Advanced NHC-Catalyzed Synthesis of 3-Arylcoumarins for High-Purity Pharmaceutical Intermediates
Advanced NHC-Catalyzed Synthesis of 3-Arylcoumarins for High-Purity Pharmaceutical Intermediates
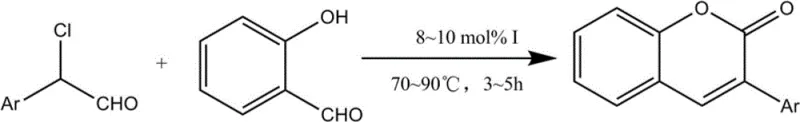
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for greener, safer, and more efficient synthetic routes. A pivotal advancement in this domain is detailed in patent CN102757416B, which discloses a novel method for synthesizing 3-arylcoumarins utilizing N-heterocyclic carbene (NHC) organocatalysis. This technology represents a significant departure from legacy methods, offering a metal-free pathway that aligns perfectly with modern regulatory standards for pharmaceutical intermediates. By leveraging the unique ability of NHCs to invert the polarity of aldehydes (Umpolung), this process enables the direct coupling of readily available 2-chloro-2-arylacetaldehydes and salicylaldehydes. For R&D directors and procurement specialists alike, this patent offers a compelling value proposition: a robust, scalable, and environmentally benign route to high-value coumarin scaffolds that are essential in drug discovery and agrochemical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-arylcoumarins has relied heavily on the Perkin reaction or variations involving diazonium salts, both of which present substantial operational and safety challenges for industrial scale-up. The Perkin reaction, while conceptually simple, typically necessitates the use of strong bases and high-temperature reflux conditions, often leading to harsh reaction environments that can degrade sensitive functional groups and generate complex impurity profiles. Alternatively, methods employing diazonium salts, such as those reported by Lesya, suffer from severe stability issues; these reagents are notoriously unstable in dry states and pose significant explosion risks upon heating or impact. Consequently, these traditional pathways not only compromise operator safety but also result in difficult post-treatment procedures due to the formation of numerous by-products, ultimately inflating the cost of goods and extending production lead times for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in patent CN102757416B introduces a transformative approach that circumvents these historical bottlenecks through the use of biomimetic N-heterocyclic carbene catalysts. This novel strategy operates under mild, weakly alkaline conditions, typically utilizing triethylamine as a base, thereby eliminating the need for corrosive strong acids or bases. Crucially, the process is entirely metal-free, which inherently solves the persistent industry challenge of removing trace heavy metal residues—a step that is both costly and technically demanding in API manufacturing. By employing easily preparable starting materials like 2-chloro-2-arylacetaldehyde and salicylaldehyde, the reaction proceeds smoothly in ethyl acetate, a green solvent, to deliver 3-arylcoumarins with high efficiency. This shift not only simplifies the workflow but also drastically reduces the environmental footprint, positioning it as an ideal candidate for sustainable cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into NHC-Catalyzed Cyclization
The core innovation of this synthesis lies in the sophisticated mechanistic role played by the N-heterocyclic carbene (NHC) catalyst, which fundamentally alters the reactivity of the aldehyde substrates. Under the action of a base, the NHC precursor generates the active carbene species in situ, which then attacks the carbonyl carbon of the salicylaldehyde. This interaction triggers a polarity inversion, known as Umpolung, converting the traditionally electrophilic carbonyl carbon into a nucleophilic acyl anion equivalent. This nucleophilic species is then poised to attack the electrophilic center of the 2-chloro-2-arylacetaldehyde. This unique activation mode allows for the construction of the carbon-carbon bond under exceptionally mild conditions that would be inaccessible via conventional enolate chemistry. The subsequent intramolecular cyclization and elimination of the chloride ion finalize the formation of the coumarin lactone ring, showcasing the elegance of organocatalysis in building complex heterocyclic systems.
Beyond the primary bond formation, the reaction design incorporates strategic elements to control impurities and drive conversion. The inclusion of 4Å molecular sieves serves a dual purpose: acting as a desiccant to sequester water produced during the condensation and shifting the equilibrium towards the product side according to Le Chatelier's principle. This ensures that the reaction proceeds to completion without the need for excessive reagent loading or prolonged reaction times. Furthermore, the choice of a triazole-based NHC precursor provides a stable yet reactive catalytic cycle that minimizes catalyst decomposition, a common issue in organocatalysis that can lead to colored impurities. The result is a clean reaction profile that facilitates straightforward downstream processing, such as simple filtration and concentration, yielding products with high purity suitable for direct use in subsequent synthetic steps or rigorous QC testing.
