminates the need for hazardous chlorine gas or sodium nitrite, reducing the risk of toxic gas release and equipment corrosion. The reaction byproducts are primarily water and sodium sulfate, aligning with green chemical safety standards.
Q: Does this clean production method affect the purity and physical properties of the final accelerator?
A: Yes, the method produces tetrabenzylthiuram disulfide with purity exceeding 98% and a melting point of 129-134°C. The stepwise crystallization effectively removes impurities, ensuring the product is free from nitrosamines and suitable for high-end rubber applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrabenzylthiuram Disulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting sustainable and efficient manufacturing technologies to meet the evolving needs of the global rubber industry. Our team of expert engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN110256318B are translated into reliable industrial reality. We are committed to delivering high-purity tetrabenzylthiuram disulfide that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our expertise in green chemistry and process optimization, we help our partners secure a stable supply of high-performance rubber additives that comply with the most demanding environmental and safety standards.
We invite procurement leaders and technical directors to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce overall manufacturing costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our solutions align perfectly with your production goals. Together, we can drive the adoption of cleaner, more efficient chemical manufacturing practices that benefit both your bottom line and the environment.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
