Advanced Sulfuric Acid-Assisted Chlorination for High-Purity Photoinitiator Intermediates and Commercial Scale-Up
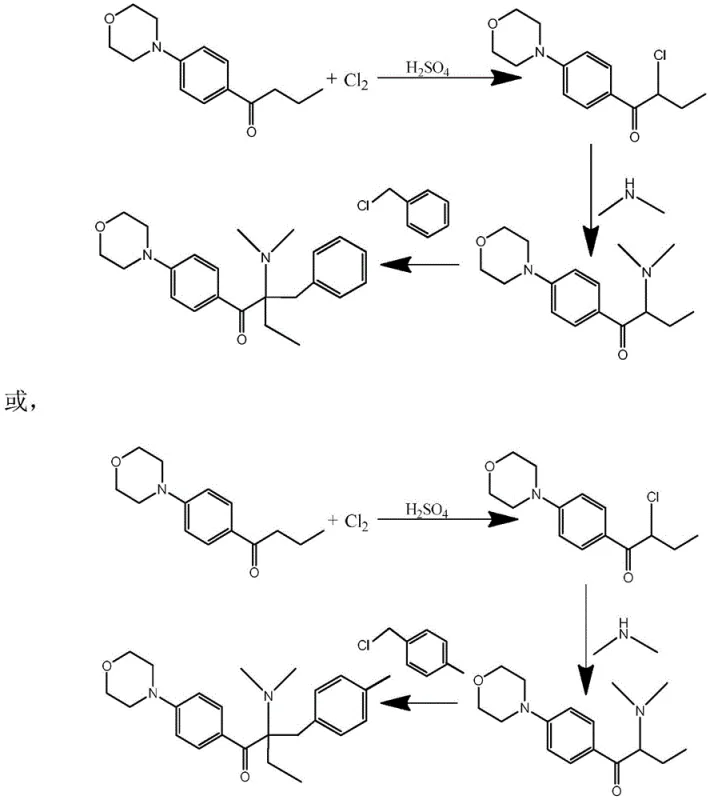
The chemical industry is constantly seeking more efficient pathways to synthesize critical intermediates, particularly for the high-value sector of UV-curing materials and photoinitiators. A significant technological breakthrough in this domain is detailed in patent CN114315760A, which discloses a novel preparation method for 2-chloro-1-(4-morpholinylphenyl)-1-butanone. This compound serves as a pivotal precursor for advanced alpha-aminoketone photoinitiators, widely utilized in coatings and inks for their superior thermal stability and curing speed. The core innovation lies in the strategic use of sulfuric acid as an auxiliary agent during the chlorination of 1-(4-morpholinylphenyl)-1-butanone. Unlike traditional methods that struggle with selectivity issues or rely on expensive brominating agents, this process leverages specific acid concentrations ranging from 30% to 85% to modulate the electronic environment of the substrate. This adjustment not only enhances the yield of the alpha-chlorinated product but also significantly mitigates the formation of undesirable ring-substituted byproducts, representing a substantial leap forward in process chemistry for reliable photoinitiator intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-aminoketone photoinitiators such as 2-benzyl-2-dimethylamino-1-(4-morpholinophenyl)-1-butanone has relied heavily on bromination processes. While bromination offers reasonable selectivity due to the lower reactivity of bromine compared to chlorine, it presents severe economic and operational drawbacks for large-scale manufacturing. The cost of elemental bromine is significantly higher than that of chlorine, directly impacting the raw material expenditure. Furthermore, the recovery and recycling of bromine are technically complex, often requiring specialized equipment to handle corrosive byproducts like hydrogen bromide. In attempts to switch to cheaper chlorination routes, manufacturers have historically faced the challenge of poor regioselectivity. The morpholine group attached to the phenyl ring is inherently electron-donating, which activates the aromatic ring towards electrophilic attack. Consequently, without precise control, chlorine tends to substitute onto the benzene ring rather than the desired alpha-position of the ketone chain, leading to a complex mixture of isomers that are difficult and costly to separate, ultimately compromising the purity and yield required for high-performance applications.
The Novel Approach
The methodology described in patent CN114315760A fundamentally alters this landscape by introducing a controlled acidic environment that dictates the reaction pathway. By employing sulfuric acid with a concentration specifically tuned between 30% and 85%, the process effectively suppresses the competing ring-chlorination side reactions. This approach allows for the use of inexpensive chlorine gas while achieving selectivity profiles that rival or exceed those of bromination. The result is a streamlined synthesis where the target alpha-chloro ketone is produced with high fidelity, minimizing downstream purification burdens. This novel route not only reduces the direct cost of halogenating agents but also simplifies the waste treatment profile, as the acidic byproducts are easier to manage and recycle compared to brominated waste streams. For procurement teams focused on cost reduction in photoinitiator manufacturing, this shift from bromine to acid-assisted chlorine represents a strategic opportunity to optimize the supply chain for these critical specialty chemicals.
Mechanistic Insights into Sulfuric Acid-Assisted Chlorination
To fully appreciate the technical sophistication of this process, one must examine the electronic interactions occurring at the molecular level during the reaction. The key to the improved selectivity lies in the protonation of the morpholine nitrogen atom by the sulfuric acid. Under neutral or basic conditions, the lone pair of electrons on the nitrogen atom participates in resonance with the benzene ring, acting as a strong electron-donating group that activates the ortho and para positions for electrophilic aromatic substitution. However, in the presence of concentrated sulfuric acid, the nitrogen is protonated to form an ammonium species. This transformation converts the morpholine group from an electron-donating activator into an electron-withdrawing deactivator. This electronic reversal dramatically reduces the electron density on the benzene ring, rendering it far less susceptible to attack by the electrophilic chlorine species. Consequently, the chlorination reaction is directed almost exclusively towards the enolizable alpha-position of the ketone side chain, where the reaction kinetics remain favorable.