How to Synthesize 3-Phenylcoumarin Efficiently
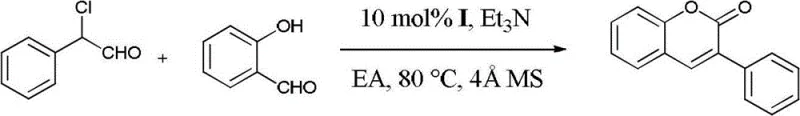
To translate this patented technology into practical application, the synthesis of 3-phenylcoumarin serves as an exemplary model for the broader class of 3-arylcoumarins. The protocol is designed for operational simplicity, requiring standard laboratory equipment such as Schlenk tubes or sealed reactors capable of maintaining an inert atmosphere. The process begins with the careful preparation of the catalytic system, where the NHC precursor and drying agents are loaded prior to the introduction of liquid reagents. This sequence ensures that the active catalyst is generated in a controlled environment, maximizing its turnover number. The use of anhydrous ethyl acetate as the solvent not only dissolves the organic substrates effectively but also aligns with green chemistry principles, replacing more hazardous chlorinated solvents often used in legacy coumarin syntheses.
- Load the NHC precursor (e.g., triazole type) and 4Å molecular sieves into a sealed reaction vessel under an inert nitrogen atmosphere.
- Dissolve 2-chloro-2-arylacetaldehyde and salicylaldehyde in anhydrous ethyl acetate (EA) to form the reactant solution.
- Inject the reactant solution and triethylamine into the vessel, then heat the mixture to 70-90°C for 3-5 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-catalyzed route offers tangible strategic advantages that extend beyond mere chemical yield. The elimination of transition metal catalysts removes the necessity for expensive and time-consuming metal scavenging steps, which are often required to meet strict regulatory limits for residual metals in pharmaceutical ingredients. This simplification of the purification train directly translates to reduced processing time and lower consumption of specialized resins or adsorbents. Additionally, the use of mild reaction conditions (70-90°C) compared to the high-temperature reflux of traditional methods implies lower energy consumption per batch, contributing to a more sustainable and cost-effective manufacturing profile. The robustness of the reaction across a wide range of substrates also means that a single standardized protocol can be used to produce a diverse library of derivatives, streamlining inventory management and reducing the complexity of raw material sourcing.
- Cost Reduction in Manufacturing: The metal-free nature of this catalytic system fundamentally alters the cost structure of production by obviating the need for precious metal catalysts and the associated validation of cleaning procedures to remove metal traces. By utilizing inexpensive and commercially available organic bases like triethylamine and common solvents like ethyl acetate, the raw material costs are kept low. Furthermore, the high atom economy and the ability to drive reactions to completion using molecular sieves reduce the loss of valuable starting materials, ensuring that the overall cost of goods sold is significantly optimized without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous reagents such as 2-chloro-2-arylacetaldehydes and salicylaldehydes mitigates the supply chain risks associated with unstable intermediates like diazonium salts. These starting materials are widely available from multiple global suppliers, ensuring continuity of supply even in volatile market conditions. The simplified workup procedure, which involves basic filtration and concentration rather than complex extractions or chromatography, reduces the dependency on specialized processing equipment and skilled labor, thereby enhancing the overall resilience and flexibility of the manufacturing supply chain.
- Scalability and Environmental Compliance: From an environmental compliance perspective, this process offers a distinct advantage by generating minimal hazardous waste. The absence of heavy metals simplifies wastewater treatment protocols, and the use of ethyl acetate allows for easier solvent recovery and recycling. The reaction's tolerance to various functional groups means that protection and deprotection steps can often be avoided, shortening the synthetic sequence and reducing the total volume of chemical waste generated per kilogram of product. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this NHC-catalyzed synthesis, derived directly from the experimental data and specifications provided in the patent documentation. Understanding these nuances is critical for process chemists evaluating the feasibility of this route for their specific project needs. The answers reflect the optimized conditions found to provide the best balance between reaction rate, yield, and product purity.
Q: What are the advantages of using NHC catalysis over traditional Perkin reactions for coumarin synthesis?
A: Unlike the traditional Perkin reaction which requires harsh conditions like strong bases and high temperatures, the NHC-catalyzed method operates under mild weakly alkaline conditions (70-90°C). Furthermore, it eliminates the need for toxic metal catalysts, ensuring a cleaner product profile free from heavy metal residues, which is critical for pharmaceutical applications.
Q: What is the role of 4Å molecular sieves in this synthesis protocol?
A: The 4Å molecular sieves act as a highly effective desiccant within the reaction system. They possess a strong water absorption capacity that thoroughly removes water generated during the condensation process. This drives the chemical equilibrium towards the product side, significantly accelerating the reaction rate and improving overall yield without requiring complex azeotropic distillation setups.
Q: Can this method accommodate diverse aryl substituents on the aldehyde substrate?
A: Yes, the protocol demonstrates excellent substrate universality. It successfully accommodates various substituents including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like chloro, bromo, and fluoro. Even bulky substrates like naphthyl groups are tolerated, yielding products with high efficiency, making it versatile for generating diverse chemical libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylcoumarin Supplier
The technological potential of NHC-catalyzed synthesis for 3-arylcoumarins is immense, offering a pathway to high-quality intermediates that meet the rigorous demands of the global pharmaceutical industry. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale protocols like this are seamlessly translated into robust industrial processes. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of 3-arylcoumarin delivered meets the highest standards of quality and consistency required for API synthesis.
We invite you to engage with our technical procurement team to discuss how this metal-free technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our advanced manufacturing capabilities can support your R&D and commercialization goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