This mechanistic control is further refined by the specific concentration range of the sulfuric acid and the temperature parameters maintained between 75°C and 85°C. If the acid concentration is too low, the protonation equilibrium is insufficient to fully deactivate the ring, leading to persistent byproduct formation. Conversely, if the concentration is too high, solubility issues may arise, or the acid may promote other degradation pathways. The patent data indicates that maintaining the acid concentration within the 30% to 85% window ensures an optimal balance where the electronic effect is maximized without compromising the physical handling of the reaction mixture. Furthermore, the introduction of chlorine gas is managed with a specific profile—rapid initially to drive the reaction, then slowed to prevent over-chlorination or dichloro-substitution. This precise control over reaction variables ensures that the impurity profile remains minimal, facilitating the production of high-purity intermediates essential for sensitive photochemical applications.
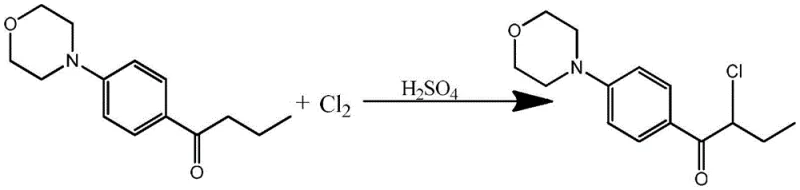
How to Synthesize 2-Chloro-1-(4-morpholinylphenyl)-1-butanone Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and phase separation steps outlined in the patent documentation. The process begins with the mixing of the ketone substrate and the sulfuric acid auxiliary agent, followed by the controlled introduction of chlorine gas under heated conditions. Monitoring the reaction progress via liquid chromatography is crucial to determine the endpoint and prevent over-reaction. Once the chlorination is complete, the workup procedure involves the addition of an organic solvent, such as toluene or dichloromethane, along with a calculated amount of water. This step is critical for inducing phase separation, allowing the sulfuric acid layer to be isolated and potentially recycled, while the organic layer retains the product. The detailed standardized synthesis steps, including specific mass ratios and temperature ramps, are provided below to guide process engineers in replicating this high-efficiency protocol.
- Mix 1-(4-morpholinylphenyl)-1-butanone with 30%-85% sulfuric acid and heat to 75-85°C.
- Introduce chlorine gas rapidly at first, then slowly, maintaining temperature to achieve alpha-chlorination.
- Add organic solvent and water to separate the sulfuric acid phase (30%-70% concentration) for recycling.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the procurement of fine chemical intermediates, the transition to this sulfuric acid-assisted chlorination process offers compelling economic and logistical benefits. The primary advantage stems from the substitution of expensive bromine with commodity-grade chlorine gas, which drastically lowers the raw material cost basis. Additionally, the ability to recycle the sulfuric acid phase reduces the consumption of auxiliary reagents and minimizes the volume of hazardous waste requiring disposal. This aligns with modern green chemistry principles and reduces the environmental compliance burden on manufacturing facilities. The simplified purification process, resulting from higher reaction selectivity, also translates to reduced solvent usage and shorter cycle times, enhancing overall plant throughput. These factors combined create a robust supply chain model that is less vulnerable to price volatility in the halogen market and more resilient to regulatory changes regarding waste management.
- Cost Reduction in Manufacturing: The elimination of high-cost brominating agents and the implementation of an acid-recycling loop significantly lower the variable costs associated with production. By avoiding the complex recovery systems needed for bromine and reducing the load on purification columns due to fewer byproducts, the overall cost of goods sold is substantially decreased. This economic efficiency allows for more competitive pricing structures in the final photoinitiator market without sacrificing margin.
- Enhanced Supply Chain Reliability: Chlorine and sulfuric acid are bulk commodities with stable and widespread availability, unlike specialized brominating reagents which may face supply constraints. Utilizing these common chemicals ensures a steady flow of raw materials, reducing the risk of production stoppages. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in feedstock quality, ensuring consistent output and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor configurations and separation techniques familiar to chemical engineers. The ability to separate and reuse the sulfuric acid phase minimizes the generation of spent acid waste, a major environmental concern in halogenation chemistry. This reduction in waste volume simplifies effluent treatment and supports sustainability goals, making the technology attractive for expansion in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for commercial production. The answers are derived directly from the experimental data and technical disclosures found within the patent specification, providing a reliable foundation for decision-making.
Q: Why is sulfuric acid critical in this chlorination process?
A: Sulfuric acid protonates the morpholine nitrogen, converting it from an electron-donating group to an electron-withdrawing group. This electronic modulation suppresses unwanted electrophilic substitution on the benzene ring, thereby drastically improving the selectivity for the desired alpha-chlorinated product.
Q: Can the sulfuric acid be recycled in this process?
A: Yes, the process is designed for sustainability. By adjusting the water content post-reaction, the sulfuric acid phase concentration is maintained between 30% and 70%, allowing for clear phase separation from the organic layer and enabling the acid to be reused in subsequent batches.
Q: What purity levels can be achieved with this method?
A: The optimized chlorination followed by standard downstream processing yields intermediates with purity exceeding 99%. This high purity is essential for the subsequent synthesis of high-performance photoinitiators like Irgacure 369 analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-1-(4-morpholinylphenyl)-1-butanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final photochemical products. Our technical team has extensively analyzed the pathway described in CN114315760A and possesses the expertise to scale this sophisticated chlorination process from laboratory benchtop to full commercial production. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of 2-chloro-1-(4-morpholinylphenyl)-1-butanone meets the exacting standards required for high-end photoinitiator synthesis.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your operational efficiency and product quality.
